[English] 日本語
 Yorodumi
Yorodumi- EMDB-36368: Cryo-EM structure of CCHFV envelope protein Gc trimer in complex ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of CCHFV envelope protein Gc trimer in complex with Gc13 Fab | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CCHFV / envelope protein / postfusion / Bunyavirus / VIRAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationhost cell Golgi membrane / clathrin-dependent endocytosis of virus by host cell / host cell endoplasmic reticulum membrane / virus-mediated perturbation of host defense response / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / virion membrane / membrane Similarity search - Function | |||||||||
| Biological species |  Crimean-Congo hemorrhagic fever virus strain IbAr10200 / Crimean-Congo hemorrhagic fever virus strain IbAr10200 /  Crimean-Congo hemorrhagic fever virus (strain Nigeria/IbAr10200/1970) Crimean-Congo hemorrhagic fever virus (strain Nigeria/IbAr10200/1970) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||
 Authors Authors | Chong T / Cao S | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: PLoS Pathog / Year: 2024 Journal: PLoS Pathog / Year: 2024Title: Neutralizing monoclonal antibodies against the Gc fusion loop region of Crimean-Congo hemorrhagic fever virus. Authors: Liushuai Li / Tingting Chong / Lu Peng / Yajie Liu / Guibo Rao / Yan Fu / Yanni Shu / Jiamei Shen / Qinghong Xiao / Jia Liu / Jiang Li / Fei Deng / Bing Yan / Zhihong Hu / Sheng Cao / Manli Wang /  Abstract: Crimean-Congo hemorrhagic fever virus (CCHFV) is a highly pathogenic tick-borne virus, prevalent in more than 30 countries worldwide. Human infection by this virus leads to severe illness, with an ...Crimean-Congo hemorrhagic fever virus (CCHFV) is a highly pathogenic tick-borne virus, prevalent in more than 30 countries worldwide. Human infection by this virus leads to severe illness, with an average case fatality of 40%. There is currently no approved vaccine or drug to treat the disease. Neutralizing antibodies are a promising approach to treat virus infectious diseases. This study generated 37 mouse-derived specific monoclonal antibodies against CCHFV Gc subunit. Neutralization assays using pseudotyped virus and authentic CCHFV identified Gc8, Gc13, and Gc35 as neutralizing antibodies. Among them, Gc13 had the highest neutralizing activity and binding affinity with CCHFV Gc. Consistently, Gc13, but not Gc8 or Gc35, showed in vivo protective efficacy (62.5% survival rate) against CCHFV infection in a lethal mouse infection model. Further characterization studies suggested that Gc8 and Gc13 may recognize a similar, linear epitope in domain II of CCHFV Gc, while Gc35 may recognize a different epitope in Gc. Cryo-electron microscopy of Gc-Fab complexes indicated that both Gc8 and Gc13 bind to the conserved fusion loop region and Gc13 had stronger interactions with sGc-trimers. This was supported by the ability of Gc13 to block CCHFV GP-mediated membrane fusion. Overall, this study provides new therapeutic strategies to treat CCHF and new insights into the interaction between antibodies with CCHFV Gc proteins. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36368.map.gz emd_36368.map.gz | 203.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36368-v30.xml emd-36368-v30.xml emd-36368.xml emd-36368.xml | 14.1 KB 14.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_36368.png emd_36368.png | 62.6 KB | ||
| Filedesc metadata |  emd-36368.cif.gz emd-36368.cif.gz | 5.6 KB | ||
| Others |  emd_36368_half_map_1.map.gz emd_36368_half_map_1.map.gz emd_36368_half_map_2.map.gz emd_36368_half_map_2.map.gz | 200.6 MB 200.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36368 http://ftp.pdbj.org/pub/emdb/structures/EMD-36368 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36368 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36368 | HTTPS FTP |
-Validation report
| Summary document |  emd_36368_validation.pdf.gz emd_36368_validation.pdf.gz | 777.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_36368_full_validation.pdf.gz emd_36368_full_validation.pdf.gz | 776.9 KB | Display | |
| Data in XML |  emd_36368_validation.xml.gz emd_36368_validation.xml.gz | 15.6 KB | Display | |
| Data in CIF |  emd_36368_validation.cif.gz emd_36368_validation.cif.gz | 18.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36368 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36368 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36368 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36368 | HTTPS FTP |
-Related structure data
| Related structure data |  8jkdMC  8jlwC  8jlxC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_36368.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36368.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.95 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_36368_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
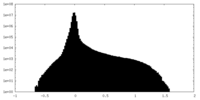
| Projections & Slices |
| ||||||||||||
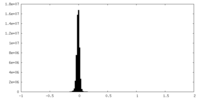
| Density Histograms |
-Half map: #1
| File | emd_36368_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
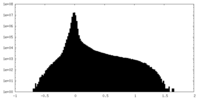
| Projections & Slices |
| ||||||||||||
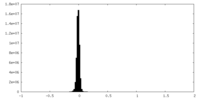
| Density Histograms |
- Sample components
Sample components
-Entire : CCHFV envelope protein Gc
| Entire | Name: CCHFV envelope protein Gc |
|---|---|
| Components |
|
-Supramolecule #1: CCHFV envelope protein Gc
| Supramolecule | Name: CCHFV envelope protein Gc / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Crimean-Congo hemorrhagic fever virus strain IbAr10200 Crimean-Congo hemorrhagic fever virus strain IbAr10200 |
-Macromolecule #1: Glycoprotein C
| Macromolecule | Name: Glycoprotein C / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Crimean-Congo hemorrhagic fever virus (strain Nigeria/IbAr10200/1970) Crimean-Congo hemorrhagic fever virus (strain Nigeria/IbAr10200/1970)Strain: Nigeria/IbAr10200/1970 |
| Molecular weight | Theoretical: 65.809711 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKNLLNSTSL ETSLSIEAPW GAINVQSTYK PTVSTANIAL SWSSVEHRGN KILVSGRSES IMKLEERTGI SWDLGVEDAS ESKLLTVSV MDLSQMYSPV FEYLSGDRQV GEWPKATCTG DCPERCGCTS STCLHKEWPH SRNWRCNPTW CWGVGTGCTC C GLDVKDLF ...String: MKNLLNSTSL ETSLSIEAPW GAINVQSTYK PTVSTANIAL SWSSVEHRGN KILVSGRSES IMKLEERTGI SWDLGVEDAS ESKLLTVSV MDLSQMYSPV FEYLSGDRQV GEWPKATCTG DCPERCGCTS STCLHKEWPH SRNWRCNPTW CWGVGTGCTC C GLDVKDLF TDYMFVKWKV EYIKTEAIVC VELTSQERQC SLIEAGTRFN LGPVTITLSE PRNIQQKLPP EIITLHPRIE EG FFDLMHV QKVLSASTVC KLQSCTHGVP GDLQVYHIGN LLKGDKVNGH LIHKIEPHFN TSWMSWDGCD LDYYCNMGDW PSC TYTGVT QHNHASFVNL LNIETDYTKN FHFHSKRVTA HGDTPQLDLK ARPTYGAGEI TVLVEVADME LHTKKIEISG LKFA SLACT GCYACSSGIS CKVRIHVDEP DELTVHVKSD DPDVVAASSS LMARKLEFGT DSTFKAFSAM PKTSLCFYIV EREHC KSCS EEDTKKCVNT KLEQPQSILI EHKGTIIGKQ NSTCTAKSRG SGGMKQIEDK IEEILSKIYH IENEIARIKK LIGEGS GGS RGPFEGKPIP NPLLGLDSTR TGHHHHHH UniProtKB: Envelopment polyprotein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | JEOL CRYO ARM 300 |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.6 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 465995 |
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller





 Z
Z Y
Y X
X