+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7tti | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human KCC1 bound with VU0463271 In an outward-open state | ||||||||||||||||||||||||||||||||||||
 Components Components | Solute carrier family 12 member 4 | ||||||||||||||||||||||||||||||||||||
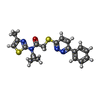
 Keywords Keywords | TRANSPORT PROTEIN/INHIBITOR / SLC12A4 / VU0463271 / Outward-open state / TRANSPORT PROTEIN-INHIBITOR complex | ||||||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationpotassium:chloride symporter activity / Cation-coupled Chloride cotransporters / chloride ion homeostasis / potassium ion homeostasis / cell volume homeostasis / potassium ion import across plasma membrane / monoatomic ion transport / potassium ion transmembrane transport / protein serine/threonine kinase binding / chloride transmembrane transport ...potassium:chloride symporter activity / Cation-coupled Chloride cotransporters / chloride ion homeostasis / potassium ion homeostasis / cell volume homeostasis / potassium ion import across plasma membrane / monoatomic ion transport / potassium ion transmembrane transport / protein serine/threonine kinase binding / chloride transmembrane transport / chemical synaptic transmission / lysosomal membrane / synapse / ATP binding / membrane / plasma membrane Similarity search - Function | ||||||||||||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.5 Å | ||||||||||||||||||||||||||||||||||||
 Authors Authors | Zhao, Y.X. / Cao, E.H. | ||||||||||||||||||||||||||||||||||||
| Funding support | 1items
| ||||||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2022 Journal: Proc Natl Acad Sci U S A / Year: 2022Title: Structure of the human cation-chloride cotransport KCC1 in an outward-open state. Authors: Yongxiang Zhao / Jiemin Shen / Qinzhe Wang / Manuel Jose Ruiz Munevar / Pietro Vidossich / Marco De Vivo / Ming Zhou / Erhu Cao /   Abstract: Cation-chloride cotransporters (CCCs) catalyze electroneutral symport of Cl with Na and/or K across membranes. CCCs are fundamental in cell volume homeostasis, transepithelia ion movement, ...Cation-chloride cotransporters (CCCs) catalyze electroneutral symport of Cl with Na and/or K across membranes. CCCs are fundamental in cell volume homeostasis, transepithelia ion movement, maintenance of intracellular Cl concentration, and neuronal excitability. Here, we present a cryoelectron microscopy structure of human K-Cl cotransporter (KCC)1 bound with the VU0463271 inhibitor in an outward-open state. In contrast to many other amino acid-polyamine-organocation transporter cousins, our first outward-open CCC structure reveals that opening the KCC1 extracellular ion permeation path does not involve hinge-bending motions of the transmembrane (TM) 1 and TM6 half-helices. Instead, rocking of TM3 and TM8, together with displacements of TM4, TM9, and a conserved intracellular loop 1 helix, underlie alternate opening and closing of extracellular and cytoplasmic vestibules. We show that KCC1 intriguingly exists in one of two distinct dimeric states via different intersubunit interfaces. Our studies provide a blueprint for understanding the mechanisms of CCCs and their inhibition by small molecule compounds. | ||||||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7tti.cif.gz 7tti.cif.gz | 227.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7tti.ent.gz pdb7tti.ent.gz | 164.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7tti.json.gz 7tti.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/tt/7tti https://data.pdbj.org/pub/pdb/validation_reports/tt/7tti ftp://data.pdbj.org/pub/pdb/validation_reports/tt/7tti ftp://data.pdbj.org/pub/pdb/validation_reports/tt/7tti | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  26116MC  7tthC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
| EM raw data |  EMPIAR-11047 (Title: Cryo-EM structure of human KCC1 bound with inhibitor VU0463271 EMPIAR-11047 (Title: Cryo-EM structure of human KCC1 bound with inhibitor VU0463271Data size: 3.9 TB Data #1: HumanKCC1-VU0463271 complex [micrographs - multiframe]) |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 120770.492 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SLC12A4, KCC1 / Production host: Homo sapiens (human) / Gene: SLC12A4, KCC1 / Production host:  Homo sapiens (human) / References: UniProt: Q9UP95 Homo sapiens (human) / References: UniProt: Q9UP95#2: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source #3: Chemical | Has ligand of interest | Y | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: human potassium-chloride cotransporter 1 bound with VU0463271 Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: DIFFRACTION / Nominal defocus max: 3500 nm / Nominal defocus min: 700 nm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.20_4459: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING ONLY | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.5 Å / Resolution method: DIFFRACTION PATTERN/LAYERLINES / Num. of particles: 81855 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller




 PDBj
PDBj