+ Open data
Open data
- Basic information
Basic information
| Entry | Database: SASBDB / ID: SASDAS7 |
|---|---|
 Sample Sample | mouse olfactomedin-1
|
| Function / homology |  Function and homology information Function and homology informationatrioventricular valve formation / neuronal signal transduction / extrinsic component of postsynaptic density membrane / extrinsic component of synaptic membrane / regulation of amyloid-beta formation / cardiac epithelial to mesenchymal transition / regulation of axon extension / regulation of neurotransmitter receptor localization to postsynaptic specialization membrane / negative regulation of amyloid-beta formation / AMPA glutamate receptor complex ...atrioventricular valve formation / neuronal signal transduction / extrinsic component of postsynaptic density membrane / extrinsic component of synaptic membrane / regulation of amyloid-beta formation / cardiac epithelial to mesenchymal transition / regulation of axon extension / regulation of neurotransmitter receptor localization to postsynaptic specialization membrane / negative regulation of amyloid-beta formation / AMPA glutamate receptor complex / positive regulation of epithelial to mesenchymal transition / axonal growth cone / synaptic membrane / amyloid-beta binding / perikaryon / positive regulation of apoptotic process / negative regulation of gene expression / axon / neuronal cell body / positive regulation of gene expression / glutamatergic synapse / endoplasmic reticulum / signal transduction / : / identical protein binding Similarity search - Function |
| Biological species |  |
 Citation Citation |  Journal: J Biol Chem / Year: 2015 Journal: J Biol Chem / Year: 2015Title: Olfactomedin-1 Has a V-shaped Disulfide-linked Tetrameric Structure. Authors: Matti F Pronker / Trusanne G A A Bos / Thomas H Sharp / Dominique M E Thies-Weesie / Bert J C Janssen /  Abstract: Olfactomedin-1 (Olfm1; also known as noelin and pancortin) is a member of the olfactomedin domain-containing superfamily and a highly expressed neuronal glycoprotein important for nervous system ...Olfactomedin-1 (Olfm1; also known as noelin and pancortin) is a member of the olfactomedin domain-containing superfamily and a highly expressed neuronal glycoprotein important for nervous system development. It binds a number of secreted proteins and cell surface-bound receptors to induce cell signaling processes. Using a combined approach of x-ray crystallography, solution scattering, analytical ultracentrifugation, and electron microscopy we determined that full-length Olfm1 forms disulfide-linked tetramers with a distinctive V-shaped architecture. The base of the "V" is formed by two disulfide-linked dimeric N-terminal domains. Each of the two V legs consists of a parallel dimeric disulfide-linked coiled coil with a C-terminal β-propeller dimer at the tips. This agrees with our crystal structure of a C-terminal coiled-coil segment and β-propeller combination (Olfm1(coil-Olf)) that reveals a disulfide-linked dimeric arrangement with the β-propeller top faces in an outward exposed orientation. Similar to its family member myocilin, Olfm1 is stabilized by calcium. The dimer-of-dimers architecture suggests a role for Olfm1 in clustering receptors to regulate signaling and sheds light on the conformation of several other olfactomedin domain family members. |
 Contact author Contact author |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
-Data source
| SASBDB page |  SASDAS7 SASDAS7 |
|---|
-Related structure data
| Related structure data | 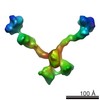 2940C  2941C  2942C  2943C  2944C  5amoC C: citing same article ( |
|---|---|
| Similar structure data |
- External links
External links
| Related items in Molecule of the Month |
|---|
-Models
| Model #298 |  Type: dummy / Software: Dammif / Radius of dummy atoms: 6.30 A / Chi-square value: 4.313  Search similar-shape structures of this assembly by Omokage search (details) Search similar-shape structures of this assembly by Omokage search (details) |
|---|
- Sample
Sample
 Sample Sample | Name: mouse olfactomedin-1 / Specimen concentration: 3.55 mg/ml |
|---|---|
| Buffer | Name: 20 mM HEPES 150 mM NaCl / Concentration: 20.00 mM / pH: 7.5 / Composition: 150 mM NaCl |
| Entity #163 | Name: Olfactomedin-1 / Type: protein / Description: Noelin / Formula weight: 64 / Num. of mol.: 4 / Source: Mus musculus / References: UniProt: O88998 Sequence: MITNWMSQTL PSLVGLNTTR LSAASGGTLD RSTGVLPTNP EESWQVYSSA QDSEGRCICT VVAPQQTMCS RDARTKQLRQ LLEKVQNMSQ SIEVLDRRTQ RDLQYVEKME NQMKGLETKF KQVEESHKQH LARQFKAIKA KMDELRPLIP VLEEYKADAK LVLQFKEEVQ ...Sequence: MITNWMSQTL PSLVGLNTTR LSAASGGTLD RSTGVLPTNP EESWQVYSSA QDSEGRCICT VVAPQQTMCS RDARTKQLRQ LLEKVQNMSQ SIEVLDRRTQ RDLQYVEKME NQMKGLETKF KQVEESHKQH LARQFKAIKA KMDELRPLIP VLEEYKADAK LVLQFKEEVQ NLTSVLNELQ EEIGAYDYDE LQSRVSNLEE RLRACMQKLA CGKLTGISDP VTVKTSGSRF GSWMTDPLAP EGDNRVWYMD GYHNNRFVRE YKSMVDFMNT DNFTSHRLPH PWSGTGQVVY NGSIYFNKFQ SHIIIRFDLK TETILKTRSL DYAGYNNMYH YAWGGHSDID LMVDENGLWA VYATNQNAGN IVISKLDPVS LQILQTWNTS YPKRSAGEAF IICGTLYVTN GYSGGTKVHY AYQTNASTYE YIDIPFQNKY SHISMLDYNP KDRALYAW |
-Experimental information
| Beam | Instrument name: ESRF BM29 / City: Grenoble / 国: France  / Type of source: X-ray synchrotron / Wavelength: 0.93 Å / Dist. spec. to detc.: 2.43 mm / Type of source: X-ray synchrotron / Wavelength: 0.93 Å / Dist. spec. to detc.: 2.43 mm | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Detector | Name: Pilatus 1M | ||||||||||||||||||||||||
| Scan |
| ||||||||||||||||||||||||
| Distance distribution function P(R) |
| ||||||||||||||||||||||||
| Result | Comments: SAXS was used to determine that Olfactomedin-1 forms tetramers in solution (confirmed by SEC-MALS and AUC). Scattering data were combined with crystallographic and negative stain electron ...Comments: SAXS was used to determine that Olfactomedin-1 forms tetramers in solution (confirmed by SEC-MALS and AUC). Scattering data were combined with crystallographic and negative stain electron tomography data to construct a model of a V-shaped dimer-of-dimers.
|
 Movie
Movie Controller
Controller






