+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM structure of ET-1 bound ETBR-DNGI complex | ||||||||||||
 マップデータ マップデータ | |||||||||||||
 試料 試料 |
| ||||||||||||
 キーワード キーワード | ENDOTHELIN / RECEPTOR / Gi / COMPLEX / MEMBRANE PROTEIN | ||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報enteric smooth muscle cell differentiation / response to endothelin / positive regulation of prostaglandin-endoperoxide synthase activity / aldosterone metabolic process / negative regulation of neuron maturation / endothelin A receptor binding / protein kinase C deactivation / chordate pharynx development / phospholipase D-activating G protein-coupled receptor signaling pathway / rhythmic excitation ...enteric smooth muscle cell differentiation / response to endothelin / positive regulation of prostaglandin-endoperoxide synthase activity / aldosterone metabolic process / negative regulation of neuron maturation / endothelin A receptor binding / protein kinase C deactivation / chordate pharynx development / phospholipase D-activating G protein-coupled receptor signaling pathway / rhythmic excitation / endothelin receptor activity / peptide hormone secretion / endothelin B receptor binding / cellular response to human chorionic gonadotropin stimulus / meiotic cell cycle process involved in oocyte maturation / positive regulation of artery morphogenesis / semaphorin-plexin signaling pathway involved in axon guidance / body fluid secretion / neural crest cell fate commitment / regulation of fever generation / vein smooth muscle contraction / glomerular endothelium development / response to prostaglandin F / sympathetic neuron axon guidance / positive regulation of sarcomere organization / noradrenergic neuron differentiation / positive regulation of renal sodium excretion / histamine secretion / leukocyte activation / maternal process involved in parturition / positive regulation of chemokine-mediated signaling pathway / positive regulation of penile erection / rough endoplasmic reticulum lumen / neuroblast migration / heparin metabolic process / posterior midgut development / developmental pigmentation / pharyngeal arch artery morphogenesis / regulation of D-glucose transmembrane transport / positive regulation of odontogenesis / epithelial fluid transport / endothelin receptor signaling pathway involved in heart process / negative regulation of hormone secretion / cardiac neural crest cell migration involved in outflow tract morphogenesis / Weibel-Palade body / response to leptin / endothelin receptor signaling pathway / response to ozone / podocyte differentiation / positive regulation of cell growth involved in cardiac muscle cell development / renal sodium ion absorption / artery smooth muscle contraction / response to sodium phosphate / glomerular filtration / renal sodium excretion / protein transmembrane transport / enteric nervous system development / axonogenesis involved in innervation / positive regulation of cation channel activity / renin secretion into blood stream / cellular response to follicle-stimulating hormone stimulus / cellular response to luteinizing hormone stimulus / melanocyte differentiation / : / regulation of pH / positive regulation of prostaglandin secretion / renal albumin absorption / regulation of epithelial cell proliferation / respiratory gaseous exchange by respiratory system / basal part of cell / cellular response to mineralocorticoid stimulus / peripheral nervous system development / positive regulation of smooth muscle contraction / response to salt / positive regulation of urine volume / positive regulation of hormone secretion / negative regulation of adenylate cyclase activity / regulation of systemic arterial blood pressure by endothelin / vasoconstriction / negative regulation of blood coagulation / type 1 angiotensin receptor binding / embryonic heart tube development / : / dorsal/ventral pattern formation / axon extension / establishment of endothelial barrier / superoxide anion generation / positive regulation of neutrophil chemotaxis / positive regulation of signaling receptor activity / cartilage development / middle ear morphogenesis / cellular response to glucocorticoid stimulus / negative regulation of protein metabolic process / neural crest cell migration / cGMP-mediated signaling / prostaglandin biosynthetic process / cellular response to fatty acid / nitric oxide transport / positive regulation of heart rate / branching involved in blood vessel morphogenesis 類似検索 - 分子機能 | ||||||||||||
| 生物種 |  Homo sapiens (ヒト) / Homo sapiens (ヒト) /  | ||||||||||||
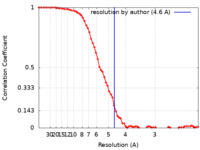
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 4.6 Å | ||||||||||||
 データ登録者 データ登録者 | Tani K / Maki-Yonekura S / Kanno R / Negami T / Hamaguchi T / Hall M / Mizoguchi A / Humbel BM / Terada T / Yonekura K / Doi T | ||||||||||||
| 資金援助 |  日本, 3件 日本, 3件
| ||||||||||||
 引用 引用 |  ジャーナル: Commun Biol / 年: 2024 ジャーナル: Commun Biol / 年: 2024タイトル: Structure of endothelin ET receptor-G complex in a conformation stabilized by unique NPxxL motif. 著者: Kazutoshi Tani / Saori Maki-Yonekura / Ryo Kanno / Tatsuki Negami / Tasuku Hamaguchi / Malgorzata Hall / Akira Mizoguchi / Bruno M Humbel / Tohru Terada / Koji Yonekura / Tomoko Doi /  要旨: Endothelin type B receptor (ETR) plays a crucial role in regulating blood pressure and humoral homeostasis, making it an important therapeutic target for related diseases. ETR activation by the ...Endothelin type B receptor (ETR) plays a crucial role in regulating blood pressure and humoral homeostasis, making it an important therapeutic target for related diseases. ETR activation by the endogenous peptide hormones endothelin (ET)-1-3 stimulates several signaling pathways, including G, G, G, G, and β-arrestin. Although the conserved NPxxY motif in transmembrane helix 7 (TM7) is important during GPCR activation, ETR possesses the lesser known NPxxL motif. In this study, we present the cryo-EM structure of the ETR-G complex, complemented by MD simulations and functional studies. These investigations reveal an unusual movement of TM7 to the intracellular side during ETR activation and the essential roles of the diverse NPxxL motif in stabilizing the active conformation of ETR and organizing the assembly of the binding pocket for the α5 helix of G protein. These findings enhance our understanding of the interactions between GPCRs and G proteins, thereby advancing the development of therapeutic strategies. | ||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_38741.map.gz emd_38741.map.gz | 26 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-38741-v30.xml emd-38741-v30.xml emd-38741.xml emd-38741.xml | 20.7 KB 20.7 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_38741_fsc.xml emd_38741_fsc.xml | 6.7 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_38741.png emd_38741.png | 43.4 KB | ||
| Filedesc metadata |  emd-38741.cif.gz emd-38741.cif.gz | 6.5 KB | ||
| その他 |  emd_38741_half_map_1.map.gz emd_38741_half_map_1.map.gz emd_38741_half_map_2.map.gz emd_38741_half_map_2.map.gz | 21.3 MB 21.3 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-38741 http://ftp.pdbj.org/pub/emdb/structures/EMD-38741 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38741 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38741 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_38741_validation.pdf.gz emd_38741_validation.pdf.gz | 848.6 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_38741_full_validation.pdf.gz emd_38741_full_validation.pdf.gz | 848.2 KB | 表示 | |
| XML形式データ |  emd_38741_validation.xml.gz emd_38741_validation.xml.gz | 12.5 KB | 表示 | |
| CIF形式データ |  emd_38741_validation.cif.gz emd_38741_validation.cif.gz | 17.5 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38741 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38741 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38741 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38741 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_38741.map.gz / 形式: CCP4 / 大きさ: 27.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_38741.map.gz / 形式: CCP4 / 大きさ: 27.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.094 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: even
| ファイル | emd_38741_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | even | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: odd
| ファイル | emd_38741_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | odd | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : ET-1 BOUND ETBR-GI COMPLEX
| 全体 | 名称: ET-1 BOUND ETBR-GI COMPLEX |
|---|---|
| 要素 |
|
-超分子 #1: ET-1 BOUND ETBR-GI COMPLEX
| 超分子 | 名称: ET-1 BOUND ETBR-GI COMPLEX / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
-分子 #1: Endothelin receptor type B
| 分子 | 名称: Endothelin receptor type B / タイプ: protein_or_peptide / ID: 1 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 38.808262 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: PGGGLAPAEV PKGDRTAGSP PRTISPPPCQ GPIEIKETFK YINTVVSCLV FVLGIIGNST LLYIIYKNKC MRNGPNILIA SLALGDLLH IVIDIPINVY KLLAEDWPFG AEMCKLVPFI QKASVGITVL SLCALSIDRY RAVASWSRIK GIGVPKWTAV E IVLIWVVS ...文字列: PGGGLAPAEV PKGDRTAGSP PRTISPPPCQ GPIEIKETFK YINTVVSCLV FVLGIIGNST LLYIIYKNKC MRNGPNILIA SLALGDLLH IVIDIPINVY KLLAEDWPFG AEMCKLVPFI QKASVGITVL SLCALSIDRY RAVASWSRIK GIGVPKWTAV E IVLIWVVS VVLAVPEAIG FDIITMDYKG SYLRICLLHP VQKTAFMQFY KTAKDWWLFS FYFCLPLAIT AFFYTLMTCE ML RKKSGMQ IALNDHLKQR REVAKTVFCL VLVFALCWLP LHLSRILKLT LYNQNDPNRC ELLSFLLVLD YIGINMASLN SCI NPIALY LVSKRFKNAF KSALCCWAQS UniProtKB: Endothelin receptor type B |
-分子 #2: Endothelin-1
| 分子 | 名称: Endothelin-1 / タイプ: protein_or_peptide / ID: 2 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 2.497951 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: CSCSSLMDKE CVYFCHLDII W UniProtKB: Endothelin-1 |
-分子 #3: Guanine nucleotide-binding protein G(i) subunit alpha-1
| 分子 | 名称: Guanine nucleotide-binding protein G(i) subunit alpha-1 タイプ: protein_or_peptide / ID: 3 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 40.415031 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI ...文字列: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI PTQQDVLRTR VKTTGIVETH FTFKDLHFKM FDVGGQRSER KKWIHCFEGV TAIIFCVALS DYDLVLAEDE EM NRMHESM KLFDSICNNK WFTDTSIILF LNKKDLFEEK IKKSPLTICY PEYAGSNTYE EAAAYIQCQF EDLNKRKDTK EIY THFTCA TDTKNVQFVF DAVTDVIIKN NLKDCGLF UniProtKB: Guanine nucleotide-binding protein G(i) subunit alpha-1 |
-分子 #4: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| 分子 | 名称: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 タイプ: protein_or_peptide / ID: 4 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 37.671102 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: PGSSGSELDQ LRQEAEQLKN QIRDARKACA DATLSQITNN IDPVGRIQMR TRRTLRGHLA KIYAMHWGTD SRLLVSASQD GKLIIWDSY TTNKVHAIPL RSSWVMTCAY APSGNYVACG GLDNICSIYN LKTREGNVRV SRELAGHTGY LSCCRFLDDN Q IVTSSGDT ...文字列: PGSSGSELDQ LRQEAEQLKN QIRDARKACA DATLSQITNN IDPVGRIQMR TRRTLRGHLA KIYAMHWGTD SRLLVSASQD GKLIIWDSY TTNKVHAIPL RSSWVMTCAY APSGNYVACG GLDNICSIYN LKTREGNVRV SRELAGHTGY LSCCRFLDDN Q IVTSSGDT TCALWDIETG QQTTTFTGHT GDVMSLSLAP DTRLFVSGAC DASAKLWDVR EGMCRQTFTG HESDINAICF FP NGNAFAT GSDDATCRLF DLRADQELMT YSHDNIICGI TSVSFSKSGR LLLAGYDDFN CNVWDALKAD RAGVLAGHDN RVS CLGVTD DGMAVATGSW DSFLKIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-分子 #5: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| 分子 | 名称: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 タイプ: protein_or_peptide / ID: 5 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 7.861143 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-分子 #6: SCFV16
| 分子 | 名称: SCFV16 / タイプ: protein_or_peptide / ID: 6 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 29.39893 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MVSAIVLYVL LAAAAHSAFA DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFT ISRDDPKNTL FLQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV M TQATSSVP ...文字列: MVSAIVLYVL LAAAAHSAFA DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFT ISRDDPKNTL FLQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV M TQATSSVP VTPGESVSIS CRSSKSLLHS NGNTYLYWFL QRPGQSPQLL IYRMSNLASG VPDRFSGSGS GTAFTLTISR LE AEDVGVY YCMQHLEYPL TFGAGTKLEL KGSLEVLFQG |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 6 mg/mL |
|---|---|
| 緩衝液 | pH: 7.5 |
| 凍結 | 凍結剤: ETHANE |
| 詳細 | This sample was monodisperse. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TECNAI ARCTICA |
|---|---|
| 撮影 | フィルム・検出器のモデル: FEI FALCON III (4k x 4k) 平均電子線量: 40.0 e/Å2 |
| 電子線 | 加速電圧: 200 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.9 µm 最小 デフォーカス(公称値): 0.7000000000000001 µm |
| 実験機器 |  モデル: Talos Arctica / 画像提供: FEI Company |
+ 画像解析
画像解析
-原子モデル構築 1
| 精密化 | プロトコル: RIGID BODY FIT |
|---|---|
| 得られたモデル |  PDB-8xwq: |
 ムービー
ムービー コントローラー
コントローラー






































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)