+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Klebsiella pneumoniae DyP peroxidase-loaded encapsulin shell | |||||||||
 Map data Map data | Klebsiella pneumoniae DyP peroxidase-loaded encapsulin shell | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Encapsulin / nanocompartment / DyP peroxidase / VIRUS LIKE PARTICLE | |||||||||
| Biological species |  Klebsiella pneumoniae (bacteria) Klebsiella pneumoniae (bacteria) | |||||||||
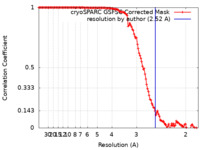
| Method | single particle reconstruction / cryo EM / Resolution: 2.52 Å | |||||||||
 Authors Authors | Andreas MP / Jones JA / Giessen TW | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Structural basis for peroxidase encapsulation inside the encapsulin from the Gram-negative pathogen Klebsiella pneumoniae. Authors: Jesse A Jones / Michael P Andreas / Tobias W Giessen /  Abstract: Encapsulins are self-assembling protein nanocompartments capable of selectively encapsulating dedicated cargo proteins, including enzymes involved in iron storage, sulfur metabolism, and stress ...Encapsulins are self-assembling protein nanocompartments capable of selectively encapsulating dedicated cargo proteins, including enzymes involved in iron storage, sulfur metabolism, and stress resistance. They represent a unique compartmentalization strategy used by many pathogens to facilitate specialized metabolic capabilities. Encapsulation is mediated by specific cargo protein motifs known as targeting peptides (TPs), though the structural basis for encapsulation of the largest encapsulin cargo class, dye-decolorizing peroxidases (DyPs), is currently unknown. Here, we characterize a DyP-containing encapsulin from the enterobacterial pathogen Klebsiella pneumoniae. By combining cryo-electron microscopy with TP and TP-binding site mutagenesis, we elucidate the molecular basis for cargo encapsulation. TP binding is mediated by cooperative hydrophobic and ionic interactions as well as shape complementarity. Our results expand the molecular understanding of enzyme encapsulation inside protein nanocompartments and lay the foundation for rationally modulating encapsulin cargo loading for biomedical and biotechnological applications. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41905.map.gz emd_41905.map.gz | 204.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41905-v30.xml emd-41905-v30.xml emd-41905.xml emd-41905.xml | 18.7 KB 18.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41905_fsc.xml emd_41905_fsc.xml | 12.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_41905.png emd_41905.png | 158.8 KB | ||
| Filedesc metadata |  emd-41905.cif.gz emd-41905.cif.gz | 6.1 KB | ||
| Others |  emd_41905_half_map_1.map.gz emd_41905_half_map_1.map.gz emd_41905_half_map_2.map.gz emd_41905_half_map_2.map.gz | 200.5 MB 200.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41905 http://ftp.pdbj.org/pub/emdb/structures/EMD-41905 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41905 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41905 | HTTPS FTP |
-Validation report
| Summary document |  emd_41905_validation.pdf.gz emd_41905_validation.pdf.gz | 1.2 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_41905_full_validation.pdf.gz emd_41905_full_validation.pdf.gz | 1.2 MB | Display | |
| Data in XML |  emd_41905_validation.xml.gz emd_41905_validation.xml.gz | 21.4 KB | Display | |
| Data in CIF |  emd_41905_validation.cif.gz emd_41905_validation.cif.gz | 27.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41905 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41905 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41905 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41905 | HTTPS FTP |
-Related structure data
| Related structure data |  8u50MC  8u4zC  8u51C M: atomic model generated by this map C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41905.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41905.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Klebsiella pneumoniae DyP peroxidase-loaded encapsulin shell | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.91 Å | ||||||||||||||||||||||||||||||||||||
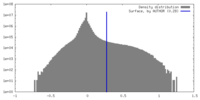
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half Map 1
| File | emd_41905_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 | ||||||||||||
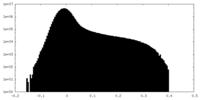
| Projections & Slices |
| ||||||||||||
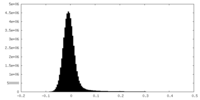
| Density Histograms |
-Half map: Half Map 2
| File | emd_41905_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
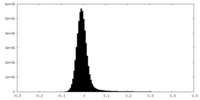
| Density Histograms |
- Sample components
Sample components
-Entire : Klebsiella pneumoniae DyP peroxidase-loaded encapsulin shell
| Entire | Name: Klebsiella pneumoniae DyP peroxidase-loaded encapsulin shell |
|---|---|
| Components |
|
-Supramolecule #1: Klebsiella pneumoniae DyP peroxidase-loaded encapsulin shell
| Supramolecule | Name: Klebsiella pneumoniae DyP peroxidase-loaded encapsulin shell type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Klebsiella pneumoniae (bacteria) Klebsiella pneumoniae (bacteria) |
-Supramolecule #2: Klebsiella pneumoniae family 1 encapsulin shell
| Supramolecule | Name: Klebsiella pneumoniae family 1 encapsulin shell / type: complex / ID: 2 / Parent: 1 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Klebsiella pneumoniae (bacteria) Klebsiella pneumoniae (bacteria) |
-Supramolecule #3: Klebsiella pneumoniae encapsulated DyP peroxidase
| Supramolecule | Name: Klebsiella pneumoniae encapsulated DyP peroxidase / type: complex / ID: 3 / Parent: 2 |
|---|---|
| Source (natural) | Organism:  Klebsiella pneumoniae (bacteria) Klebsiella pneumoniae (bacteria) |
-Macromolecule #1: Klebsiella pneumoniae family 1 encapsulin shell
| Macromolecule | Name: Klebsiella pneumoniae family 1 encapsulin shell / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Klebsiella pneumoniae (bacteria) Klebsiella pneumoniae (bacteria) |
| Molecular weight | Theoretical: 28.841129 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MNNLHRELAP VSDAAWEQIE EEASRTLKRF LAARRVVDVS DPQGPAFSAV GTGHVTRLEG PGDSVGAVKR QSQPVVEFRV PFILTRQAI DDVERGSQDS DWSPLKEAAR KIAGAEDRAV FDGYAAAGIG GIRPQSSNSP LTLPVAASGY PDVIARALDQ L RVAGVNGP ...String: MNNLHRELAP VSDAAWEQIE EEASRTLKRF LAARRVVDVS DPQGPAFSAV GTGHVTRLEG PGDSVGAVKR QSQPVVEFRV PFILTRQAI DDVERGSQDS DWSPLKEAAR KIAGAEDRAV FDGYAAAGIG GIRPQSSNSP LTLPVAASGY PDVIARALDQ L RVAGVNGP YHLVLGENAY TLITSGNEDG YPVLQHIHRL IDGEIVWAPA IEGGVLLSTR GGDFAMDIGQ DISIGYLSHT AT HVELYLQ ESFTFRTLTS EAVVSLLPSE D |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3.0 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
Details: 20 mM Tris pH 7.5, 150 mM NaCl | |||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. / Pretreatment - Atmosphere: AIR / Details: Glow discharged for 60 seconds at 5 mA | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295.15 K / Instrument: FEI VITROBOT MARK IV / Details: Blot force: 20 Blot time: 4 seconds. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3710 pixel / Digitization - Dimensions - Height: 3838 pixel / Number grids imaged: 1 / Number real images: 1183 / Average exposure time: 5.0 sec. / Average electron dose: 41.67 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 45000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| Details | Initial fitting was performed using ChimeraX v1.2.5. The model was then manually refined and mutated using Coot v9.8.1 followed by real-space refinement using Phenix v1.20.1-4487-000. |
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT / Overall B value: 95.2 / Target criteria: Cross-correlation coefficient |
| Output model |  PDB-8u50: |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)