+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1223 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | The dynamics of signal triggering in a gp130-receptor complex. | |||||||||
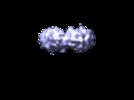
 Map data Map data | This is a cEM map of the extracellular portion of the IL11 hexameric complex | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 30.0 Å | |||||||||
 Authors Authors | Matadeen R / Hon W / Jones EY / Fuller S | |||||||||
 Citation Citation |  Journal: Structure / Year: 2007 Journal: Structure / Year: 2007Title: The dynamics of signal triggering in a gp130-receptor complex. Authors: Rishi Matadeen / Wai-Ching Hon / John K Heath / E Yvonne Jones / Stephen Fuller /  Abstract: gp130 is a shared signal-transducing membrane-associated receptor for several hematopoietic cytokines. The 30 A resolution cryo-electron microscopy (cryo-EM) structure of the Interleukin 11(IL-11)-IL- ...gp130 is a shared signal-transducing membrane-associated receptor for several hematopoietic cytokines. The 30 A resolution cryo-electron microscopy (cryo-EM) structure of the Interleukin 11(IL-11)-IL-11 Receptor-gp130 extracellular complex reveals the architecture and dynamics of this gp130-containing signaling complex. Normal-mode analysis reveals a repertoire of conformational changes that could function in signal triggering. This suggests a concerted mechanism of signaling involving all the components of the complex. This could provide a general mechanism of signal transfer for cytokines utilizing the JAK-STAT signaling cascade. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1223.map.gz emd_1223.map.gz | 499.2 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1223-v30.xml emd-1223-v30.xml emd-1223.xml emd-1223.xml | 10 KB 10 KB | Display Display |  EMDB header EMDB header |
| Images |  1223.gif 1223.gif | 6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1223 http://ftp.pdbj.org/pub/emdb/structures/EMD-1223 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1223 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1223 | HTTPS FTP |
-Related structure data
| Similar structure data |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_1223.map.gz / Format: CCP4 / Size: 1.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1223.map.gz / Format: CCP4 / Size: 1.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is a cEM map of the extracellular portion of the IL11 hexameric complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 3.3 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : IL11-IL11-R-gp130
| Entire | Name: IL11-IL11-R-gp130 |
|---|---|
| Components |
|
-Supramolecule #1000: IL11-IL11-R-gp130
| Supramolecule | Name: IL11-IL11-R-gp130 / type: sample / ID: 1000 / Oligomeric state: hexamer / Number unique components: 3 |
|---|---|
| Molecular weight | Theoretical: 270 KDa |
-Macromolecule #1: Interleukin-11
| Macromolecule | Name: Interleukin-11 / type: protein_or_peptide / ID: 1 / Name.synonym: IL-11 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
-Macromolecule #2: Interleukin-11 Receptor
| Macromolecule | Name: Interleukin-11 Receptor / type: protein_or_peptide / ID: 2 / Name.synonym: IL-11R / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
-Macromolecule #3: Glycoprotein 130
| Macromolecule | Name: Glycoprotein 130 / type: protein_or_peptide / ID: 3 / Name.synonym: gp130 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7.5 Details: 5mM HEPES 100mM NaCl 1mM DTT 0.25% B-octylglucopyranoside |
| Grid | Details: 300 mesh holy carbon |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % |
- Electron microscopy
Electron microscopy
| Microscope | FEI/PHILIPS CM200FEG |
|---|---|
| Alignment procedure | Legacy - Astigmatism: objective lens astigmatism was corrected at 100,000 times magnification |
| Image recording | Category: CCD / Film or detector model: KODAK SO-163 FILM / Digitization - Sampling interval: 8.3 µm / Number real images: 15 / Bits/pixel: 8 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 50100 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2 mm / Nominal defocus max: 5.0 µm / Nominal defocus min: 3.0 µm / Nominal magnification: 50000 |
| Sample stage | Specimen holder: side entry / Specimen holder model: GATAN LIQUID NITROGEN |
- Image processing
Image processing
| CTF correction | Details: Each particle |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C2 (2 fold cyclic) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 30.0 Å / Resolution method: OTHER / Software - Name: Imagic / Number images used: 830 |
| Final two d classification | Number classes: 100 |
-Atomic model buiding 1
| Software | Name: URO |
|---|---|
| Details | Protocol: Rigid Body |
| Refinement | Space: RECIPROCAL / Protocol: RIGID BODY FIT |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















