+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | TRPV1 in nanodisc bound with PIP2-Br4 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | TRPV1 in nanodisc bound with PIP2-Br4 MEMBRANE PROTEIN / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of iodide transmembrane transport / positive regulation of membrane depolarization / negative regulation of establishment of blood-brain barrier / response to capsazepine / sensory perception of mechanical stimulus / peptide secretion / cellular response to temperature stimulus / temperature-gated ion channel activity / positive regulation of sensory perception of pain / detection of chemical stimulus involved in sensory perception of pain ...negative regulation of iodide transmembrane transport / positive regulation of membrane depolarization / negative regulation of establishment of blood-brain barrier / response to capsazepine / sensory perception of mechanical stimulus / peptide secretion / cellular response to temperature stimulus / temperature-gated ion channel activity / positive regulation of sensory perception of pain / detection of chemical stimulus involved in sensory perception of pain / positive regulation of renal sodium excretion / TRP channels / negative regulation of axon regeneration / positive regulation of cardiac muscle cell differentiation / smooth muscle contraction involved in micturition / fever generation / detection of temperature stimulus involved in thermoception / thermoception / urinary bladder smooth muscle contraction / response to pH / glutamate secretion / monoatomic cation transmembrane transporter activity / negative regulation of systemic arterial blood pressure / excitatory extracellular ligand-gated monoatomic ion channel activity / dendritic spine membrane / negative regulation of heart rate / response to acidic pH / positive regulation of urine volume / response to pain / diet induced thermogenesis / cellular response to alkaloid / temperature homeostasis / cellular response to cytokine stimulus / cellular response to ATP / intracellularly gated calcium channel activity / detection of temperature stimulus involved in sensory perception of pain / negative regulation of mitochondrial membrane potential / calcium ion import across plasma membrane / behavioral response to pain / positive regulation of vasoconstriction / monoatomic ion channel activity / ligand-gated monoatomic ion channel activity / monoatomic cation channel activity / cellular response to acidic pH / extracellular ligand-gated monoatomic ion channel activity / phosphatidylinositol binding / sensory perception of pain / axon terminus / positive regulation of excitatory postsynaptic potential / sarcoplasmic reticulum / lipid metabolic process / phosphoprotein binding / microglial cell activation / cellular response to nerve growth factor stimulus / cellular response to growth factor stimulus / response to peptide hormone / GABA-ergic synapse / cellular response to tumor necrosis factor / calcium ion transmembrane transport / calcium channel activity / positive regulation of nitric oxide biosynthetic process / calcium ion transport / transmembrane signaling receptor activity / cellular response to heat / sensory perception of taste / response to heat / positive regulation of cytosolic calcium ion concentration / monoatomic ion transmembrane transport / protein homotetramerization / calmodulin binding / postsynaptic membrane / neuron projection / positive regulation of apoptotic process / external side of plasma membrane / neuronal cell body / dendrite / negative regulation of transcription by RNA polymerase II / ATP binding / membrane / metal ion binding / identical protein binding / nucleus / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
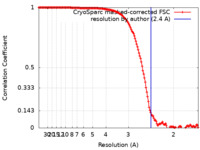
| Method | single particle reconstruction / cryo EM / Resolution: 2.4 Å | |||||||||
 Authors Authors | Arnold WR / Julius D / Cheng Y | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Structural basis of TRPV1 modulation by endogenous bioactive lipids. Authors: William R Arnold / Adamo Mancino / Frank R Moss / Adam Frost / David Julius / Yifan Cheng /  Abstract: TRP ion channels are modulated by phosphoinositide lipids, but the underlying structural mechanisms remain unclear. The capsaicin- and heat-activated receptor, TRPV1, has served as a model for ...TRP ion channels are modulated by phosphoinositide lipids, but the underlying structural mechanisms remain unclear. The capsaicin- and heat-activated receptor, TRPV1, has served as a model for deciphering lipid modulation, which is relevant to understanding how pro-algesic agents enhance channel activity in the setting of inflammatory pain. Identification of a pocket within the TRPV1 transmembrane core has provided initial clues as to how phosphoinositide lipids bind to and regulate the channel. Here we show that this regulatory pocket in rat TRPV1 can accommodate diverse lipid species, including the inflammatory lipid lysophosphatidic acid, whose actions are determined by their specific modes of binding. Furthermore, we show that an empty-pocket channel lacking an endogenous phosphoinositide lipid assumes an agonist-like state, even at low temperature, substantiating the concept that phosphoinositide lipids serve as negative TRPV1 modulators whose ejection from the binding pocket is a critical step toward activation by thermal or chemical stimuli. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41873.map.gz emd_41873.map.gz | 258.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41873-v30.xml emd-41873-v30.xml emd-41873.xml emd-41873.xml | 15.3 KB 15.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41873_fsc.xml emd_41873_fsc.xml | 14.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_41873.png emd_41873.png | 88.9 KB | ||
| Filedesc metadata |  emd-41873.cif.gz emd-41873.cif.gz | 6 KB | ||
| Others |  emd_41873_half_map_1.map.gz emd_41873_half_map_1.map.gz emd_41873_half_map_2.map.gz emd_41873_half_map_2.map.gz | 254.1 MB 254.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41873 http://ftp.pdbj.org/pub/emdb/structures/EMD-41873 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41873 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41873 | HTTPS FTP |
-Related structure data
| Related structure data |  8u43MC  8t0cC  8t0eC  8t0yC  8t10C  8t3lC  8t3mC  8u2zC  8u30C  8u3aC  8u3cC  8u3jC  8u3lC  8u4dC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41873.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41873.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.835 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_41873_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
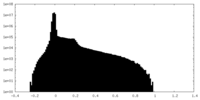
| Projections & Slices |
| ||||||||||||
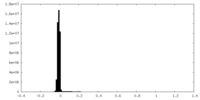
| Density Histograms |
-Half map: #1
| File | emd_41873_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
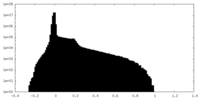
| Projections & Slices |
| ||||||||||||
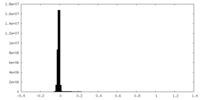
| Density Histograms |
- Sample components
Sample components
-Entire : TRPV1 in nanodisc bound with PIP2-Br4
| Entire | Name: TRPV1 in nanodisc bound with PIP2-Br4 |
|---|---|
| Components |
|
-Supramolecule #1: TRPV1 in nanodisc bound with PIP2-Br4
| Supramolecule | Name: TRPV1 in nanodisc bound with PIP2-Br4 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 688 KDa |
-Macromolecule #1: Transient receptor potential cation channel subfamily V member 1
| Macromolecule | Name: Transient receptor potential cation channel subfamily V member 1 type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 72.888227 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MGSRLYDRRS IFDAVAQSNC QELESLLPFL QRSKKRLTDS EFKDPETGKT CLLKAMLNLH NGQNDTIALL LDVARKTDSL KQFVNASYT DSYYKGQTAL HIAIERRNMT LVTLLVENGA DVQAAANGDF FKKTKGRPGF YFGELPLSLA ACTNQLAIVK F LLQNSWQP ...String: MGSRLYDRRS IFDAVAQSNC QELESLLPFL QRSKKRLTDS EFKDPETGKT CLLKAMLNLH NGQNDTIALL LDVARKTDSL KQFVNASYT DSYYKGQTAL HIAIERRNMT LVTLLVENGA DVQAAANGDF FKKTKGRPGF YFGELPLSLA ACTNQLAIVK F LLQNSWQP ADISARDSVG NTVLHALVEV ADNTVDNTKF VTSMYNEILI LGAKLHPTLK LEEITNRKGL TPLALAASSG KI GVLAYIL QREIHEPECR HLSRKFTEWA YGPVHSSLYD LSCIDTCEKN SVLEVIAYSS SETPNRHDML LVEPLNRLLQ DKW DRFVKR IFYFNFFVYC LYMIIFTAAA YYRPVEGLPP YKLKNTVGDY FRVTGEILSV SGGVYFFFRG IQYFLQRRPS LKSL FVDSY SEILFFVQSL FMLVSVVLYF SQRKEYVASM VFSLAMGWTN MLYYTRGFQQ MGIYAVMIEK MILRDLCRFM FVYLV FLFG FSTAVVTLIE DGKYNSLYST CLELFKFTIG MGDLEFTENY DFKAVFIILL LAYVILTYIL LLNMLIALMG ETVNKI AQE SKNIWKLQRA ITILDTEKSF LKCMRKAFRS GKLLQVGFTP DGKDDYRWCF RVDEVNWTTW NTNVGIINED PG UniProtKB: Transient receptor potential cation channel subfamily V member 1 |
-Macromolecule #2: (2S)-2-[(9,10-dibromooctadecanoyl)oxy]-3-{[(S)-hydroxy{[(1R,2R,3S...
| Macromolecule | Name: (2S)-2-[(9,10-dibromooctadecanoyl)oxy]-3-{[(S)-hydroxy{[(1R,2R,3S,4R,5R,6S)-2,3,6-trihydroxy-4,5-bis(phosphonooxy)cyclohexyl]oxy}phosphoryl]oxy}propyl (9R,10S)-9,10-dibromooctadecanoate type: ligand / ID: 2 / Number of copies: 4 / Formula: V5H |
|---|---|
| Molecular weight | Theoretical: 1.342682 KDa |
-Macromolecule #3: 1,2-DIOLEOYL-SN-GLYCERO-3-PHOSPHOCHOLINE
| Macromolecule | Name: 1,2-DIOLEOYL-SN-GLYCERO-3-PHOSPHOCHOLINE / type: ligand / ID: 3 / Number of copies: 4 / Formula: PCW |
|---|---|
| Molecular weight | Theoretical: 787.121 Da |
| Chemical component information |  ChemComp-PCW: |
-Macromolecule #4: SODIUM ION
| Macromolecule | Name: SODIUM ION / type: ligand / ID: 4 / Number of copies: 1 |
|---|---|
| Molecular weight | Theoretical: 22.99 Da |
-Macromolecule #5: water
| Macromolecule | Name: water / type: ligand / ID: 5 / Number of copies: 130 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2.1 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 45.8 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)