+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Ralmitaront(RO-6889450)-bound hTAAR1-Gs protein complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Complex / Agonist / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationAmine ligand-binding receptors / trace-amine receptor activity / G protein-coupled receptor activity / electron transport chain / Olfactory Signaling Pathway / Activation of the phototransduction cascade / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor ...Amine ligand-binding receptors / trace-amine receptor activity / G protein-coupled receptor activity / electron transport chain / Olfactory Signaling Pathway / Activation of the phototransduction cascade / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Glucagon signaling in metabolic regulation / G protein-coupled acetylcholine receptor signaling pathway / G-protein activation / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G beta:gamma signalling through CDC42 / Vasopressin regulates renal water homeostasis via Aquaporins / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Glucagon-type ligand receptors / Sensory perception of sweet, bitter, and umami (glutamate) taste / photoreceptor disc membrane / G alpha (z) signalling events / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / cellular response to catecholamine stimulus / sensory perception of taste / ADP signalling through P2Y purinoceptor 1 / adenylate cyclase-activating dopamine receptor signaling pathway / G beta:gamma signalling through PI3Kgamma / GPER1 signaling / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / cellular response to prostaglandin E stimulus / Inactivation, recovery and regulation of the phototransduction cascade / G-protein beta-subunit binding / heterotrimeric G-protein complex / G alpha (12/13) signalling events / extracellular vesicle / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / GTPase binding / retina development in camera-type eye / Ca2+ pathway / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events / fibroblast proliferation / G alpha (s) signalling events / G alpha (q) signalling events / Ras protein signal transduction / cell population proliferation / Extra-nuclear estrogen signaling / periplasmic space / electron transfer activity / iron ion binding / G protein-coupled receptor signaling pathway / lysosomal membrane / GTPase activity / heme binding / synapse / protein-containing complex binding / signal transduction / extracellular exosome / membrane / plasma membrane / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.23 Å | |||||||||
 Authors Authors | Xu Z / Guo LL / Zhao C / Shen SY / Sun JP / Shao ZH | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Ligand recognition and G-protein coupling of trace amine receptor TAAR1. Authors: Zheng Xu / Lulu Guo / Jingjing Yu / Siyuan Shen / Chao Wu / Weifeng Zhang / Chang Zhao / Yue Deng / Xiaowen Tian / Yuying Feng / Hanlin Hou / Lantian Su / Hongshuang Wang / Shuo Guo / Heli ...Authors: Zheng Xu / Lulu Guo / Jingjing Yu / Siyuan Shen / Chao Wu / Weifeng Zhang / Chang Zhao / Yue Deng / Xiaowen Tian / Yuying Feng / Hanlin Hou / Lantian Su / Hongshuang Wang / Shuo Guo / Heli Wang / Kexin Wang / Peipei Chen / Jie Zhao / Xiaoyu Zhang / Xihao Yong / Lin Cheng / Lunxu Liu / Shengyong Yang / Fan Yang / Xiaohui Wang / Xiao Yu / Yunfei Xu / Jin-Peng Sun / Wei Yan / Zhenhua Shao /  Abstract: Trace-amine-associated receptors (TAARs), a group of biogenic amine receptors, have essential roles in neurological and metabolic homeostasis. They recognize diverse endogenous trace amines and ...Trace-amine-associated receptors (TAARs), a group of biogenic amine receptors, have essential roles in neurological and metabolic homeostasis. They recognize diverse endogenous trace amines and subsequently activate a range of G-protein-subtype signalling pathways. Notably, TAAR1 has emerged as a promising therapeutic target for treating psychiatric disorders. However, the molecular mechanisms underlying its ability to recognize different ligands remain largely unclear. Here we present nine cryo-electron microscopy structures, with eight showing human and mouse TAAR1 in a complex with an array of ligands, including the endogenous 3-iodothyronamine, two antipsychotic agents, the psychoactive drug amphetamine and two identified catecholamine agonists, and one showing 5-HTR in a complex with an antipsychotic agent. These structures reveal a rigid consensus binding motif in TAAR1 that binds to endogenous trace amine stimuli and two extended binding pockets that accommodate diverse chemotypes. Combined with mutational analysis, functional assays and molecular dynamic simulations, we elucidate the structural basis of drug polypharmacology and identify the species-specific differences between human and mouse TAAR1. Our study provides insights into the mechanism of ligand recognition and G-protein selectivity by TAAR1, which may help in the discovery of ligands or therapeutic strategies for neurological and metabolic disorders. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36403.map.gz emd_36403.map.gz | 59.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36403-v30.xml emd-36403-v30.xml emd-36403.xml emd-36403.xml | 20.3 KB 20.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_36403.png emd_36403.png | 47.3 KB | ||
| Filedesc metadata |  emd-36403.cif.gz emd-36403.cif.gz | 6.5 KB | ||
| Others |  emd_36403_half_map_1.map.gz emd_36403_half_map_1.map.gz emd_36403_half_map_2.map.gz emd_36403_half_map_2.map.gz | 59.2 MB 59.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36403 http://ftp.pdbj.org/pub/emdb/structures/EMD-36403 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36403 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36403 | HTTPS FTP |
-Validation report
| Summary document |  emd_36403_validation.pdf.gz emd_36403_validation.pdf.gz | 856.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_36403_full_validation.pdf.gz emd_36403_full_validation.pdf.gz | 856.1 KB | Display | |
| Data in XML |  emd_36403_validation.xml.gz emd_36403_validation.xml.gz | 12.2 KB | Display | |
| Data in CIF |  emd_36403_validation.cif.gz emd_36403_validation.cif.gz | 14.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36403 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36403 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36403 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36403 | HTTPS FTP |
-Related structure data
| Related structure data |  8jlpMC  8jljC  8jlkC  8jlnC  8jloC  8jlqC  8jlrC  8jsoC  8jspC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_36403.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36403.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_36403_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
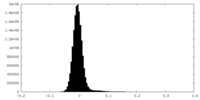
| Density Histograms |
-Half map: #2
| File | emd_36403_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
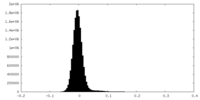
| Density Histograms |
- Sample components
Sample components
-Entire : hTAAR1-bound RAL in complex with Gs heterotrimer
| Entire | Name: hTAAR1-bound RAL in complex with Gs heterotrimer |
|---|---|
| Components |
|
-Supramolecule #1: hTAAR1-bound RAL in complex with Gs heterotrimer
| Supramolecule | Name: hTAAR1-bound RAL in complex with Gs heterotrimer / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|
-Supramolecule #2: Gs heterotrimer
| Supramolecule | Name: Gs heterotrimer / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1-#2, #4 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: RAL-bound hTAAR1
| Supramolecule | Name: RAL-bound hTAAR1 / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Gs
| Macromolecule | Name: Gs / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 43.117918 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGCTLRNEEK AQREANKKIE KQLQKDKQVY RATHRLLLLG AGESGKNTIV KQMRIYHVNG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI ...String: MGCTLRNEEK AQREANKKIE KQLQKDKQVY RATHRLLLLG AGESGKNTIV KQMRIYHVNG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI PTQQDVLRTR VKTSGIFETK FQVDKVNFHM FDVGAQRDER RKWIQCFNDV TAIIFVVASS SYNMVIREDN QT NRLQAAL KLFDSIWNNK WLRDTSVILF LNKQDLLAEK VLAGKSKIED YFPEFARYTT PEDATPEPGE DPRVTRAKYF IRD EFLRIS TASGDGRHYC YPHFTCSVDT ENIRRVFNDC RDIIQRMHLR QYELL |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.141793 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MHHHHLEVLF QGPGSSGSEL DQLRQEAEQL KNQIRDARKA CADATLSQIT NNIDPVGRIQ MRTRRTLRGH LAKIYAMHWG TDSRLLVSA SQDGKLIIWD SYTTNKVHAI PLRSSWVMTC AYAPSGNYVA CGGLDNICSI YNLKTREGNV RVSRELAGHT G YLSCCRFL ...String: MHHHHLEVLF QGPGSSGSEL DQLRQEAEQL KNQIRDARKA CADATLSQIT NNIDPVGRIQ MRTRRTLRGH LAKIYAMHWG TDSRLLVSA SQDGKLIIWD SYTTNKVHAI PLRSSWVMTC AYAPSGNYVA CGGLDNICSI YNLKTREGNV RVSRELAGHT G YLSCCRFL DDNQIVTSSG DTTCALWDIE TGQQTTTFTG HTGDVMSLSL APDTRLFVSG ACDASAKLWD VREGMCRQTF TG HESDINA ICFFPNGNAF ATGSDDATCR LFDLRADQEL MTYSHDNIIC GITSVSFSKS GRLLLAGYDD FNCNVWDALK ADR AGVLAG HDNRVSCLGV TDDGMAVATG SWDSFLKIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Soluble cytochrome b562,Trace amine-associated receptor 1
| Macromolecule | Name: Soluble cytochrome b562,Trace amine-associated receptor 1 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 54.128133 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DYKDDDDKHH HHHHHHHHEN LYFQGADLED NWETLNDNLK VIEKADNAAQ VKDALTKMRA AALDAQKATP PKLEDKSPDS PEMKDFRHG FDILVGQIDD ALKLANEGKV KEAQAAAEQL KTTRNAYIQK YLMMPFCHNI INISCVKNNW SNDVRASLYS L MVLIILTT ...String: DYKDDDDKHH HHHHHHHHEN LYFQGADLED NWETLNDNLK VIEKADNAAQ VKDALTKMRA AALDAQKATP PKLEDKSPDS PEMKDFRHG FDILVGQIDD ALKLANEGKV KEAQAAAEQL KTTRNAYIQK YLMMPFCHNI INISCVKNNW SNDVRASLYS L MVLIILTT LVGNLIVIVS ISHFKQLHTP TNWLIHSMAT VDFLLGCLVM PYSMVRSAEH CWYFGEVFCK IHTSTDIMLS SA SIFHLSF ISIDRYYAVC DPLRYKAKMN ILVICVMIFI SWSVPAVFAF GMIFLELNFK GAEEIYYKHV HCRGGCSVFF SKI SGVLTF MTSFYIPGSI MLCVYYRIYL IAKEQARLIS DANQKLQIGL EMKNGISQSK ERKAVKTLGI VMGVFLICWC PFFI CTVMD PFLHYIIPPT LNDVLIWFGY LNSTFNPMVY AFFYPWFRKA LKMMLFGKIF QKDSSRCKLF LELSS UniProtKB: Soluble cytochrome b562, Trace amine-associated receptor 1 |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #5: 5-ethyl-4-methyl-~{N}-[4-[(2~{S})-morpholin-2-yl]phenyl]-1~{H}-py...
| Macromolecule | Name: 5-ethyl-4-methyl-~{N}-[4-[(2~{S})-morpholin-2-yl]phenyl]-1~{H}-pyrazole-3-carboxamide type: ligand / ID: 5 / Number of copies: 1 / Formula: UJU |
|---|---|
| Molecular weight | Theoretical: 314.382 Da |
| Chemical component information | 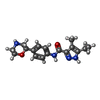 ChemComp-UJU: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 57.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.23 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 3205589 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller



































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)