+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | MicroED structure of the C11 cysteine protease clostripain | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Hydrolase / protease / thiol protease | |||||||||
| Function / homology | clostripain / Peptidase C11, Clostripain Clostridium species / Peptidase C11, clostripain / Clostripain family / cysteine-type endopeptidase activity / proteolysis / Clostripain Function and homology information Function and homology information | |||||||||
| Biological species |  Hathewaya histolytica (bacteria) Hathewaya histolytica (bacteria) | |||||||||
| Method | electron crystallography / cryo EM / Resolution: 2.5 Å | |||||||||
 Authors Authors | Ruma YN / Bu G / Hattne J / Gonen T | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: J Struct Biol X / Year: 2024 Journal: J Struct Biol X / Year: 2024Title: MicroED structure of the C11 cysteine protease clostripain. Authors: Yasmeen N Ruma / Guanhong Bu / Johan Hattne / Tamir Gonen /  Abstract: Clostripain secreted from is the founding member of the C11 family of Clan CD cysteine peptidases, which is an important group of peptidases secreted by numerous bacteria. Clostripain is an arginine- ...Clostripain secreted from is the founding member of the C11 family of Clan CD cysteine peptidases, which is an important group of peptidases secreted by numerous bacteria. Clostripain is an arginine-specific endopeptidase. Because of its efficacy as a cysteine peptidase, it is widely used in laboratory settings. Despite its importance the structure of clostripain remains unsolved. Here we describe the first structure of an active form of clostripain determined at 2.5 Å resolution using microcrystal electron diffraction (MicroED). The structure was determined from a single nanocrystal after focused ion beam milling. The structure of clostripain shows a typical Clan CD α/β/α sandwich architecture and the Cys231/His176 catalytic dyad in the active site. It has a large electronegative substrate binding pocket showing its ability to accommodate large and diverse substrates. A loop in the heavy chain formed between residues 452 and 457 is potentially important for substrate binding. In conclusion, this result demonstrates the importance of MicroED to determine the unknown structure of macromolecules such as clostripain, which can be further used as a platform to study substrate binding and design of potential inhibitors against this class of peptidases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_45623.map.gz emd_45623.map.gz | 4.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-45623-v30.xml emd-45623-v30.xml emd-45623.xml emd-45623.xml | 15.1 KB 15.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_45623.png emd_45623.png | 204.3 KB | ||
| Filedesc metadata |  emd-45623.cif.gz emd-45623.cif.gz | 6.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-45623 http://ftp.pdbj.org/pub/emdb/structures/EMD-45623 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-45623 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-45623 | HTTPS FTP |
-Validation report
| Summary document |  emd_45623_validation.pdf.gz emd_45623_validation.pdf.gz | 616.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_45623_full_validation.pdf.gz emd_45623_full_validation.pdf.gz | 615.7 KB | Display | |
| Data in XML |  emd_45623_validation.xml.gz emd_45623_validation.xml.gz | 4.4 KB | Display | |
| Data in CIF |  emd_45623_validation.cif.gz emd_45623_validation.cif.gz | 4.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45623 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45623 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45623 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45623 | HTTPS FTP |
-Related structure data
| Related structure data |  9cipMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_45623.map.gz / Format: CCP4 / Size: 5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_45623.map.gz / Format: CCP4 / Size: 5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||
| Voxel size |
| ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 18 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Clostripain
| Entire | Name: Clostripain |
|---|---|
| Components |
|
-Supramolecule #1: Clostripain
| Supramolecule | Name: Clostripain / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Hathewaya histolytica (bacteria) Hathewaya histolytica (bacteria) |
| Molecular weight | Theoretical: 59 KDa |
-Macromolecule #1: Clostripain
| Macromolecule | Name: Clostripain / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Hathewaya histolytica (bacteria) Hathewaya histolytica (bacteria) |
| Molecular weight | Theoretical: 59.805293 KDa |
| Sequence | String: MLRRKVSTLL MTALITTSFL NSKPVYANPV TKSKDNNLKE VQQVTSKSNK NKNQKVTIMY YCDADNNLEG SLLNDIEEMK TGYKDSPNL NLIALVDRSP RYSSDEKVLG EDFSDTRLYK IEHNKANRLD GKNEFPEIST TSKYEANMGD PEVLKKFIDY C KSNYEADK ...String: MLRRKVSTLL MTALITTSFL NSKPVYANPV TKSKDNNLKE VQQVTSKSNK NKNQKVTIMY YCDADNNLEG SLLNDIEEMK TGYKDSPNL NLIALVDRSP RYSSDEKVLG EDFSDTRLYK IEHNKANRLD GKNEFPEIST TSKYEANMGD PEVLKKFIDY C KSNYEADK YVLIMANHGG GAREKSNPRL NRAICWDDSN LDKNGEADCL YMGEISDHLT EKQSVDLLAF DACLMGTAEV AY QYRPGNG GFSADTLVAS SPVVWGPGFK YDKIFDRIKA GGGTNNEDDL TLGGKEQNFD PATITNEQLG ALFVEEQRDS THA NGRYDQ HLSFYDLKKA ESVKRAIDNL AVNLSNENKK SEIEKLRGSG IHTDLMHYFD EYSEGEWVEY PYFDVYDLCE KINK SENFS SKTKDLASNA MNKLNEMIVY SFGDPSNNFK EGKNGLSIFL PNGDKKYSTY YTSTKIPHWT MQSWYNSIDT VKYGL NPYG KLSWCKDGQD PEINKVGNWF ELLDSWFDKT NDVTGGVNHY QW UniProtKB: Clostripain |
-Macromolecule #2: SODIUM ION
| Macromolecule | Name: SODIUM ION / type: ligand / ID: 2 / Number of copies: 7 |
|---|---|
| Molecular weight | Theoretical: 22.99 Da |
-Macromolecule #3: water
| Macromolecule | Name: water / type: ligand / ID: 3 / Number of copies: 6 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | electron crystallography |
| Aggregation state | 3D array |
- Sample preparation
Sample preparation
| Buffer | pH: 5.6 Component:
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY ARRAY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 45 sec. | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 293 K / Instrument: LEICA EM GP | ||||||||||||
| Crystal formation | Temperature: 293.0 K / Time: 1.0 DAY |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Temperature | Min: 93.0 K / Max: 93.0 K |
| Specialist optics | Energy filter - Name: TFS Selectris / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Number grids imaged: 1 / Number real images: 252 / Number diffraction images: 252 / Average exposure time: 5.0 sec. / Average electron dose: 0.04 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: DIFFRACTION / Nominal defocus max: 0.0 µm / Nominal defocus min: 0.0 µm / Camera length: 2500 mm |
| Sample stage | Cooling holder cryogen: NITROGEN / Tilt angle: -20.0, 70.0 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.5 Å / Resolution method: DIFFRACTION PATTERN/LAYERLINES |
|---|---|
| Molecular replacement | Software - Name: PHENIX (ver. 1.21.1_5287) |
| Merging software list | Software - Name: XSCALE (ver. Jun 30, 2023) |
| Crystallography statistics | Number intensities measured: 131476 / Number structure factors: 32113 / Fourier space coverage: 86.8 / R merge: 0.279 / Overall phase error: 34.49 / Overall phase residual: 0 / Phase error rejection criteria: 0 / High resolution: 2.5 Å / Shell - Shell ID: 1 / Shell - High resolution: 2.5 Å / Shell - Low resolution: 2.56 Å / Shell - Number structure factors: 2318 / Shell - Phase residual: 54.5 / Shell - Fourier space coverage: 86.5 / Shell - Multiplicity: 4.1 |
 Movie
Movie Controller
Controller



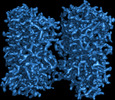
 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)