[English] 日本語
 Yorodumi
Yorodumi- EMDB-43392: Structure of VCP in complex with an ATPase activator and ADP (D2 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of VCP in complex with an ATPase activator and ADP (D2 domains only, hexameric form) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | activator / complex / ATPase / AAA protein / HYDROLASE / HYDROLASE-ACTIVATOR complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of Lys63-specific deubiquitinase activity / flavin adenine dinucleotide catabolic process / positive regulation of oxidative phosphorylation / VCP-NSFL1C complex / endosome to lysosome transport via multivesicular body sorting pathway / protein-DNA covalent cross-linking repair / endoplasmic reticulum stress-induced pre-emptive quality control / cellular response to arsenite ion / Derlin-1 retrotranslocation complex / BAT3 complex binding ...positive regulation of Lys63-specific deubiquitinase activity / flavin adenine dinucleotide catabolic process / positive regulation of oxidative phosphorylation / VCP-NSFL1C complex / endosome to lysosome transport via multivesicular body sorting pathway / protein-DNA covalent cross-linking repair / endoplasmic reticulum stress-induced pre-emptive quality control / cellular response to arsenite ion / Derlin-1 retrotranslocation complex / BAT3 complex binding / positive regulation of protein K63-linked deubiquitination / deubiquitinase activator activity / aggresome assembly / regulation of protein localization to chromatin / vesicle-fusing ATPase / mitotic spindle disassembly / VCP-NPL4-UFD1 AAA ATPase complex / NADH metabolic process / cellular response to misfolded protein / stress granule disassembly / K48-linked polyubiquitin modification-dependent protein binding / positive regulation of mitochondrial membrane potential / negative regulation of protein localization to chromatin / ubiquitin-modified protein reader activity / retrograde protein transport, ER to cytosol / regulation of aerobic respiration / positive regulation of ATP biosynthetic process / regulation of synapse organization / ATPase complex / ubiquitin-specific protease binding / ubiquitin-like protein ligase binding / MHC class I protein binding / RHOH GTPase cycle / autophagosome maturation / HSF1 activation / polyubiquitin modification-dependent protein binding / proteasomal protein catabolic process / Protein methylation / endoplasmic reticulum to Golgi vesicle-mediated transport / translesion synthesis / interstrand cross-link repair / negative regulation of smoothened signaling pathway / ERAD pathway / ATP metabolic process / Attachment and Entry / endoplasmic reticulum unfolded protein response / viral genome replication / lipid droplet / proteasome complex / Josephin domain DUBs / N-glycan trimming in the ER and Calnexin/Calreticulin cycle / Hh mutants are degraded by ERAD / macroautophagy / Defective CFTR causes cystic fibrosis / Hedgehog ligand biogenesis / Translesion Synthesis by POLH / positive regulation of protein-containing complex assembly / ABC-family proteins mediated transport / establishment of protein localization / ADP binding / autophagy / Aggrephagy / cytoplasmic stress granule / positive regulation of non-canonical NF-kappaB signal transduction / positive regulation of protein catabolic process / activation of cysteine-type endopeptidase activity involved in apoptotic process / KEAP1-NFE2L2 pathway / azurophil granule lumen / double-strand break repair / Ovarian tumor domain proteases / positive regulation of proteasomal ubiquitin-dependent protein catabolic process / positive regulation of canonical Wnt signaling pathway / E3 ubiquitin ligases ubiquitinate target proteins / Neddylation / site of double-strand break / cellular response to heat / ubiquitin-dependent protein catabolic process / regulation of apoptotic process / protein phosphatase binding / proteasome-mediated ubiquitin-dependent protein catabolic process / secretory granule lumen / ficolin-1-rich granule lumen / Attachment and Entry / protein ubiquitination / protein domain specific binding / intracellular membrane-bounded organelle / DNA repair / glutamatergic synapse / lipid binding / ubiquitin protein ligase binding / DNA damage response / Neutrophil degranulation / endoplasmic reticulum membrane / perinuclear region of cytoplasm / endoplasmic reticulum / ATP hydrolysis activity / protein-containing complex / RNA binding / extracellular exosome / extracellular region Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
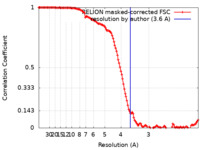
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Jones NH / Urnivicius L / Kapoor TM | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation | Journal: bioRxiv / Year: 2023 Title: Allosteric activation of VCP, a AAA unfoldase, by small molecule mimicry. Authors: N H Jones / Q Liu / L Urnavicius / N E Dahan / L E Vostal / T M Kapoor Abstract: The loss of function of AAA (ATPases associated with diverse cellular activities) mechanoenzymes has been linked to diseases, and small molecules that activate these proteins can be powerful tools to ...The loss of function of AAA (ATPases associated with diverse cellular activities) mechanoenzymes has been linked to diseases, and small molecules that activate these proteins can be powerful tools to probe mechanisms and test therapeutic hypotheses. Unlike chemical inhibitors that can bind a single conformational state to block enzyme activity, activator binding must be permissive to different conformational states needed for enzyme function. However, we do not know how AAA proteins can be activated by small molecules. Here, we focus on valosin-containing protein (VCP)/p97, a AAA unfoldase whose loss of function has been linked to protein aggregation-based disorders, to identify druggable sites for chemical activators. We identified VCP Activator 1 (VA1), a compound that dose-dependently stimulates VCP ATPase activity up to ∼3-fold. Our cryo-EM studies resulted in structures (∼2.9-3.5 Å-resolution) of VCP in apo and ADP-bound states, and revealed VA1 binding an allosteric pocket near the C-terminus in both states. Engineered mutations in the VA1 binding site confer resistance to VA1, and furthermore, modulate VCP activity to a similar level as VA1-mediated activation. The VA1 binding site can alternatively be occupied by a phenylalanine residue in the VCP C-terminal tail, a motif that is post-translationally modified and interacts with cofactors. Together, our findings uncover a druggable allosteric site and a mechanism of enzyme regulation that can be tuned through small molecule mimicry. SIGNIFICANCE: The loss of function of valosin-containing protein (VCP/p97), a mechanoenzyme from the AAA superfamily that hydrolyzes ATP and uses the released energy to extract or unfold substrate ...SIGNIFICANCE: The loss of function of valosin-containing protein (VCP/p97), a mechanoenzyme from the AAA superfamily that hydrolyzes ATP and uses the released energy to extract or unfold substrate proteins, is linked to protein aggregation-based disorders. However, druggable allosteric sites to activate VCP, or any AAA mechanoenzyme, have not been identified. Here, we report cryo-EM structures of VCP in two states in complex with VA1, a compound we identified that dose-dependently stimulates VCP's ATP hydrolysis activity. The VA1 binding site can also be occupied by a phenylalanine residue in the VCP C-terminal tail, suggesting that VA1 acts through mimicry of this interaction. Our study reveals a druggable allosteric site and a mechanism of enzyme regulation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_43392.map.gz emd_43392.map.gz | 177.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-43392-v30.xml emd-43392-v30.xml emd-43392.xml emd-43392.xml | 19.6 KB 19.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_43392_fsc.xml emd_43392_fsc.xml | 13 KB | Display |  FSC data file FSC data file |
| Images |  emd_43392.png emd_43392.png | 54.6 KB | ||
| Filedesc metadata |  emd-43392.cif.gz emd-43392.cif.gz | 6.7 KB | ||
| Others |  emd_43392_half_map_1.map.gz emd_43392_half_map_1.map.gz emd_43392_half_map_2.map.gz emd_43392_half_map_2.map.gz | 150 MB 150.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-43392 http://ftp.pdbj.org/pub/emdb/structures/EMD-43392 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43392 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43392 | HTTPS FTP |
-Validation report
| Summary document |  emd_43392_validation.pdf.gz emd_43392_validation.pdf.gz | 871.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_43392_full_validation.pdf.gz emd_43392_full_validation.pdf.gz | 871 KB | Display | |
| Data in XML |  emd_43392_validation.xml.gz emd_43392_validation.xml.gz | 20.6 KB | Display | |
| Data in CIF |  emd_43392_validation.cif.gz emd_43392_validation.cif.gz | 27.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-43392 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-43392 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-43392 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-43392 | HTTPS FTP |
-Related structure data
| Related structure data |  8vovMC  8vkuC  8vlsC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_43392.map.gz / Format: CCP4 / Size: 190.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_43392.map.gz / Format: CCP4 / Size: 190.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.035 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_43392_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_43392_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of VCP with ADP and ATPase activator small molecule VAA1
| Entire | Name: Complex of VCP with ADP and ATPase activator small molecule VAA1 |
|---|---|
| Components |
|
-Supramolecule #1: Complex of VCP with ADP and ATPase activator small molecule VAA1
| Supramolecule | Name: Complex of VCP with ADP and ATPase activator small molecule VAA1 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Transitional endoplasmic reticulum ATPase
| Macromolecule | Name: Transitional endoplasmic reticulum ATPase / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO / EC number: vesicle-fusing ATPase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 89.43682 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASGADSKGD DLSTAILKQK NRPNRLIVDE AINEDNSVVS LSQPKMDELQ LFRGDTVLLK GKKRREAVCI VLSDDTCSDE KIRMNRVVR NNLRVRLGDV ISIQPCPDVK YGKRIHVLPI DDTVEGITGN LFEVYLKPYF LEAYRPIRKG DIFLVRGGMR A VEFKVVET ...String: MASGADSKGD DLSTAILKQK NRPNRLIVDE AINEDNSVVS LSQPKMDELQ LFRGDTVLLK GKKRREAVCI VLSDDTCSDE KIRMNRVVR NNLRVRLGDV ISIQPCPDVK YGKRIHVLPI DDTVEGITGN LFEVYLKPYF LEAYRPIRKG DIFLVRGGMR A VEFKVVET DPSPYCIVAP DTVIHCEGEP IKREDEEESL NEVGYDDIGG CRKQLAQIKE MVELPLRHPA LFKAIGVKPP RG ILLYGPP GTGKTLIARA VANETGAFFF LINGPEIMSK LAGESESNLR KAFEEAEKNA PAIIFIDELD AIAPKREKTH GEV ERRIVS QLLTLMDGLK QRAHVIVMAA TNRPNSIDPA LRRFGRFDRE VDIGIPDATG RLEILQIHTK NMKLADDVDL EQVA NETHG HVGADLAALC SEAALQAIRK KMDLIDLEDE TIDAEVMNSL AVTMDDFRWA LSQSNPSALR ETVVEVPQVT WEDIG GLED VKRELQELVQ YPVEHPDKFL KFGMTPSKGV LFYGPPGCGK TLLAKAIANE CQANFISIKG PELLTMWFGE SEANVR EIF DKARQAAPCV LFFDELDSIA KARGGNIGDG GGAADRVINQ ILTEMDGMST KKNVFIIGAT NRPDIIDPAI LRPGRLD QL IYIPLPDEKS RVAILKANLR KSPVAKDVDL EFLAKMTNGF SGADLTEICQ RACKLAIRES IESEIRRERE RQTNPSAM E VEEDDPVPEI RRDHFEEAMR FARRSVSDND IRKYEMFAQT LQQSRGFGSF RFPSGNQGGA GPSQGSGGGT GGSVYTEDN DDDLYG UniProtKB: Transitional endoplasmic reticulum ATPase |
-Macromolecule #2: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 6 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Macromolecule #3: (3R)-N-[2-(ethylsulfanyl)phenyl]-3-(1-oxo-1,3-dihydro-2H-isoindol...
| Macromolecule | Name: (3R)-N-[2-(ethylsulfanyl)phenyl]-3-(1-oxo-1,3-dihydro-2H-isoindol-2-yl)butanamide type: ligand / ID: 3 / Number of copies: 6 / Formula: A1AC1 |
|---|---|
| Molecular weight | Theoretical: 354.466 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
Details: 50 mM K.HEPES pH 7.5, 25 mM KCl, 2.5 mM MgCl2, 2.5 mM GSH, 0.5% DMSO, 0.01% FOM | |||||||||||||||||||||
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. | |||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 38.3 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.5 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)