[English] 日本語
 Yorodumi
Yorodumi- EMDB-41643: Langya Virus G glycoprotein (LayV G) with stabilizing mutations -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Langya Virus G glycoprotein (LayV G) with stabilizing mutations | |||||||||
 Map data Map data | sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | LayVG / LayV / G protein / attachment protein / attachment glycoprotein / Langya virus / protein design / protein stabilization / Structural Genomics / Seattle Structural Genomics Center for Infectious Disease / SSGCID / VIRAL PROTEIN | |||||||||
| Biological species |  Langya virus Langya virus | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | McCallum M / Veesler D / Seattle Structural Genomics Center for Infectious Disease (SSGCID) | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2024 Journal: Proc Natl Acad Sci U S A / Year: 2024Title: Structure and design of Langya virus glycoprotein antigens. Authors: Zhaoqian Wang / Matthew McCallum / Lianying Yan / Cecily A Gibson / William Sharkey / Young-Jun Park / Ha V Dang / Moushimi Amaya / Ashley Person / Christopher C Broder / David Veesler /  Abstract: Langya virus (LayV) is a recently discovered henipavirus (HNV), isolated from febrile patients in China. HNV entry into host cells is mediated by the attachment (G) and fusion (F) glycoproteins which ...Langya virus (LayV) is a recently discovered henipavirus (HNV), isolated from febrile patients in China. HNV entry into host cells is mediated by the attachment (G) and fusion (F) glycoproteins which are the main targets of neutralizing antibodies. We show here that the LayV F and G glycoproteins promote membrane fusion with human, mouse, and hamster target cells using a different, yet unknown, receptor than Nipah virus (NiV) and Hendra virus (HeV) and that NiV- and HeV-elicited monoclonal and polyclonal antibodies do not cross-react with LayV F and G. We determined cryoelectron microscopy structures of LayV F, in the prefusion and postfusion states, and of LayV G, revealing their conformational landscape and distinct antigenicity relative to NiV and HeV. We computationally designed stabilized LayV G constructs and demonstrate the generalizability of an HNV F prefusion-stabilization strategy. Our data will support the development of vaccines and therapeutics against LayV and closely related HNVs. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41643.map.gz emd_41643.map.gz | 306.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41643-v30.xml emd-41643-v30.xml emd-41643.xml emd-41643.xml | 18.2 KB 18.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41643.png emd_41643.png | 106.2 KB | ||
| Filedesc metadata |  emd-41643.cif.gz emd-41643.cif.gz | 6 KB | ||
| Others |  emd_41643_additional_1.map.gz emd_41643_additional_1.map.gz emd_41643_half_map_1.map.gz emd_41643_half_map_1.map.gz emd_41643_half_map_2.map.gz emd_41643_half_map_2.map.gz | 162.2 MB 300.9 MB 300.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41643 http://ftp.pdbj.org/pub/emdb/structures/EMD-41643 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41643 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41643 | HTTPS FTP |
-Related structure data
| Related structure data |  8tviMC  8tvbC  8tveC  8tvfC  8tvgC  8tvhC  8vwpC C: citing same article ( M: atomic model generated by this map |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41643.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41643.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.89 Å | ||||||||||||||||||||||||||||||||||||
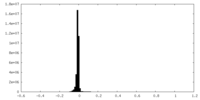
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: unsharpened map
| File | emd_41643_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
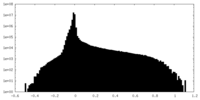
| Density Histograms |
-Half map: half map b
| File | emd_41643_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map b | ||||||||||||
| Projections & Slices |
| ||||||||||||
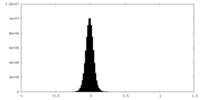
| Density Histograms |
-Half map: half map a
| File | emd_41643_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map a | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Stabilized Langya virus attachment glycoprotein LayVG
| Entire | Name: Stabilized Langya virus attachment glycoprotein LayVG |
|---|---|
| Components |
|
-Supramolecule #1: Stabilized Langya virus attachment glycoprotein LayVG
| Supramolecule | Name: Stabilized Langya virus attachment glycoprotein LayVG / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Langya virus Langya virus |
-Macromolecule #1: Langya virus attachment glycoprotein G
| Macromolecule | Name: Langya virus attachment glycoprotein G / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Langya virus Langya virus |
| Molecular weight | Theoretical: 68.051281 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MTRLTVLALL AGLLASSRAS GPHHHHHHHH GSSPQAEELK AIKEELKAIK EELKAIKEEL KAIQAMLKII QDEVNSKLEM LVSLDQLVK TEIKPKVSLI NTAVSVSIPA QISNLQTKVL QKLVYLEESI TKQCTCNPLS GIFPTTKPPP PPTDKPDDDT T DDDKVDTT ...String: MTRLTVLALL AGLLASSRAS GPHHHHHHHH GSSPQAEELK AIKEELKAIK EELKAIKEEL KAIQAMLKII QDEVNSKLEM LVSLDQLVK TEIKPKVSLI NTAVSVSIPA QISNLQTKVL QKLVYLEESI TKQCTCNPLS GIFPTTKPPP PPTDKPDDDT T DDDKVDTT IKPVEYYKPD GCNKTNDHFT MQPGVNFYTV PNLGPSSSSA DECYTNPSFS IGSSIYMFSQ EIRKTDCTTG EI LSIQIVL GRIVDKGQQG PQASPLLVWS VPNPKIINSC AVAAGDETGW VLCSVTLTAA SGEPIPHMFD GFWLYKFEPD TEV VAYRIT GFAYLLDKVY DSVFIGKGGG IQRGNDLYFQ MFGLSRNRQS IKALCEHGSC LGTGGGGYQV LCDRAVMSFG SEES LISNA YLKVNDVASG KPTIISQTFP PSDSYKGSNG RIYTIGERYG IYLAPSSWNR YLRFGLTPDI SVRSTTWLKE KDPIM KVLT TCTNTDKDMC PEICNTRGYQ DIFPLSEDSS FYTYIGITPS NEGTKSFVAV KDDAGHVASI TILPNYYSIT SATISC FMY KEEIWCIAVT EGRKQKENPQ RIYAHSYRVQ KMCFNIKPAS VVTSLPSNVT IRS |
-Macromolecule #2: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 2 / Number of copies: 4 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #3: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 3 / Number of copies: 4 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Material: GOLD |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 47.3 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.4 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.2 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 145167 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)