[English] 日本語
 Yorodumi
Yorodumi- EMDB-42268: Cryo-EM Structure of the Ro5256390-bound hTA1-Gs heterotrimer sig... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
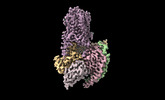
| Title | Cryo-EM Structure of the Ro5256390-bound hTA1-Gs heterotrimer signaling complex | |||||||||||||||
 Map data Map data | Primary Map | |||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | GPCR / Signaling Complex / Trace Amine-associated Receptor / MEMBRANE PROTEIN | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationadenylate cyclase regulator activity / regulation of parathyroid hormone secretion / COPI-coated Golgi to ER transport vesicle / post-embryonic body morphogenesis / Amine ligand-binding receptors / response to parathyroid hormone / trace-amine receptor activity / hair follicle placode formation / genomic imprinting / sensory perception of chemical stimulus ...adenylate cyclase regulator activity / regulation of parathyroid hormone secretion / COPI-coated Golgi to ER transport vesicle / post-embryonic body morphogenesis / Amine ligand-binding receptors / response to parathyroid hormone / trace-amine receptor activity / hair follicle placode formation / genomic imprinting / sensory perception of chemical stimulus / tissue homeostasis / mu-type opioid receptor binding / developmental growth / endochondral ossification / positive regulation of sodium ion transport / corticotropin-releasing hormone receptor 1 binding / D1 dopamine receptor binding / embryonic cranial skeleton morphogenesis / energy reserve metabolic process / positive regulation of osteoclast differentiation / beta2-adrenergic receptor activity / positive regulation of mini excitatory postsynaptic potential / embryonic hindlimb morphogenesis / positive regulation of cAMP-dependent protein kinase activity / positive regulation of AMPA receptor activity / cartilage development / positive regulation of autophagosome maturation / norepinephrine binding / norepinephrine-epinephrine-mediated vasodilation involved in regulation of systemic arterial blood pressure / heat generation / Adrenoceptors / alkylglycerophosphoethanolamine phosphodiesterase activity / activation of transmembrane receptor protein tyrosine kinase activity / beta-2 adrenergic receptor binding / negative regulation of smooth muscle contraction / positive regulation of lipophagy / skin development / negative regulation of multicellular organism growth / negative regulation of G protein-coupled receptor signaling pathway / response to psychosocial stress / endosome to lysosome transport / adrenergic receptor signaling pathway / cellular response to glucagon stimulus / diet induced thermogenesis / neuronal dense core vesicle / adenylate cyclase-activating adrenergic receptor signaling pathway / positive regulation of protein kinase A signaling / adenylate cyclase activator activity / adenylate cyclase binding / smooth muscle contraction / G-protein alpha-subunit binding / alpha-tubulin binding / regulation of signal transduction / trans-Golgi network membrane / bone resorption / potassium channel regulator activity / positive regulation of osteoblast differentiation / positive regulation of bone mineralization / brown fat cell differentiation / regulation of sodium ion transport / ruffle / insulin-like growth factor receptor binding / ionotropic glutamate receptor binding / negative regulation of blood pressure / bone development / response to cold / post-embryonic development / receptor-mediated endocytosis / G-protein beta/gamma-subunit complex binding / G protein activity / platelet aggregation / skeletal system development / cognition / G protein-coupled receptor activity / adenylate cyclase-activating G protein-coupled receptor signaling pathway / electron transport chain / clathrin-coated endocytic vesicle membrane / positive regulation of protein serine/threonine kinase activity / positive regulation of insulin secretion / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / Olfactory Signaling Pathway / multicellular organism growth / Activation of the phototransduction cascade / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Glucagon signaling in metabolic regulation / recycling endosome / G protein-coupled acetylcholine receptor signaling pathway / sarcolemma / G-protein activation / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G beta:gamma signalling through CDC42 / Vasopressin regulates renal water homeostasis via Aquaporins / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Glucagon-type ligand receptors Similarity search - Function | |||||||||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /   | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.35 Å | |||||||||||||||
 Authors Authors | Zilberg G / Warren AL / Parpounas AK / Wacker D | |||||||||||||||
| Funding support |  United States, 4 items United States, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Molecular basis of human trace amine-associated receptor 1 activation. Authors: Gregory Zilberg / Alexandra K Parpounas / Audrey L Warren / Shifan Yang / Daniel Wacker /  Abstract: The human trace amine-associated receptor 1 (hTAAR1, hTA1) is a key regulator of monoaminergic neurotransmission and the actions of psychostimulants. Despite preclinical research demonstrating its ...The human trace amine-associated receptor 1 (hTAAR1, hTA1) is a key regulator of monoaminergic neurotransmission and the actions of psychostimulants. Despite preclinical research demonstrating its tractability as a drug target, its molecular mechanisms of activation remain unclear. Moreover, poorly understood pharmacological differences between rodent and human TA1 complicate the translation of findings from preclinical disease models into novel pharmacotherapies. To elucidate hTA1's mechanisms on the molecular scale and investigate the underpinnings of its divergent pharmacology from rodent orthologs, we herein report the structure of the human TA1 receptor in complex with a Gαs heterotrimer. Our structure reveals shared structural elements with other TAARs, as well as with its closest monoaminergic orthologue, the serotonin receptor 5-HT4R. We further find that a single mutation dramatically shifts the selectivity of hTA1 towards that of its rodent orthologues, and report on the effects of substituting residues to those found in serotonin and dopamine receptors. Strikingly, we also discover that the atypical antipsychotic medication and pan-monoaminergic antagonist asenapine potently and efficaciously activates hTA1. Together our studies provide detailed insight into hTA1 structure and function, contrast its molecular pharmacology with that of related receptors, and uncover off-target activities of monoaminergic drugs at hTA1. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42268.map.gz emd_42268.map.gz | 31.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42268-v30.xml emd-42268-v30.xml emd-42268.xml emd-42268.xml | 20.4 KB 20.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_42268.png emd_42268.png | 44.3 KB | ||
| Filedesc metadata |  emd-42268.cif.gz emd-42268.cif.gz | 7 KB | ||
| Others |  emd_42268_half_map_1.map.gz emd_42268_half_map_1.map.gz emd_42268_half_map_2.map.gz emd_42268_half_map_2.map.gz | 59.4 MB 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42268 http://ftp.pdbj.org/pub/emdb/structures/EMD-42268 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42268 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42268 | HTTPS FTP |
-Validation report
| Summary document |  emd_42268_validation.pdf.gz emd_42268_validation.pdf.gz | 727.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_42268_full_validation.pdf.gz emd_42268_full_validation.pdf.gz | 726.9 KB | Display | |
| Data in XML |  emd_42268_validation.xml.gz emd_42268_validation.xml.gz | 12.4 KB | Display | |
| Data in CIF |  emd_42268_validation.cif.gz emd_42268_validation.cif.gz | 14.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42268 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42268 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42268 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42268 | HTTPS FTP |
-Related structure data
| Related structure data |  8uhbMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_42268.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42268.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Primary Map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.069 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half map 1
| File | emd_42268_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 2
| File | emd_42268_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : hTA1-Gs-Nb35 complex (Ro5256390)
| Entire | Name: hTA1-Gs-Nb35 complex (Ro5256390) |
|---|---|
| Components |
|
-Supramolecule #1: hTA1-Gs-Nb35 complex (Ro5256390)
| Supramolecule | Name: hTA1-Gs-Nb35 complex (Ro5256390) / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Soluble cytochrome b562,Beta-2 adrenergic receptor,Trace amine-as...
| Macromolecule | Name: Soluble cytochrome b562,Beta-2 adrenergic receptor,Trace amine-associated receptor 1 type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 60.019816 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDAKLQTMH HHHHHHHHHE NLYFQGGTTA DLEDNWETLN DNLKVIEKAD NAAQVKDALT KMRAAALDA QKATPPKLED KSPDSPEMKD FRHGFDILVG QIDDALKLAN EGKVKEAQAA AEQLKTTRNA YIQKYLMGQP G NGSAFLLA ...String: MKTIIALSYI FCLVFADYKD DDDAKLQTMH HHHHHHHHHE NLYFQGGTTA DLEDNWETLN DNLKVIEKAD NAAQVKDALT KMRAAALDA QKATPPKLED KSPDSPEMKD FRHGFDILVG QIDDALKLAN EGKVKEAQAA AEQLKTTRNA YIQKYLMGQP G NGSAFLLA PNRSHAPDHD VTQQRDEMMP FCHNIINISC VKNNWSNDVR ASLYSLMVLI ILTTLVGNLI VIVSISHFKQ LH TPTNWLI HSMATVDFLL GCLVMPYSMV RSAEHCWYFG EVFCKIHTST DIMLSSASIW HLSFISIDRY YAVCDPLRYK AKM NILVIC VMIFISWSVP AVFAFGMIFL ELNFKGAEEI YYKHVHCRGG CSVFFSKISG VLTFMTSFYI PGSIMLCVYY RIYL IAKEQ ARLISDANQK LQIGLEMKNG ISQSKERKAV KTLGIVMGVF LICWCPFFIC TVMDPFLHYI IPPTLNDVLI WFGYL NSTF NPMVYAFFYP WFRKALKMML FGKIFQKDSS RCKLFLELSS UniProtKB: Soluble cytochrome b562, Beta-2 adrenergic receptor, Trace amine-associated receptor 1 |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.418086 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MHHHHHHLEV LFQGPGSSGS ELDQLRQEAE QLKNQIRDAR KACADATLSQ ITNNIDPVGR IQMRTRRTLR GHLAKIYAMH WGTDSRLLV SASQDGKLII WDSYTTNKVH AIPLRSSWVM TCAYAPSGNY VACGGLDNIC SIYNLKTREG NVRVSRELAG H TGYLSCCR ...String: MHHHHHHLEV LFQGPGSSGS ELDQLRQEAE QLKNQIRDAR KACADATLSQ ITNNIDPVGR IQMRTRRTLR GHLAKIYAMH WGTDSRLLV SASQDGKLII WDSYTTNKVH AIPLRSSWVM TCAYAPSGNY VACGGLDNIC SIYNLKTREG NVRVSRELAG H TGYLSCCR FLDDNQIVTS SGDTTCALWD IETGQQTTTF TGHTGDVMSL SLAPDTRLFV SGACDASAKL WDVREGMCRQ TF TGHESDI NAICFFPNGN AFATGSDDAT CRLFDLRADQ ELMTYSHDNI ICGITSVSFS KSGRLLLAGY DDFNCNVWDA LKA DRAGVL AGHDNRVSCL GVTDDGMAVA TGSWDSFLKI WN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 45.803598 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG EGGEEDPQAA RSNSDGEKA TKVQDIKNNL KEAIETIVAA MSNLVPPVEL ANPENQFRVD YILSVMNVPN FDFPPEFYEH AKALWEDEGV R ACYERSNE ...String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG EGGEEDPQAA RSNSDGEKA TKVQDIKNNL KEAIETIVAA MSNLVPPVEL ANPENQFRVD YILSVMNVPN FDFPPEFYEH AKALWEDEGV R ACYERSNE YQLIDCAQYF LDKIDVIKQA EYMPTEQDLL RCRVLTSGIF ETKFQVDKVN FHMFDVGAQR DERRKWIQCF ND VTAIIFV VASSSYNMVI REDNQTNRLQ EALKLFDSIW NNKWLRDTSV ILFLNKQDLL AEKVLAGKSK IEDYFPEFAR YTT PEDATP EPGEDPRVTR AKYFIRDEFL RISTASGDGR HYCYPHFTCS VDTENIRRVF NDCRDIIQRM HLRQYELL UniProtKB: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #5: Nanobody 35
| Macromolecule | Name: Nanobody 35 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 14.71432 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: QVQLQESGGG LVQPGGSLRL SCAASGFTFS NYKMNWVRQA PGKGLEWVSD ISQSGASISY TGSVKGRFTI SRDNAKNTLY LQMNSLKPE DTAVYYCARC PAPFTRDCFD VTSTTYAYRG QGTQVTVSSH HHHHH |
-Macromolecule #6: (2R,4S)-4-[(2S)-2-phenylbutyl]-1,3-oxazolidin-2-amine
| Macromolecule | Name: (2R,4S)-4-[(2S)-2-phenylbutyl]-1,3-oxazolidin-2-amine / type: ligand / ID: 6 / Number of copies: 1 / Formula: WV8 |
|---|---|
| Molecular weight | Theoretical: 218.295 Da |
| Chemical component information |  ChemComp-WV8: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.2 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 |
| Vitrification | Cryogen name: ETHANE / Instrument: LEICA EM GP |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 53.88 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.35 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 626370 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
 Movie
Movie Controller
Controller



























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)