[English] 日本語
 Yorodumi
Yorodumi- EMDB-1079: Electron crystallography reveals the structure of metarhodopsin I. -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1079 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Electron crystallography reveals the structure of metarhodopsin I. | |||||||||
 Map data Map data | Density map of a rhodopsin photostationary state, highly enriched in metarhodopsin I | |||||||||
 Sample Sample |
| |||||||||
| Function / homology | G protein-coupled opsin signaling pathway => GO:0016056 / Rhodopsin Function and homology information Function and homology information | |||||||||
| Biological species |  | |||||||||
| Method | electron crystallography / cryo EM / Resolution: 5.5 Å | |||||||||
 Authors Authors | Ruprecht JJ / Mielke T / Vogel R / Villa C / Schertler GFX | |||||||||
 Citation Citation |  Journal: EMBO J / Year: 2004 Journal: EMBO J / Year: 2004Title: Electron crystallography reveals the structure of metarhodopsin I. Authors: Jonathan J Ruprecht / Thorsten Mielke / Reiner Vogel / Claudio Villa / Gebhard F X Schertler /  Abstract: Rhodopsin is the prototypical G protein-coupled receptor, responsible for detection of dim light in vision. Upon absorption of a photon, rhodopsin undergoes structural changes, characterised by ...Rhodopsin is the prototypical G protein-coupled receptor, responsible for detection of dim light in vision. Upon absorption of a photon, rhodopsin undergoes structural changes, characterised by distinct photointermediates. Currently, only the ground-state structure has been described. We have determined a density map of a photostationary state highly enriched in metarhodopsin I, to a resolution of 5.5 A in the membrane plane, by electron crystallography. The map shows density for helix 8, the cytoplasmic loops, the extracellular plug, all tryptophan residues, an ordered cholesterol molecule and the beta-ionone ring. Comparison of this map with X-ray structures of the ground state reveals that metarhodopsin I formation does not involve large rigid-body movements of helices, but there is a rearrangement close to the bend of helix 6, at the level of the retinal chromophore. There is no gradual build-up of the large conformational change known to accompany metarhodopsin II formation. The protein remains in a conformation similar to that of the ground state until late in the photobleaching process. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1079.map.gz emd_1079.map.gz | 14.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1079-v30.xml emd-1079-v30.xml emd-1079.xml emd-1079.xml | 12.3 KB 12.3 KB | Display Display |  EMDB header EMDB header |
| Images |  1079.gif 1079.gif | 73.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1079 http://ftp.pdbj.org/pub/emdb/structures/EMD-1079 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1079 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1079 | HTTPS FTP |
-Related structure data
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_1079.map.gz / Format: CCP4 / Size: 18.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1079.map.gz / Format: CCP4 / Size: 18.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Density map of a rhodopsin photostationary state, highly enriched in metarhodopsin I | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 18 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Bovine Metarhodopsin I
| Entire | Name: Bovine Metarhodopsin I |
|---|---|
| Components |
|
-Supramolecule #1000: Bovine Metarhodopsin I
| Supramolecule | Name: Bovine Metarhodopsin I / type: sample / ID: 1000 Details: Illuminated to form a photostationary state highly enriched in metarhodopsin I Oligomeric state: monomer / Number unique components: 1 |
|---|---|
| Molecular weight | Experimental: 39 KDa / Theoretical: 39 KDa |
-Macromolecule #1: Rhodopsin
| Macromolecule | Name: Rhodopsin / type: protein_or_peptide / ID: 1 / Name.synonym: Rh / Details: Illuminated to form photostationary state / Number of copies: 1 / Oligomeric state: Monomer / Recombinant expression: No / Database: NCBI |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Experimental: 39 KDa / Theoretical: 39 KDa |
| Sequence | GO: G protein-coupled opsin signaling pathway => GO:0016056 / InterPro: Rhodopsin |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | electron crystallography |
| Aggregation state | 2D array |
- Sample preparation
Sample preparation
| Concentration | 0.5 mg/mL |
|---|---|
| Buffer | pH: 7 Details: 20mM HEPES pH 7, 100 mM NaCl, 10 mM MgCl2, 3 mM NaN3, 4 mM DTT, 4 mM mercaptoethanol, 2.5 % (v/v) isopropanol |
| Grid | Details: 300 mesh molybdenum grid |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 75 % / Chamber temperature: 77 K / Instrument: GATAN CRYOPLUNGE 3 Details: Vitrification instrument: Gatan cryoplunge. Vitrification carried out in temperature and humidity-controlled chamber Timed resolved state: Vitrified after illumination of specimen Method: Blot for 20 sec before plunging |
| Details | 11 day dialysis at 18 deg C |
| Crystal formation | Details: 11 day dialysis at 18 deg C |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F30 |
|---|---|
| Temperature | Min: 77 K / Max: 77 K / Average: 77 K |
| Alignment procedure | Legacy - Astigmatism: corrected at 230,000 times magnification |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: ZEISS SCAI / Digitization - Sampling interval: 7 µm / Number real images: 87 / Average electron dose: 15 e/Å2 |
| Tilt angle min | 0 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 59000 / Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Cs: 2.3 mm / Nominal defocus max: 1.41 µm / Nominal defocus min: 0.27 µm / Nominal magnification: 60000 |
| Sample stage | Specimen holder: Side entry liquid-nitrogen cooled Gatan holder Specimen holder model: GATAN LIQUID NITROGEN / Tilt angle max: 60 / Tilt series - Axis1 - Min angle: 0 ° / Tilt series - Axis1 - Max angle: 60 ° |
| Experimental equipment |  Model: Tecnai F30 / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 5.5 Å / Resolution method: OTHER / Software - Name: MRC |
|---|---|
| Crystal parameters | Unit cell - A: 58.8 Å / Unit cell - B: 83.7 Å / Unit cell - C: 200.0 Å / Unit cell - γ: 90 ° / Unit cell - α: 90 ° / Unit cell - β: 90 ° / Plane group: P 2 21 21 |
| CTF correction | Details: Each image |
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Chain ID: B |
|---|---|
| Software | Name: O |
| Details | PDBEntryID_givenInChain. Protocol: Rigid Body |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
 Movie
Movie Controller
Controller


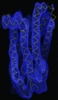
 Z (Sec.)
Z (Sec.) X (Row.)
X (Row.) Y (Col.)
Y (Col.)






















