+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7z9c | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | E.coli gyrase holocomplex with 217 bp DNA and albicidin | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Components Components |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Keywords Keywords | ISOMERASE / type II topoisomerase / antibiotic / albicidin / DNA gyrase | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationAction of antimicrobials / negative regulation of DNA-templated DNA replication / DNA topoisomerase type II (double strand cut, ATP-hydrolyzing) complex / DNA negative supercoiling activity / DNA topoisomerase type II (double strand cut, ATP-hydrolyzing) activity / DNA topoisomerase (ATP-hydrolysing) / Antimicrobial resistance / DNA topological change / ATP-dependent activity, acting on DNA / DNA-templated DNA replication ...Action of antimicrobials / negative regulation of DNA-templated DNA replication / DNA topoisomerase type II (double strand cut, ATP-hydrolyzing) complex / DNA negative supercoiling activity / DNA topoisomerase type II (double strand cut, ATP-hydrolyzing) activity / DNA topoisomerase (ATP-hydrolysing) / Antimicrobial resistance / DNA topological change / ATP-dependent activity, acting on DNA / DNA-templated DNA replication / chromosome / response to xenobiotic stimulus / response to antibiotic / DNA-templated transcription / DNA binding / ATP binding / membrane / metal ion binding / identical protein binding / cytoplasm / cytosol Similarity search - Function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological species |   Escherichia phage Mu (virus) Escherichia phage Mu (virus) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.06 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Authors Authors | Ghilarov, D. / Heddle, J.G.H. / Suessmuth, R. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Funding support |  United Kingdom, United Kingdom,  Poland, 2items Poland, 2items
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Catal / Year: 2023 Journal: Nat Catal / Year: 2023Title: Molecular mechanism of topoisomerase poisoning by the peptide antibiotic albicidin. Authors: Elizabeth Michalczyk / Kay Hommernick / Iraj Behroz / Marcel Kulike / Zuzanna Pakosz-Stępień / Lukasz Mazurek / Maria Seidel / Maria Kunert / Karine Santos / Holger von Moeller / Bernhard ...Authors: Elizabeth Michalczyk / Kay Hommernick / Iraj Behroz / Marcel Kulike / Zuzanna Pakosz-Stępień / Lukasz Mazurek / Maria Seidel / Maria Kunert / Karine Santos / Holger von Moeller / Bernhard Loll / John B Weston / Andi Mainz / Jonathan G Heddle / Roderich D Süssmuth / Dmitry Ghilarov /    Abstract: The peptide antibiotic albicidin is a DNA topoisomerase inhibitor with low-nanomolar bactericidal activity towards fluoroquinolone-resistant Gram-negative pathogens. However, its mode of action is ...The peptide antibiotic albicidin is a DNA topoisomerase inhibitor with low-nanomolar bactericidal activity towards fluoroquinolone-resistant Gram-negative pathogens. However, its mode of action is poorly understood. We determined a 2.6 Å resolution cryoelectron microscopy structure of a ternary complex between topoisomerase DNA gyrase, a 217 bp double-stranded DNA fragment and albicidin. Albicidin employs a dual binding mechanism where one end of the molecule obstructs the crucial gyrase dimer interface, while the other intercalates between the fragments of cleaved DNA substrate. Thus, albicidin efficiently locks DNA gyrase, preventing it from religating DNA and completing its catalytic cycle. Two additional structures of this trapped state were determined using synthetic albicidin analogues that demonstrate improved solubility, and activity against a range of gyrase variants and topoisomerase IV. The extraordinary promiscuity of the DNA-intercalating region of albicidins and their excellent performance against fluoroquinolone-resistant bacteria holds great promise for the development of last-resort antibiotics. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7z9c.cif.gz 7z9c.cif.gz | 686.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7z9c.ent.gz pdb7z9c.ent.gz | 545.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7z9c.json.gz 7z9c.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/z9/7z9c https://data.pdbj.org/pub/pdb/validation_reports/z9/7z9c ftp://data.pdbj.org/pub/pdb/validation_reports/z9/7z9c ftp://data.pdbj.org/pub/pdb/validation_reports/z9/7z9c | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  14570MC  7z9gC  7z9kC  7z9mC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-DNA gyrase subunit ... , 2 types, 4 molecules ACBD
| #1: Protein | Mass: 97854.305 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Strain: K12 / Gene: gyrA, hisW, nalA, parD, b2231, JW2225 / Production host:  References: UniProt: P0AES4, DNA topoisomerase (ATP-hydrolysing) #2: Protein | Mass: 90891.734 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Strain: K12 Gene: gyrB, acrB, cou, himB, hisU, nalC, parA, pcbA, b3699, JW5625 Production host:  References: UniProt: P0AES6, DNA topoisomerase (ATP-hydrolysing) |
|---|
-DNA chain , 4 types, 4 molecules EFGH
| #3: DNA chain | Mass: 4335.827 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Escherichia phage Mu (virus) / Production host: Escherichia phage Mu (virus) / Production host:  |
|---|---|
| #4: DNA chain | Mass: 4321.856 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Escherichia phage Mu (virus) / Production host: Escherichia phage Mu (virus) / Production host:  |
| #5: DNA chain | Mass: 5444.558 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Escherichia phage Mu (virus) / Production host: Escherichia phage Mu (virus) / Production host:  |
| #6: DNA chain | Mass: 5487.561 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Escherichia phage Mu (virus) / Production host: Escherichia phage Mu (virus) / Production host:  |
-Non-polymers , 3 types, 13 molecules 
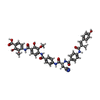



| #7: Chemical | | #8: Chemical | ChemComp-BWH / | #9: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Experimental value: NO | ||||||||||||||||||||||||
| Source (natural) |
| ||||||||||||||||||||||||
| Source (recombinant) |
| ||||||||||||||||||||||||
| Buffer solution | pH: 8 | ||||||||||||||||||||||||
| Buffer component | Conc.: 20 mM / Name: HEPES | ||||||||||||||||||||||||
| Specimen | Conc.: 12 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 95 % / Chamber temperature: 283 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 3000 nm / Nominal defocus min: 900 nm |
| Image recording | Electron dose: 40 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.19.2_4158: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 2040353 | ||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.06 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 23435 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






 PDBj
PDBj








































