+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8fne | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
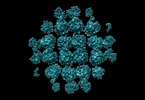
| Title | phiPA3 PhuN Tetramer, p2 | |||||||||
 Components Components | Maltose/maltodextrin-binding periplasmic protein, PhuN | |||||||||
 Keywords Keywords | VIRAL PROTEIN / phiPA3 protein / shell protein / PhuN / phage nucleus | |||||||||
| Function / homology |  Function and homology information Function and homology informationdetection of maltose stimulus / maltose transport complex / carbohydrate transport / carbohydrate transmembrane transporter activity / maltose binding / maltose transport / maltodextrin transmembrane transport / ATP-binding cassette (ABC) transporter complex, substrate-binding subunit-containing / ATP-binding cassette (ABC) transporter complex / cell chemotaxis ...detection of maltose stimulus / maltose transport complex / carbohydrate transport / carbohydrate transmembrane transporter activity / maltose binding / maltose transport / maltodextrin transmembrane transport / ATP-binding cassette (ABC) transporter complex, substrate-binding subunit-containing / ATP-binding cassette (ABC) transporter complex / cell chemotaxis / outer membrane-bounded periplasmic space / host cell cytoplasm / periplasmic space / DNA damage response / membrane Similarity search - Function | |||||||||
| Biological species |   Pseudomonas phage PhiPA3 (virus) Pseudomonas phage PhiPA3 (virus) | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Nieweglowska, E.S. / Brilot, A.F. / Mendez-Moran, M. / Kokontis, C. / Baek, M. / Li, J. / Cheng, Y. / Baker, D. / Bondy-Denomy, J. / Agard, D.A. | |||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: The ϕPA3 phage nucleus is enclosed by a self-assembling 2D crystalline lattice. Authors: Eliza S Nieweglowska / Axel F Brilot / Melissa Méndez-Moran / Claire Kokontis / Minkyung Baek / Junrui Li / Yifan Cheng / David Baker / Joseph Bondy-Denomy / David A Agard /  Abstract: To protect themselves from host attack, numerous jumbo bacteriophages establish a phage nucleus-a micron-scale, proteinaceous structure encompassing the replicating phage DNA. Bacteriophage and host ...To protect themselves from host attack, numerous jumbo bacteriophages establish a phage nucleus-a micron-scale, proteinaceous structure encompassing the replicating phage DNA. Bacteriophage and host proteins associated with replication and transcription are concentrated inside the phage nucleus while other phage and host proteins are excluded, including CRISPR-Cas and restriction endonuclease host defense systems. Here, we show that nucleus fragments isolated from ϕPA3 infected Pseudomonas aeruginosa form a 2-dimensional lattice, having p2 or p4 symmetry. We further demonstrate that recombinantly purified primary Phage Nuclear Enclosure (PhuN) protein spontaneously assembles into similar 2D sheets with p2 and p4 symmetry. We resolve the dominant p2 symmetric state to 3.9 Å by cryo-EM. Our structure reveals a two-domain core, organized into quasi-symmetric tetramers. Flexible loops and termini mediate adaptable inter-tetramer contacts that drive subunit assembly into a lattice and enable the adoption of different symmetric states. While the interfaces between subunits are mostly well packed, two are open, forming channels that likely have functional implications for the transport of proteins, mRNA, and small molecules. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8fne.cif.gz 8fne.cif.gz | 799.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8fne.ent.gz pdb8fne.ent.gz | 607 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8fne.json.gz 8fne.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8fne_validation.pdf.gz 8fne_validation.pdf.gz | 1019.3 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8fne_full_validation.pdf.gz 8fne_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  8fne_validation.xml.gz 8fne_validation.xml.gz | 64.3 KB | Display | |
| Data in CIF |  8fne_validation.cif.gz 8fne_validation.cif.gz | 99.9 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fn/8fne https://data.pdbj.org/pub/pdb/validation_reports/fn/8fne ftp://data.pdbj.org/pub/pdb/validation_reports/fn/8fne ftp://data.pdbj.org/pub/pdb/validation_reports/fn/8fne | HTTPS FTP |
-Related structure data
| Related structure data | 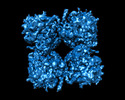 29310MC  8fv5C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 110126.078 Da / Num. of mol.: 8 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Pseudomonas phage PhiPA3 (virus) Pseudomonas phage PhiPA3 (virus)Gene: malE, b4034, JW3994, 053 / Production host:  |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: 2D ARRAY / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Core tetramer assembly (p2) of the phiPA3 bacteriophage PhuN protein Type: COMPLEX / Entity ID: all / Source: RECOMBINANT | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Experimental value: NO | ||||||||||||||||||||||||||||
| Source (natural) | Organism:  Pseudomonas phage PhiPA3 (virus) Pseudomonas phage PhiPA3 (virus) | ||||||||||||||||||||||||||||
| Source (recombinant) | Organism:  | ||||||||||||||||||||||||||||
| Buffer solution | pH: 6.5 Details: 0.25 cOmplete Protease Inhibitor Tablet also included | ||||||||||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 283.15 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 105000 X / Nominal defocus max: 2500 nm / Nominal defocus min: 800 nm / Cs: 2.7 mm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 67 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CTF correction | Type: NONE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C2 (2 fold cyclic) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.9 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 29303 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT Details: The initial model was generated using AlphaFold based on sequence alone. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Source name: AlphaFold / Type: in silico model |
 Movie
Movie Controller
Controller




 PDBj
PDBj


