[English] 日本語
 Yorodumi
Yorodumi- EMDB-40735: Structure of human ULK1 complex core (2:2:2 stoichiometry) of the... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of human ULK1 complex core (2:2:2 stoichiometry) of the ATG13(450-517) mutant | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Autophagy / Protein kinase / Complex / IMMUNE SYSTEM | |||||||||
| Function / homology |  Function and homology information Function and homology informationneuron projection regeneration / omegasome membrane / negative regulation of collateral sprouting / Atg1/ULK1 kinase complex / response to mitochondrial depolarisation / glycophagy / positive regulation of autophagosome assembly / ribophagy / protein localization to phagophore assembly site / phagophore assembly site membrane ...neuron projection regeneration / omegasome membrane / negative regulation of collateral sprouting / Atg1/ULK1 kinase complex / response to mitochondrial depolarisation / glycophagy / positive regulation of autophagosome assembly / ribophagy / protein localization to phagophore assembly site / phagophore assembly site membrane / autophagy of mitochondrion / piecemeal microautophagy of the nucleus / pexophagy / RAB GEFs exchange GTP for GDP on RABs / protein kinase regulator activity / regulation of tumor necrosis factor-mediated signaling pathway / axon extension / phagophore assembly site / TBC/RABGAPs / kinase activator activity / reticulophagy / : / response to starvation / Macroautophagy / Receptor Mediated Mitophagy / cellular response to stress / autophagosome membrane / positive regulation of cell size / regulation of macroautophagy / negative regulation of protein-containing complex assembly / autophagosome assembly / mitophagy / extrinsic apoptotic signaling pathway / protein-membrane adaptor activity / positive regulation of autophagy / autophagosome / cellular response to nutrient levels / peptidyl-serine phosphorylation / negative regulation of extrinsic apoptotic signaling pathway / regulation of autophagy / macroautophagy / liver development / Regulation of TNFR1 signaling / recycling endosome / positive regulation of JNK cascade / autophagy / small GTPase binding / neuron projection development / intracellular protein localization / protein autophosphorylation / heart development / GTPase binding / nuclear membrane / molecular adaptor activity / defense response to virus / protein phosphorylation / mitochondrial outer membrane / non-specific serine/threonine protein kinase / lysosome / negative regulation of cell population proliferation / axon / innate immune response / protein serine kinase activity / protein serine/threonine kinase activity / endoplasmic reticulum membrane / protein kinase binding / protein-containing complex binding / signal transduction / mitochondrion / ATP binding / identical protein binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.46 Å | |||||||||
 Authors Authors | Chen M / Hurley JH | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: bioRxiv Journal: bioRxivTitle: Structure and activation of the human autophagy-initiating ULK1C:PI3KC3-C1 supercomplex Authors: Chen M / Ren X / Cook A / Hurley JH | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_40735.map.gz emd_40735.map.gz | 167.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-40735-v30.xml emd-40735-v30.xml emd-40735.xml emd-40735.xml | 20 KB 20 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_40735.png emd_40735.png | 54.9 KB | ||
| Filedesc metadata |  emd-40735.cif.gz emd-40735.cif.gz | 6.6 KB | ||
| Others |  emd_40735_half_map_1.map.gz emd_40735_half_map_1.map.gz emd_40735_half_map_2.map.gz emd_40735_half_map_2.map.gz | 165 MB 165 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-40735 http://ftp.pdbj.org/pub/emdb/structures/EMD-40735 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40735 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40735 | HTTPS FTP |
-Related structure data
| Related structure data |  8srmMC  8soiC  8sorC  8sqzC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_40735.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_40735.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.115 Å | ||||||||||||||||||||||||||||||||||||
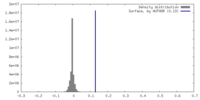
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: half map B
| File | emd_40735_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map B | ||||||||||||
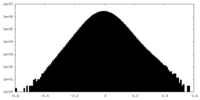
| Projections & Slices |
| ||||||||||||
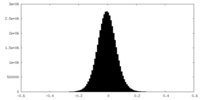
| Density Histograms |
-Half map: half map A
| File | emd_40735_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
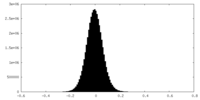
| Density Histograms |
- Sample components
Sample components
-Entire : Human autophagy initiation ULK1 complex core
| Entire | Name: Human autophagy initiation ULK1 complex core |
|---|---|
| Components |
|
-Supramolecule #1: Human autophagy initiation ULK1 complex core
| Supramolecule | Name: Human autophagy initiation ULK1 complex core / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 210 KDa |
-Macromolecule #1: RB1-inducible coiled-coil protein 1
| Macromolecule | Name: RB1-inducible coiled-coil protein 1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 73.325633 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MKLYVFLVNT GTTLTFDTEL TVQTVADLKH AIQSKYKIAI QHQVLVVNGG ECMAADRRVC TYSAGTDTNP IFLFNKEMIL CDRPPAIPK TTFSTENDME IKVEESLMMP AVFHTVASRT QLALEMYEVA KKLCSFCEGL VHDEHLQHQG WAAIMANLED C SNSYQKLL ...String: MKLYVFLVNT GTTLTFDTEL TVQTVADLKH AIQSKYKIAI QHQVLVVNGG ECMAADRRVC TYSAGTDTNP IFLFNKEMIL CDRPPAIPK TTFSTENDME IKVEESLMMP AVFHTVASRT QLALEMYEVA KKLCSFCEGL VHDEHLQHQG WAAIMANLED C SNSYQKLL FKFESIYSNY LQSIEDIKLK LTHLGTAVSV MAKIPLLECL TRHSYRECLG RLDSLPEHED SEKAEMKRST EL VLSPDMP RTTNESLLTS FPKSVEHVSP DTADAESGKE IRESCQSTVH QQDETTIDTK DGDLPFFNVS LLDWINVQDR PND VESLVR KCFDSMSRLD PRIIRPFIAE CRQTIAKLDN QNMKAIKGLE DRLYALDQMI ASCGRLVNEQ KELAQGFLAN QKRA ENLKD ASVLPDLCLS HANQLMIMLQ NHRKLLDIKQ KCTTAKQELA NNLHVRLKWC CFVMLHADQD GEKLQALLRL VIELL ERVK IVEALSTVPQ MYCLAVVEVV RRKMFIKHYR EWAGALVKDG KRLYEAEKSK RESFGKLFRK SFLRNRLFRG LDSWPP SFC TQKPRKFDCE LPDISLKDLQ FLQSFCPSEV QPFLRVPLLC DFEPLHQHVL ALHNLVKAAQ SLDEMSQTIT DLLSEQK UniProtKB: RB1-inducible coiled-coil protein 1 |
-Macromolecule #2: Serine/threonine-protein kinase ULK1
| Macromolecule | Name: Serine/threonine-protein kinase ULK1 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO / EC number: non-specific serine/threonine protein kinase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 24.190145 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MEQEHTEILR GLRFTLLFVQ HVLEIAALKG SASEAAGGPE YQLQESVVAD QISLLSREWG FAEQLVLYLK VAELLSSGLQ SAIDQIRAG KLCLSSTVKQ VVRRLNELYK ASVVSCQGLS LRLQRFFLDK QRLLDRIHSI TAERLIFSHA VQMVQSAALD E MFQHREGC ...String: MEQEHTEILR GLRFTLLFVQ HVLEIAALKG SASEAAGGPE YQLQESVVAD QISLLSREWG FAEQLVLYLK VAELLSSGLQ SAIDQIRAG KLCLSSTVKQ VVRRLNELYK ASVVSCQGLS LRLQRFFLDK QRLLDRIHSI TAERLIFSHA VQMVQSAALD E MFQHREGC VPRYHKALLL LEGLQHMLSD QADIENVTKC KLCIERRLSA LLTGICA UniProtKB: Serine/threonine-protein kinase ULK1 |
-Macromolecule #3: Autophagy-related protein 13
| Macromolecule | Name: Autophagy-related protein 13 / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 8.150997 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: KPAFSKDDIL PMDLGTFYRE FQNPPQLSSL SIDIGAQSMA EDLDSLPEKL AVHEKNVREF DAFVETLQGS DEA UniProtKB: Autophagy-related protein 13 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.35 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| |||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR / Details: 25 mA | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number grids imaged: 1 / Number real images: 2286 / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 36000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | Chain - Source name: AlphaFold / Chain - Initial model type: in silico model |
|---|---|
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
| Output model |  PDB-8srm: |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)