[English] 日本語
 Yorodumi
Yorodumi- EMDB-44491: Cryo-EM structure of the HIV-1 WITO IDL Env trimer in complex wit... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
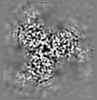
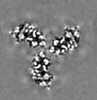
| Title | Cryo-EM structure of the HIV-1 WITO IDL Env trimer in complex with PGT122 Fab | |||||||||
 Map data Map data | sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CD4 / HIV-1 / SOSIP / Vaccine / gp120 / gp41 / VIRAL PROTEIN-IMMUNE SYSTEM complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated perturbation of host defense response / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / viral protein processing / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell ...symbiont-mediated perturbation of host defense response / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / viral protein processing / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity / membrane Similarity search - Function | |||||||||
| Biological species |   Human immunodeficiency virus 1 / Human immunodeficiency virus 1 /  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.5 Å | |||||||||
 Authors Authors | Gorman J / Kwong PD | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2024 Journal: Cell Rep / Year: 2024Title: Design of soluble HIV-1 envelope trimers free of covalent gp120-gp41 bonds with prevalent native-like conformation. Authors: Peng Zhang / Jason Gorman / Yaroslav Tsybovsky / Maolin Lu / Qingbo Liu / Vinay Gopan / Mamta Singh / Yin Lin / Huiyi Miao / Yuna Seo / Alice Kwon / Adam S Olia / Gwo-Yu Chuang / Hui Geng / ...Authors: Peng Zhang / Jason Gorman / Yaroslav Tsybovsky / Maolin Lu / Qingbo Liu / Vinay Gopan / Mamta Singh / Yin Lin / Huiyi Miao / Yuna Seo / Alice Kwon / Adam S Olia / Gwo-Yu Chuang / Hui Geng / Yen-Ting Lai / Tongqing Zhou / John R Mascola / Walther Mothes / Peter D Kwong / Paolo Lusso /   Abstract: Soluble HIV-1 envelope (Env) trimers may serve as effective vaccine immunogens. The widely utilized SOSIP trimers have been paramount for structural studies, but the disulfide bond they feature ...Soluble HIV-1 envelope (Env) trimers may serve as effective vaccine immunogens. The widely utilized SOSIP trimers have been paramount for structural studies, but the disulfide bond they feature between gp120 and gp41 constrains intersubunit mobility and may alter antigenicity. Here, we report an alternative strategy to generate stabilized soluble Env trimers free of covalent gp120-gp41 bonds. Stabilization was achieved by introducing an intrasubunit disulfide bond between the inner and outer domains of gp120, defined as interdomain lock (IDL). Correctly folded IDL trimers displaying a native-like antigenic profile were produced for HIV-1 Envs of different clades. Importantly, the IDL design abrogated CD4 binding while not affecting recognition by potent neutralizing antibodies to the CD4-binding site. By cryoelectron microscopy, IDL trimers were shown to adopt a closed prefusion configuration, while single-molecule fluorescence resonance energy transfer documented a high prevalence of native-like conformation. Thus, IDL trimers may be promising candidates as vaccine immunogens. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_44491.map.gz emd_44491.map.gz | 156 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-44491-v30.xml emd-44491-v30.xml emd-44491.xml emd-44491.xml | 25.4 KB 25.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_44491.png emd_44491.png | 87.1 KB | ||
| Masks |  emd_44491_msk_1.map emd_44491_msk_1.map | 166.4 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-44491.cif.gz emd-44491.cif.gz | 7.2 KB | ||
| Others |  emd_44491_additional_1.map.gz emd_44491_additional_1.map.gz emd_44491_additional_2.map.gz emd_44491_additional_2.map.gz emd_44491_half_map_1.map.gz emd_44491_half_map_1.map.gz emd_44491_half_map_2.map.gz emd_44491_half_map_2.map.gz | 32.7 MB 8 MB 154.4 MB 154.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-44491 http://ftp.pdbj.org/pub/emdb/structures/EMD-44491 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-44491 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-44491 | HTTPS FTP |
-Related structure data
| Related structure data |  9bf6MC  9berC  9bewC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_44491.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_44491.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0733 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_44491_msk_1.map emd_44491_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: unsharpened map
| File | emd_44491_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: resolve map
| File | emd_44491_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | resolve map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map A
| File | emd_44491_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map B
| File | emd_44491_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM Structure of the HIV-1 WITO IDL Env trimer in complex wit...
| Entire | Name: Cryo-EM Structure of the HIV-1 WITO IDL Env trimer in complex with PGT122 Fab |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM Structure of the HIV-1 WITO IDL Env trimer in complex wit...
| Supramolecule | Name: Cryo-EM Structure of the HIV-1 WITO IDL Env trimer in complex with PGT122 Fab type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
-Macromolecule #1: Envelope glycoprotein gp41
| Macromolecule | Name: Envelope glycoprotein gp41 / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
| Molecular weight | Theoretical: 17.381771 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: AVTLGAVFLG FLGAAGSTMG AASLTLTVQA RLLLSGIVQQ QSNLLRAPEA QQHMLQLTVW GIKQLQARVL AIERYLKDQQ LLGIWGCSG KLICTTTVPW NTSWSNKSYD YIWNNMTWMQ WEREIDNYTG FIYTLIEESQ NQQEKNELEL LELD UniProtKB: Envelope glycoprotein gp160 |
-Macromolecule #2: Envelope glycoprotein gp120
| Macromolecule | Name: Envelope glycoprotein gp120 / type: protein_or_peptide / ID: 2 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
| Molecular weight | Theoretical: 53.827133 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: AEQLWVTVYY GVPVWREANT TLFCASDAKA YDTEVHNVWA THACVPTDPN PQEVVMGNVT EDFNMWKNNM VEQMHEDIIS LWCQSLKPC VKLTPLCVTL HCTNVTISST NGSTANVTMR EEMKNCSFNT TTVIRDKIQK EYALFYKLDI VPIEGKNTNT S YRLINCNT ...String: AEQLWVTVYY GVPVWREANT TLFCASDAKA YDTEVHNVWA THACVPTDPN PQEVVMGNVT EDFNMWKNNM VEQMHEDIIS LWCQSLKPC VKLTPLCVTL HCTNVTISST NGSTANVTMR EEMKNCSFNT TTVIRDKIQK EYALFYKLDI VPIEGKNTNT S YRLINCNT SVITQACPKV SFEPIPIHYC APAGFAILKC NNKTFNGKGP CRNVSTVQCT HGIKPVVSTQ LLLNGSLAEE DI IIRSENF TNNGKNIIVQ LKEPVKINCT RPGNNTRRSI NIGPGRAFYA TGAIIGDIRK AHCNISTEQW NNTLTQIVDK LRE QFGN(UNK)K TIIFNQSSGG DPEVVMHTFN CGGEFFYCNS TQLFNSTWFN NGTSTWNSTA DNITLPCRIK QVINMWQEVG GCGAMYAPP IRGQIDCSSN ITGLILTRDG GSNSSQNETF RPGGGNMKDN WRSELYKYKV VKIEPLGIAP TRAKRRVVQR R RRRR UniProtKB: Envelope glycoprotein gp160 |
-Macromolecule #3: PGT122 heavy chain
| Macromolecule | Name: PGT122 heavy chain / type: protein_or_peptide / ID: 3 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 14.838731 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QVHLQESGPG LVKPSETLSL TCNVSGTLVR DNYWSWIRQP LGKQPEWIGY VHDSGDTNYN PSLKSRVHLS LDKSKNLVSL RLTGVTAAD SAIYYCATTK HGRRIYGVVA FKEWFTYFYM DVWGKGTSVT VSS |
-Macromolecule #4: PGT122 light chain
| Macromolecule | Name: PGT122 light chain / type: protein_or_peptide / ID: 4 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 11.423534 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: TFVSVAPGQT ARITCGEESL GSRSVIWYQQ RPGQAPSLII YNNNDRPSGI PDRFSGSPGS TFGTTATLTI TSVEAGDEAD YYCHIWDSR RPTNWVFGEG TTLIVL |
-Macromolecule #8: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 8 / Number of copies: 45 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #9: PHOSPHATE ION
| Macromolecule | Name: PHOSPHATE ION / type: ligand / ID: 9 / Number of copies: 1 / Formula: PO4 |
|---|---|
| Molecular weight | Theoretical: 94.971 Da |
| Chemical component information |  ChemComp-PO4: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Details: PBS |
| Grid | Model: C-flat-1.2/1.3 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 293 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Average exposure time: 10.0 sec. / Average electron dose: 70.77 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 4.7 µm / Nominal defocus min: 0.1 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | Chain - Source name: AlphaFold / Chain - Initial model type: in silico model |
|---|---|
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT |
| Output model |  PDB-9bf6: |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)