+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8bf8 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
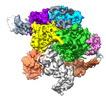
| Title | ISDra2 TnpB in complex with reRNA | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords | RNA BINDING PROTEIN / Transposon / TnpB / reRNA | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationtransposition / Hydrolases; Acting on ester bonds; Endodeoxyribonucleases producing 5'-phosphomonoesters / endonuclease activity / DNA recombination / DNA binding / RNA binding / metal ion binding Similarity search - Function | |||||||||||||||
| Biological species |  Deinococcus radiodurans R1 (radioresistant) Deinococcus radiodurans R1 (radioresistant) | |||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.8 Å | |||||||||||||||
 Authors Authors | Sasnauskas, G. / Tamulaitiene, G. / Carabias, A. / Siksnys, V. / Montoya, G. / Druteika, G. / Silanskas, A. / Venclovas, C. / Karvelis, T. / Kazlauskas, D. | |||||||||||||||
| Funding support |  Denmark, 4items Denmark, 4items
| |||||||||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: TnpB structure reveals minimal functional core of Cas12 nuclease family. Authors: Giedrius Sasnauskas / Giedre Tamulaitiene / Gytis Druteika / Arturo Carabias / Arunas Silanskas / Darius Kazlauskas / Česlovas Venclovas / Guillermo Montoya / Tautvydas Karvelis / Virginijus Siksnys /  Abstract: The widespread TnpB proteins of IS200/IS605 transposon family have recently emerged as the smallest RNA-guided nucleases capable of targeted genome editing in eukaryotic cells. Bioinformatic analysis ...The widespread TnpB proteins of IS200/IS605 transposon family have recently emerged as the smallest RNA-guided nucleases capable of targeted genome editing in eukaryotic cells. Bioinformatic analysis identified TnpB proteins as the likely predecessors of Cas12 nucleases, which along with Cas9 are widely used for targeted genome manipulation. Whereas Cas12 family nucleases are well characterized both biochemically and structurally, the molecular mechanism of TnpB remains unknown. Here we present the cryogenic-electron microscopy structures of the Deinococcus radiodurans TnpB-reRNA (right-end transposon element-derived RNA) complex in DNA-bound and -free forms. The structures reveal the basic architecture of TnpB nuclease and the molecular mechanism for DNA target recognition and cleavage that is supported by biochemical experiments. Collectively, these results demonstrate that TnpB represents the minimal structural and functional core of the Cas12 protein family and provide a framework for developing TnpB-based genome editing tools. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8bf8.cif.gz 8bf8.cif.gz | 148 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8bf8.ent.gz pdb8bf8.ent.gz | 96.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8bf8.json.gz 8bf8.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8bf8_validation.pdf.gz 8bf8_validation.pdf.gz | 1.1 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8bf8_full_validation.pdf.gz 8bf8_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  8bf8_validation.xml.gz 8bf8_validation.xml.gz | 28.5 KB | Display | |
| Data in CIF |  8bf8_validation.cif.gz 8bf8_validation.cif.gz | 40.3 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bf/8bf8 https://data.pdbj.org/pub/pdb/validation_reports/bf/8bf8 ftp://data.pdbj.org/pub/pdb/validation_reports/bf/8bf8 ftp://data.pdbj.org/pub/pdb/validation_reports/bf/8bf8 | HTTPS FTP |
-Related structure data
| Related structure data | 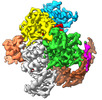 16016MC  8ex9C  8exaC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 46484.289 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Deinococcus radiodurans R1 (radioresistant) Deinococcus radiodurans R1 (radioresistant)Strain: ATCC 13939 / DSM 20539 / JCM 16871 / LMG 4051 / NBRC 15346 / NCIMB 9279 / R1 / VKM B-1422 Gene: tnpB, orf2, tnpC, DR_0666, DR_0978, DR_1381, DR_1593, DR_1651, DR_1933, DR_2324 Production host:  References: UniProt: Q7DF80, Hydrolases; Acting on ester bonds; Endodeoxyribonucleases producing 5'-phosphomonoesters |
|---|---|
| #2: RNA chain | Mass: 48408.633 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Deinococcus radiodurans R1 (radioresistant) Deinococcus radiodurans R1 (radioresistant)Production host:  |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: ISDra2 TnpB binary complex / Type: COMPLEX / Details: ISDra2 TnpB in complex with reRNA / Entity ID: all / Source: RECOMBINANT | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.10 MDa / Experimental value: YES | ||||||||||||||||||||
| Source (natural) | Organism:  Deinococcus radiodurans (radioresistant) Deinococcus radiodurans (radioresistant) | ||||||||||||||||||||
| Source (recombinant) | Organism:  | ||||||||||||||||||||
| Buffer solution | pH: 8 | ||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R1.2/1.3 | ||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 95 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2000 nm / Nominal defocus min: 1000 nm / Cs: 2.7 mm |
| Specimen holder | Cryogen: NITROGEN |
| Image recording | Electron dose: 38 e/Å2 / Detector mode: COUNTING / Film or detector model: FEI FALCON III (4k x 4k) / Num. of grids imaged: 1 / Num. of real images: 6688 |
| Image scans | Width: 4000 / Height: 4000 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| |||||||||||||||||||||||||
| CTF correction | Type: NONE | |||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 4420084 | |||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | |||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 985959 / Num. of class averages: 1 / Symmetry type: POINT | |||||||||||||||||||||||||
| Refinement | Cross valid method: NONE |
 Movie
Movie Controller
Controller




 PDBj
PDBj





























