[English] 日本語
 Yorodumi
Yorodumi- PDB-6xez: Structure of SARS-CoV-2 replication-transcription complex bound t... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6xez | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of SARS-CoV-2 replication-transcription complex bound to nsp13 helicase - nsp13(2)-RTC | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TRANSFERASE/HYDROLASE/RNA / RNA-dependent RNA polymerase / viral replication-transcription complex / transcription / viral proteins / TRANSFERASE-HYDROLASE-RNA complex | ||||||
| Function / homology |  Function and homology information Function and homology informationprotein guanylyltransferase activity / RNA endonuclease activity, producing 3'-phosphomonoesters / mRNA guanylyltransferase activity / 5'-3' RNA helicase activity / Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs ...protein guanylyltransferase activity / RNA endonuclease activity, producing 3'-phosphomonoesters / mRNA guanylyltransferase activity / 5'-3' RNA helicase activity / Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs / Translation of Replicase and Assembly of the Replication Transcription Complex / TRAF3-dependent IRF activation pathway / Replication of the SARS-CoV-2 genome / snRNP Assembly / double membrane vesicle viral factory outer membrane / Hydrolases; Acting on ester bonds; Exoribonucleases producing 5'-phosphomonoesters / 5'-3' DNA helicase activity / SARS coronavirus main proteinase / host cell endosome / host cell endoplasmic reticulum-Golgi intermediate compartment / 3'-5'-RNA exonuclease activity / symbiont-mediated suppression of host toll-like receptor signaling pathway / symbiont-mediated degradation of host mRNA / mRNA guanylyltransferase / symbiont-mediated suppression of host ISG15-protein conjugation / G-quadruplex RNA binding / SARS-CoV-2 modulates host translation machinery / omega peptidase activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of IRF3 activity / mRNA (guanine-N7)-methyltransferase / host cell Golgi apparatus / methyltransferase cap1 / symbiont-mediated perturbation of host ubiquitin-like protein modification / mRNA (nucleoside-2'-O-)-methyltransferase activity / DNA helicase / mRNA 5'-cap (guanine-N7-)-methyltransferase activity / ubiquitinyl hydrolase 1 / cysteine-type deubiquitinase activity / Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases / single-stranded RNA binding / host cell endoplasmic reticulum membrane / host cell perinuclear region of cytoplasm / viral protein processing / lyase activity / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / RNA helicase / induction by virus of host autophagy / copper ion binding / RNA-directed RNA polymerase / viral RNA genome replication / cysteine-type endopeptidase activity / RNA-dependent RNA polymerase activity / virus-mediated perturbation of host defense response / DNA-templated transcription / lipid binding / host cell nucleus / SARS-CoV-2 activates/modulates innate and adaptive immune responses / ATP hydrolysis activity / proteolysis / RNA binding / zinc ion binding / ATP binding / membrane Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.5 Å | ||||||
 Authors Authors | Chen, J. / Malone, B. / Llewellyn, E.C. / Campbell, E.A. / Darst, S.A. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Cell / Year: 2020 Journal: Cell / Year: 2020Title: Structural Basis for Helicase-Polymerase Coupling in the SARS-CoV-2 Replication-Transcription Complex. Authors: James Chen / Brandon Malone / Eliza Llewellyn / Michael Grasso / Patrick M M Shelton / Paul Dominic B Olinares / Kashyap Maruthi / Edward T Eng / Hasan Vatandaslar / Brian T Chait / Tarun M ...Authors: James Chen / Brandon Malone / Eliza Llewellyn / Michael Grasso / Patrick M M Shelton / Paul Dominic B Olinares / Kashyap Maruthi / Edward T Eng / Hasan Vatandaslar / Brian T Chait / Tarun M Kapoor / Seth A Darst / Elizabeth A Campbell /   Abstract: SARS-CoV-2 is the causative agent of the 2019-2020 pandemic. The SARS-CoV-2 genome is replicated and transcribed by the RNA-dependent RNA polymerase holoenzyme (subunits nsp7/nsp8/nsp12) along with a ...SARS-CoV-2 is the causative agent of the 2019-2020 pandemic. The SARS-CoV-2 genome is replicated and transcribed by the RNA-dependent RNA polymerase holoenzyme (subunits nsp7/nsp8/nsp12) along with a cast of accessory factors. One of these factors is the nsp13 helicase. Both the holo-RdRp and nsp13 are essential for viral replication and are targets for treating the disease COVID-19. Here we present cryoelectron microscopic structures of the SARS-CoV-2 holo-RdRp with an RNA template product in complex with two molecules of the nsp13 helicase. The Nidovirales order-specific N-terminal domains of each nsp13 interact with the N-terminal extension of each copy of nsp8. One nsp13 also contacts the nsp12 thumb. The structure places the nucleic acid-binding ATPase domains of the helicase directly in front of the replicating-transcribing holo-RdRp, constraining models for nsp13 function. We also observe ADP-Mg bound in the nsp12 N-terminal nidovirus RdRp-associated nucleotidyltransferase domain, detailing a new pocket for anti-viral therapy development. #1:  Journal: bioRxiv / Year: 2020 Journal: bioRxiv / Year: 2020Title: Structural basis for helicase-polymerase coupling in the SARS-CoV-2 replication-transcription complex. Authors: James Chen / Brandon Malone / Eliza Llewellyn / Michael Grasso / Patrick M M Shelton / Paul Dominic B Olinares / Kashyap Maruthi / Ed Eng / Hasan Vatandaslar / Brian T Chait / Tarun Kapoor / ...Authors: James Chen / Brandon Malone / Eliza Llewellyn / Michael Grasso / Patrick M M Shelton / Paul Dominic B Olinares / Kashyap Maruthi / Ed Eng / Hasan Vatandaslar / Brian T Chait / Tarun Kapoor / Seth A Darst / Elizabeth A Campbell /   Abstract: SARS-CoV-2 is the causative agent of the 2019-2020 pandemic. The SARS-CoV-2 genome is replicated-transcribed by the RNA-dependent RNA polymerase holoenzyme (subunits nsp7/nsp82/nsp12) along with a ...SARS-CoV-2 is the causative agent of the 2019-2020 pandemic. The SARS-CoV-2 genome is replicated-transcribed by the RNA-dependent RNA polymerase holoenzyme (subunits nsp7/nsp82/nsp12) along with a cast of accessory factors. One of these factors is the nsp13 helicase. Both the holo-RdRp and nsp13 are essential for viral replication and are targets for treating the disease COVID-19. Here we present cryo-electron microscopic structures of the SARS-CoV-2 holo-RdRp with an RNA template-product in complex with two molecules of the nsp13 helicase. The Nidovirus-order-specific N-terminal domains of each nsp13 interact with the N-terminal extension of each copy of nsp8. One nsp13 also contacts the nsp12-thumb. The structure places the nucleic acid-binding ATPase domains of the helicase directly in front of the replicating-transcribing holo-RdRp, constraining models for nsp13 function. We also observe ADP-Mg2+ bound in the nsp12 N-terminal nidovirus RdRp-associated nucleotidyltransferase domain, detailing a new pocket for anti-viral therapeutic development. #2:  Journal: bioRxiv / Year: 2020 Journal: bioRxiv / Year: 2020Title: Structural basis for helicase-polymerase coupling in the SARS-CoV-2 replication-transcription complex. Authors: James Chen / Brandon Malone / Eliza Llewellyn / Michael Grasso / Patrick M M Shelton / Paul Dominic B Olinares / Kashyap Maruthi / Ed Eng / Hasan Vatandaslar / Brian T Chait / Tarun Kapoor / ...Authors: James Chen / Brandon Malone / Eliza Llewellyn / Michael Grasso / Patrick M M Shelton / Paul Dominic B Olinares / Kashyap Maruthi / Ed Eng / Hasan Vatandaslar / Brian T Chait / Tarun Kapoor / Seth A Darst / Elizabeth A Campbell /   Abstract: SARS-CoV-2 is the causative agent of the 2019-2020 pandemic. The SARS-CoV-2 genome is replicated-transcribed by the RNA-dependent RNA polymerase holoenzyme (subunits nsp7/nsp82/nsp12) along with a ...SARS-CoV-2 is the causative agent of the 2019-2020 pandemic. The SARS-CoV-2 genome is replicated-transcribed by the RNA-dependent RNA polymerase holoenzyme (subunits nsp7/nsp82/nsp12) along with a cast of accessory factors. One of these factors is the nsp13 helicase. Both the holo-RdRp and nsp13 are essential for viral replication and are targets for treating the disease COVID-19. Here we present cryo-electron microscopic structures of the SARS-CoV-2 holo-RdRp with an RNA template-product in complex with two molecules of the nsp13 helicase. The Nidovirus-order-specific N-terminal domains of each nsp13 interact with the N-terminal extension of each copy of nsp8. One nsp13 also contacts the nsp12-thumb. The structure places the nucleic acid-binding ATPase domains of the helicase directly in front of the replicating-transcribing holo-RdRp, constraining models for nsp13 function. We also observe ADP-Mg2+ bound in the nsp12 N-terminal nidovirus RdRp-associated nucleotidyltransferase domain, detailing a new pocket for anti-viral therapeutic development. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6xez.cif.gz 6xez.cif.gz | 497.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6xez.ent.gz pdb6xez.ent.gz | 395.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6xez.json.gz 6xez.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6xez_validation.pdf.gz 6xez_validation.pdf.gz | 1.3 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6xez_full_validation.pdf.gz 6xez_full_validation.pdf.gz | 1.4 MB | Display | |
| Data in XML |  6xez_validation.xml.gz 6xez_validation.xml.gz | 87.1 KB | Display | |
| Data in CIF |  6xez_validation.cif.gz 6xez_validation.cif.gz | 133.1 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/xe/6xez https://data.pdbj.org/pub/pdb/validation_reports/xe/6xez ftp://data.pdbj.org/pub/pdb/validation_reports/xe/6xez ftp://data.pdbj.org/pub/pdb/validation_reports/xe/6xez | HTTPS FTP |
-Related structure data
| Related structure data |  22160MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 2 types, 3 molecules AEF
| #1: Protein | Mass: 106780.977 Da / Num. of mol.: 1 / Fragment: UNP residues 4393-5324 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Gene: rep, 1a-1b / Production host:  |
|---|---|
| #4: Protein | Mass: 67354.039 Da / Num. of mol.: 2 / Fragment: UNP residues 5325-5925 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Gene: rep, 1a-1b / Production host:  |
-Non-structural protein ... , 2 types, 3 molecules BDC
| #2: Protein | Mass: 22034.242 Da / Num. of mol.: 2 / Fragment: UNP residues 3943-4140 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Gene: rep, 1a-1b / Production host:  #3: Protein | | Mass: 9748.385 Da / Num. of mol.: 1 / Fragment: UNP residues 3860-3942 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Gene: rep, 1a-1b / Production host:  |
|---|
-RNA chain , 2 types, 2 molecules PT
| #5: RNA chain | Mass: 11141.644 Da / Num. of mol.: 1 / Source method: obtained synthetically Source: (synth.)  |
|---|---|
| #6: RNA chain | Mass: 17573.391 Da / Num. of mol.: 1 / Source method: obtained synthetically Source: (synth.)  |
-Non-polymers , 5 types, 19 molecules 


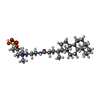





| #7: Chemical | ChemComp-ZN / #8: Chemical | #9: Chemical | #10: Chemical | #11: Chemical | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: SARS-CoV-2 replication/transcription complex bound to nsp13 helicase - nsp13(2)-RTC Type: COMPLEX / Entity ID: #1-#6 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 8 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Details: unspecified |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 65 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.17.1_3660: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.5 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 58942 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj











































