[English] 日本語
 Yorodumi
Yorodumi- EMDB-43490: Endogenous trans-translation complex with tmRNA*SmpB in the P sit... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Endogenous trans-translation complex with tmRNA*SmpB in the P site and alanyl-tRNA in the A site and deacyl-tRNA in the E site of E. coli 70S ribosome | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | endogenous tmRNA / tmRNA decoding / A-minor interactions / SmpB / cryo-EM / alanyl-tRNA / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationtrans-translation-dependent protein tagging / trans-translation / stringent response / misfolded RNA binding / Group I intron splicing / RNA folding / positive regulation of ribosome biogenesis / translational termination / DnaA-L2 complex / negative regulation of translational initiation ...trans-translation-dependent protein tagging / trans-translation / stringent response / misfolded RNA binding / Group I intron splicing / RNA folding / positive regulation of ribosome biogenesis / translational termination / DnaA-L2 complex / negative regulation of translational initiation / negative regulation of DNA-templated DNA replication initiation / mRNA regulatory element binding translation repressor activity / positive regulation of RNA splicing / cytosolic ribosome assembly / ribosome assembly / assembly of large subunit precursor of preribosome / transcription antitermination / regulation of cell growth / DNA-templated transcription termination / maintenance of translational fidelity / mRNA 5'-UTR binding / regulation of translation / large ribosomal subunit / ribosomal small subunit assembly / transferase activity / ribosome binding / ribosomal small subunit biogenesis / 5S rRNA binding / small ribosomal subunit / ribosomal large subunit assembly / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / negative regulation of translation / rRNA binding / structural constituent of ribosome / ribosome / translation / ribonucleoprotein complex / response to antibiotic / mRNA binding / RNA binding / zinc ion binding / membrane / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Teran D / Zhang Y / Korostelev AA | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Front Microbiol / Year: 2024 Journal: Front Microbiol / Year: 2024Title: Endogenous trans-translation structure visualizes the decoding of the first tmRNA alanine codon. Authors: David Teran / Ying Zhang / Andrei A Korostelev /  Abstract: Ribosomes stall on truncated or otherwise damaged mRNAs. Bacteria rely on ribosome rescue mechanisms to replenish the pool of ribosomes available for translation. Trans-translation, the main ribosome- ...Ribosomes stall on truncated or otherwise damaged mRNAs. Bacteria rely on ribosome rescue mechanisms to replenish the pool of ribosomes available for translation. Trans-translation, the main ribosome-rescue pathway, uses a circular hybrid transfer-messenger RNA (tmRNA) to restart translation and label the resulting peptide for degradation. Previous studies have visualized how tmRNA and its helper protein SmpB interact with the stalled ribosome to establish a new open reading frame. As tmRNA presents the first alanine codon via a non-canonical mRNA path in the ribosome, the incoming alanyl-tRNA must rearrange the tmRNA molecule to read the codon. Here, we describe cryo-EM analyses of an endogenous ribosome-tmRNA complex with tRNA accommodated in the A site. The flexible adenosine-rich tmRNA linker, which connects the mRNA-like domain with the codon, is stabilized by the minor groove of the canonically positioned anticodon stem of tRNA. This ribosome complex can also accommodate a tRNA near the E (exit) site, bringing insights into the translocation and dissociation of the tRNA that decoded the defective mRNA prior to tmRNA binding. Together, these structures uncover a key step of ribosome rescue, in which the ribosome starts translating the tmRNA reading frame. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_43490.map.gz emd_43490.map.gz | 620.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-43490-v30.xml emd-43490-v30.xml emd-43490.xml emd-43490.xml | 74 KB 74 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_43490.png emd_43490.png | 96.3 KB | ||
| Filedesc metadata |  emd-43490.cif.gz emd-43490.cif.gz | 14.1 KB | ||
| Others |  emd_43490_half_map_1.map.gz emd_43490_half_map_1.map.gz emd_43490_half_map_2.map.gz emd_43490_half_map_2.map.gz | 258.6 MB 258.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-43490 http://ftp.pdbj.org/pub/emdb/structures/EMD-43490 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43490 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43490 | HTTPS FTP |
-Related structure data
| Related structure data |  8vs9MC  8vsaC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_43490.map.gz / Format: CCP4 / Size: 669.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_43490.map.gz / Format: CCP4 / Size: 669.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.87 Å | ||||||||||||||||||||||||||||||||||||
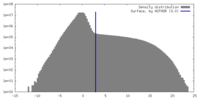
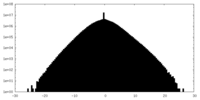
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_43490_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
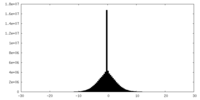
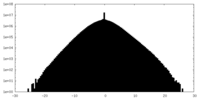
| Density Histograms |
-Half map: #1
| File | emd_43490_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
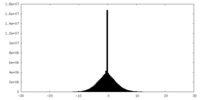
| Density Histograms |
- Sample components
Sample components
+Entire : CryoEM of tmRNA and SMPB bound to P site and alanyl-tRNA bound to...
+Supramolecule #1: CryoEM of tmRNA and SMPB bound to P site and alanyl-tRNA bound to...
+Macromolecule #1: 16S ribosomal RNA
+Macromolecule #2: 23S ribosomal RNA
+Macromolecule #3: 5S ribosomal RNA
+Macromolecule #4: A-tRNA
+Macromolecule #5: E-tRNA
+Macromolecule #59: TMRN
+Macromolecule #6: 50S ribosomal protein L2
+Macromolecule #7: 50S ribosomal protein L3
+Macromolecule #8: 50S ribosomal protein L4
+Macromolecule #9: 50S ribosomal protein L5
+Macromolecule #10: 50S ribosomal protein L6
+Macromolecule #11: Large ribosomal subunit protein bL9
+Macromolecule #12: 50S ribosomal protein L1
+Macromolecule #13: 50S ribosomal protein L10
+Macromolecule #14: 50S ribosomal protein L11
+Macromolecule #15: 50S ribosomal protein L13
+Macromolecule #16: 50S ribosomal protein L14
+Macromolecule #17: 50S ribosomal protein L15
+Macromolecule #18: 50S ribosomal protein L16
+Macromolecule #19: Large ribosomal subunit protein bL17
+Macromolecule #20: 50S ribosomal protein L18
+Macromolecule #21: 50S ribosomal protein L19
+Macromolecule #22: 50S ribosomal protein L20
+Macromolecule #23: Ribosomal protein L21
+Macromolecule #24: 50S ribosomal protein L22
+Macromolecule #25: 50S ribosomal protein L23
+Macromolecule #26: 50S ribosomal protein L24
+Macromolecule #27: 50S ribosomal protein L25
+Macromolecule #28: 50S ribosomal protein L27
+Macromolecule #29: 50S ribosomal protein L28
+Macromolecule #30: 50S ribosomal protein L29
+Macromolecule #31: Large ribosomal subunit protein uL30
+Macromolecule #32: Large ribosomal subunit protein bL31
+Macromolecule #33: 50S ribosomal protein L32
+Macromolecule #34: Large ribosomal subunit protein bL33
+Macromolecule #35: 50S ribosomal protein L34
+Macromolecule #36: 50S ribosomal protein L35
+Macromolecule #37: 50S ribosomal protein L36
+Macromolecule #38: 30S ribosomal protein S2
+Macromolecule #39: 30S ribosomal protein S3
+Macromolecule #40: 30S ribosomal protein S4
+Macromolecule #41: 30S ribosomal protein S5
+Macromolecule #42: 30S ribosomal protein S6
+Macromolecule #43: 30S ribosomal protein S7
+Macromolecule #44: 30S ribosomal protein S8
+Macromolecule #45: Small ribosomal subunit protein uS9
+Macromolecule #46: 30S ribosomal protein S10
+Macromolecule #47: 30S ribosomal protein S11
+Macromolecule #48: 30S ribosomal protein S12
+Macromolecule #49: 30S ribosomal protein S13
+Macromolecule #50: Small ribosomal subunit protein uS14
+Macromolecule #51: Small ribosomal subunit protein uS15
+Macromolecule #52: 30S ribosomal protein S16
+Macromolecule #53: Small ribosomal subunit protein uS17
+Macromolecule #54: 30S ribosomal protein S18
+Macromolecule #55: 30S ribosomal protein S19
+Macromolecule #56: 30S ribosomal protein S20
+Macromolecule #57: 30S ribosomal protein S21
+Macromolecule #58: SsrA-binding protein
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: OTHER |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK I |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 29.9 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.1 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)