[English] 日本語
 Yorodumi
Yorodumi- EMDB-42993: DNA elongation complex (configuration 2) of Xenopus laevis DNA po... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | DNA elongation complex (configuration 2) of Xenopus laevis DNA polymerase alpha-primase | ||||||||||||
 Map data Map data | DNA elongation complex (configuration 2) of Xenopus laevis DNA polymerase alpha-primase | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Primase / DNA polymerase / chimeric RNA-DNA primer / RNA/DNA hybrid / DNA replication / DNA synthesis / REPLICATION / TRANSFERASE-DNA-RNA complex | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationalpha DNA polymerase:primase complex / lagging strand elongation / DNA replication, synthesis of primer / mitotic DNA replication initiation / DNA strand elongation involved in DNA replication / leading strand elongation / DNA replication origin binding / DNA replication initiation / double-strand break repair via nonhomologous end joining / nuclear matrix ...alpha DNA polymerase:primase complex / lagging strand elongation / DNA replication, synthesis of primer / mitotic DNA replication initiation / DNA strand elongation involved in DNA replication / leading strand elongation / DNA replication origin binding / DNA replication initiation / double-strand break repair via nonhomologous end joining / nuclear matrix / Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases / DNA-directed RNA polymerase activity / nuclear envelope / single-stranded DNA binding / 4 iron, 4 sulfur cluster binding / DNA-directed DNA polymerase / DNA-directed DNA polymerase activity / DNA replication / nucleotide binding / chromatin binding / chromatin / nucleolus / DNA binding / zinc ion binding / nucleoplasm / metal ion binding / nucleus Similarity search - Function | ||||||||||||
| Biological species | |||||||||||||
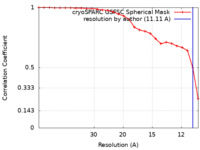
| Method | single particle reconstruction / cryo EM / Resolution: 11.11 Å | ||||||||||||
 Authors Authors | Mullins EA / Durie CL / Ohi MD / Chazin WJ / Eichman BF | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: A mechanistic model of primer synthesis from catalytic structures of DNA polymerase α-primase. Authors: Elwood A Mullins / Lauren E Salay / Clarissa L Durie / Noah P Bradley / Jane E Jackman / Melanie D Ohi / Walter J Chazin / Brandt F Eichman /  Abstract: The mechanism by which polymerase α-primase (polα-primase) synthesizes chimeric RNA-DNA primers of defined length and composition, necessary for replication fidelity and genome stability, is ...The mechanism by which polymerase α-primase (polα-primase) synthesizes chimeric RNA-DNA primers of defined length and composition, necessary for replication fidelity and genome stability, is unknown. Here, we report cryo-EM structures of Xenopus laevis polα-primase in complex with primed templates representing various stages of DNA synthesis. Our data show how interaction of the primase regulatory subunit with the primer 5' end facilitates handoff of the primer to polα and increases polα processivity, thereby regulating both RNA and DNA composition. The structures detail how flexibility within the heterotetramer enables synthesis across two active sites and provide evidence that termination of DNA synthesis is facilitated by reduction of polα and primase affinities for the varied conformations along the chimeric primer-template duplex. Together, these findings elucidate a critical catalytic step in replication initiation and provide a comprehensive model for primer synthesis by polα-primase. #1:  Journal: To Be Published Journal: To Be PublishedTitle: A mechanistic model of primer synthesis from catalytic structures of DNA polymerase alpha-primase Authors: Mullins EA / Salay LE / Durie CL / Bradley NP / Jackman JE / Ohi MD / Chazin WJ / Eichman BF | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42993.map.gz emd_42993.map.gz | 917 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42993-v30.xml emd-42993-v30.xml emd-42993.xml emd-42993.xml | 28.4 KB 28.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_42993_fsc.xml emd_42993_fsc.xml | 2.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_42993.png emd_42993.png | 25.7 KB | ||
| Filedesc metadata |  emd-42993.cif.gz emd-42993.cif.gz | 8.6 KB | ||
| Others |  emd_42993_half_map_1.map.gz emd_42993_half_map_1.map.gz emd_42993_half_map_2.map.gz emd_42993_half_map_2.map.gz | 952.5 KB 952.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42993 http://ftp.pdbj.org/pub/emdb/structures/EMD-42993 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42993 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42993 | HTTPS FTP |
-Related structure data
| Related structure data |  8v6jMC  8g99C  8g9fC  8g9lC  8g9nC  8g9oC  8ucuC  8ucvC  8ucwC  8v5mC  8v5nC  8v5oC  8v6gC  8v6hC  8v6iC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_42993.map.gz / Format: CCP4 / Size: 1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42993.map.gz / Format: CCP4 / Size: 1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DNA elongation complex (configuration 2) of Xenopus laevis DNA polymerase alpha-primase | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 5.11875 Å | ||||||||||||||||||||||||||||||||||||
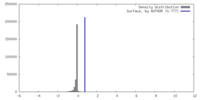
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: DNA elongation complex (configuration 2) of Xenopus laevis...
| File | emd_42993_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DNA elongation complex (configuration 2) of Xenopus laevis DNA polymerase alpha-primase (half 1) | ||||||||||||
| Projections & Slices |
| ||||||||||||
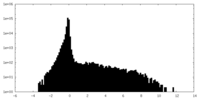
| Density Histograms |
-Half map: DNA elongation complex (configuration 2) of Xenopus laevis...
| File | emd_42993_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DNA elongation complex (configuration 2) of Xenopus laevis DNA polymerase alpha-primase (half 2) | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Polymerase alpha-primase with a DNA elongation substrate
+Supramolecule #1: Polymerase alpha-primase with a DNA elongation substrate
+Supramolecule #2: Polymerase alpha
+Supramolecule #3: Primase
+Supramolecule #4: DNA elongation substrate
+Macromolecule #1: DNA polymerase alpha catalytic subunit
+Macromolecule #2: DNA polymerase alpha subunit B
+Macromolecule #3: DNA primase large subunit
+Macromolecule #4: DNA primase
+Macromolecule #5: DNA template
+Macromolecule #6: RNA-DNA primer
+Macromolecule #7: ZINC ION
+Macromolecule #8: 2'-DEOXYGUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #9: MAGNESIUM ION
+Macromolecule #10: IRON/SULFUR CLUSTER
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.9 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Number grids imaged: 3 / Number real images: 13641 / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 45000 |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT | ||||||
| Output model |  PDB-8v6j: |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)