[English] 日本語
 Yorodumi
Yorodumi- EMDB-41585: Rod from high-resolution phycobilisome quenched by OCP (local ref... -
+ Open data
Open data
- Basic information
Basic information
| Entry | 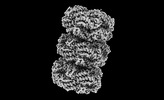 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
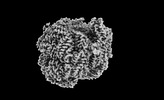
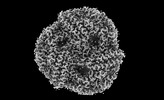
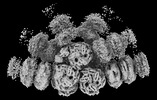
| Title | Rod from high-resolution phycobilisome quenched by OCP (local refinement) | |||||||||||||||
 Map data Map data | sharpened map | |||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | Complex / light harvesting / pigment / PHOTOSYNTHESIS | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationphycobilisome / plasma membrane-derived thylakoid membrane / photosynthesis Similarity search - Function | |||||||||||||||
| Biological species |  | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 1.9 Å | |||||||||||||||
 Authors Authors | Sauer PV / Sutter M / Cupellini L | |||||||||||||||
| Funding support |  United States, European Union, United States, European Union,  Czech Republic, 4 items Czech Republic, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2024 Journal: Sci Adv / Year: 2024Title: Structural and quantum chemical basis for OCP-mediated quenching of phycobilisomes. Authors: Paul V Sauer / Lorenzo Cupellini / Markus Sutter / Mattia Bondanza / María Agustina Domínguez Martin / Henning Kirst / David Bína / Adrian Fujiet Koh / Abhay Kotecha / Basil J Greber / ...Authors: Paul V Sauer / Lorenzo Cupellini / Markus Sutter / Mattia Bondanza / María Agustina Domínguez Martin / Henning Kirst / David Bína / Adrian Fujiet Koh / Abhay Kotecha / Basil J Greber / Eva Nogales / Tomáš Polívka / Benedetta Mennucci / Cheryl A Kerfeld /      Abstract: Cyanobacteria use large antenna complexes called phycobilisomes (PBSs) for light harvesting. However, intense light triggers non-photochemical quenching, where the orange carotenoid protein (OCP) ...Cyanobacteria use large antenna complexes called phycobilisomes (PBSs) for light harvesting. However, intense light triggers non-photochemical quenching, where the orange carotenoid protein (OCP) binds to PBS, dissipating excess energy as heat. The mechanism of efficiently transferring energy from phycocyanobilins in PBS to canthaxanthin in OCP remains insufficiently understood. Using cryo-electron microscopy, we unveiled the OCP-PBS complex structure at 1.6- to 2.1-angstrom resolution, showcasing its inherent flexibility. Using multiscale quantum chemistry, we disclosed the quenching mechanism. Identifying key protein residues, we clarified how canthaxanthin's transition dipole moment in its lowest-energy dark state becomes large enough for efficient energy transfer from phycocyanobilins. Our energy transfer model offers a detailed understanding of the atomic determinants of light harvesting regulation and antenna architecture in cyanobacteria. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41585.map.gz emd_41585.map.gz | 484.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41585-v30.xml emd-41585-v30.xml emd-41585.xml emd-41585.xml | 23.5 KB 23.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41585_fsc.xml emd_41585_fsc.xml | 16.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_41585.png emd_41585.png | 52.7 KB | ||
| Masks |  emd_41585_msk_1.map emd_41585_msk_1.map | 512 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-41585.cif.gz emd-41585.cif.gz | 6.6 KB | ||
| Others |  emd_41585_additional_1.map.gz emd_41585_additional_1.map.gz emd_41585_half_map_1.map.gz emd_41585_half_map_1.map.gz emd_41585_half_map_2.map.gz emd_41585_half_map_2.map.gz | 257.6 MB 474.9 MB 474.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41585 http://ftp.pdbj.org/pub/emdb/structures/EMD-41585 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41585 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41585 | HTTPS FTP |
-Validation report
| Summary document |  emd_41585_validation.pdf.gz emd_41585_validation.pdf.gz | 1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_41585_full_validation.pdf.gz emd_41585_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  emd_41585_validation.xml.gz emd_41585_validation.xml.gz | 26.4 KB | Display | |
| Data in CIF |  emd_41585_validation.cif.gz emd_41585_validation.cif.gz | 34.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41585 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41585 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41585 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41585 | HTTPS FTP |
-Related structure data
| Related structure data |  8troMC  8to2C  8to5C  8tpjC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41585.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41585.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sharpened map | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.727 Å | ||||||||||||||||||||
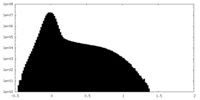
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_41585_msk_1.map emd_41585_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
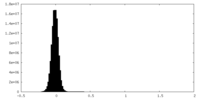
| Density Histograms |
-Additional map: refined map
| File | emd_41585_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | refined map | ||||||||||||
| Projections & Slices |
| ||||||||||||
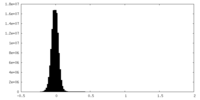
| Density Histograms |
-Half map: #2
| File | emd_41585_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_41585_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Phycobilisome bound to OCP
| Entire | Name: Phycobilisome bound to OCP |
|---|---|
| Components |
|
-Supramolecule #1: Phycobilisome bound to OCP
| Supramolecule | Name: Phycobilisome bound to OCP / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#6 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: C-phycocyanin alpha subunit
| Macromolecule | Name: C-phycocyanin alpha subunit / type: protein_or_peptide / ID: 1 / Number of copies: 18 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 17.602529 KDa |
| Sequence | String: MKTPLTEAVS TADSQGRFLS STELQIAFGR LRQANAGLQA AKALTDNAQS LVNGAAQAVY NKFPYTTQTQ GNNFAADQRG KDKCARDIG YYLRIVTYCL VAGGTGPLDE YLIAGIDEIN RTFDLSPSWY VEALKYIKAN HGLSGDARDE ANSYLDYAIN A LS UniProtKB: C-phycocyanin alpha subunit |
-Macromolecule #2: C-phycocyanin beta subunit
| Macromolecule | Name: C-phycocyanin beta subunit / type: protein_or_peptide / ID: 2 / Number of copies: 18 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 18.156451 KDa |
| Sequence | String: MFDVFTRVVS QADARGEYLS GSQLDALSAT VAEGNKRIDS VNRITGNASA IVSNAARALF AEQPQLIQPG G(MEN)AYTS RRM AACLRDMEII LRYVTYATFT GDASVLEDRC LNGLRETYVA LGVPGASVAA GVQKMKEAAL DIVNDPNGIT RGDCSAI VA EIAGYFDRAA AAVA UniProtKB: C-phycocyanin beta subunit |
-Macromolecule #3: Phycobilisome rod-core linker polypeptide CpcG
| Macromolecule | Name: Phycobilisome rod-core linker polypeptide CpcG / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 28.938758 KDa |
| Sequence | String: MALPLLNYAP KSQNVRVEGY EIGSEEKPVV FTTENILSSS DMDNLIEAAY RQIFFHAFKW DREKVLESQL RNGQITVRDF VRGLLLSNT FRNSFYEKNS NYRFVEHCVQ KILGRDVYSE REKIAWSIVV ATKGYQGLID DLLNSDEYLN NFGYDTVPYQ R RRNLPGRE ...String: MALPLLNYAP KSQNVRVEGY EIGSEEKPVV FTTENILSSS DMDNLIEAAY RQIFFHAFKW DREKVLESQL RNGQITVRDF VRGLLLSNT FRNSFYEKNS NYRFVEHCVQ KILGRDVYSE REKIAWSIVV ATKGYQGLID DLLNSDEYLN NFGYDTVPYQ R RRNLPGRE AGELPFNIKS PRYDAYHRRQ LGFPQIVWQN EVRRFIPQEK KLTAGNPMNF LGMARSINPA ANTIPKVSAQ NI NIEASVP RR UniProtKB: Phycobilisome rod-core linker polypeptide CpcG |
-Macromolecule #4: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated...
| Macromolecule | Name: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 1 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 32.558607 KDa |
| Sequence | String: MAITTAASRL GVAPYNESRP VELRPDFSLD DAKMVIRAVY RQVLGNDYIM DSERLKGAES LLTNGSISVR EFVRTVAKSE LYKKKFLYN NFQTRVIELN YKHLLGRAPF SEDEVIFHLD LYENQGFDAD IDSYIDSVEY QENFGENIVP YYRFNNQVGD R TVGFTRMF ...String: MAITTAASRL GVAPYNESRP VELRPDFSLD DAKMVIRAVY RQVLGNDYIM DSERLKGAES LLTNGSISVR EFVRTVAKSE LYKKKFLYN NFQTRVIELN YKHLLGRAPF SEDEVIFHLD LYENQGFDAD IDSYIDSVEY QENFGENIVP YYRFNNQVGD R TVGFTRMF RLYRGYANSD RSQLERSSSR LATELGQNTV SAIVGPSGSN AGWAYRPSRA GNTPAKALGG TVPFGQASKL FR VEITAIS APGYPKVRRS NKAVIVPFEQ LNQTLQQINR LGGKVASITP ASLS UniProtKB: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 1 |
-Macromolecule #5: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated...
| Macromolecule | Name: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 2 type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 30.836346 KDa |
| Sequence | String: MTSLVSAQRL GIVAVDEAIP LELRSRSTEE EVDAVILAVY RQVLGNDHLM SQERLTSAES LLRGREISVR DFVRAVALSE VYRQKFFHS NPQNRFIELN YKHLLGRAPY DQSEIAFHTD LYHQGGYEAE INSYIDSVEY TENFGDWVVP YFRGFATQRN Q KTVGFSRS ...String: MTSLVSAQRL GIVAVDEAIP LELRSRSTEE EVDAVILAVY RQVLGNDHLM SQERLTSAES LLRGREISVR DFVRAVALSE VYRQKFFHS NPQNRFIELN YKHLLGRAPY DQSEIAFHTD LYHQGGYEAE INSYIDSVEY TENFGDWVVP YFRGFATQRN Q KTVGFSRS FQVYRGYATS DRSQGNGSRS RLTRELARNT ASPVYAGSTA ESLRGTSAGS RNQMYRLQVI QGAAPGRGTR VR RGKAEYL VSYDNLSAKL QQINRQGDTV TMISLA UniProtKB: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 2 |
-Macromolecule #6: Phycobilisome 8.9 kDa linker polypeptide, phycocyanin-associated, rod
| Macromolecule | Name: Phycobilisome 8.9 kDa linker polypeptide, phycocyanin-associated, rod type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 9.333342 KDa |
| Sequence | String: MLGQSSLVGY SNTQAANRVF VYEVSGLRQT DANENSAHDI RRSGSVFIKV PYARMNDEMR RISRLGGTIV NIRPYQADSN EQN UniProtKB: Phycobilisome 8.9 kDa linker polypeptide, phycocyanin-associated, rod |
-Macromolecule #7: PHYCOCYANOBILIN
| Macromolecule | Name: PHYCOCYANOBILIN / type: ligand / ID: 7 / Number of copies: 54 / Formula: CYC |
|---|---|
| Molecular weight | Theoretical: 588.694 Da |
| Chemical component information | 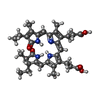 ChemComp-CYC: |
-Macromolecule #8: water
| Macromolecule | Name: water / type: ligand / ID: 8 / Number of copies: 3396 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3.8 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Vitrification | Cryogen name: ETHANE-PROPANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.2 µm / Nominal defocus min: 0.4 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





 Z
Z Y
Y X
X