+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | The Arabidopsis CLCa transporter bound with chloride, ATP and PIP2 | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Transporter / ATP / PIP2 / MEMBRANE PROTEIN | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報nitrate:proton symporter activity / nitrate transmembrane transport / nitrate transmembrane transporter activity / response to nitrate / plant-type vacuole membrane / voltage-gated chloride channel activity / chloride transport / chloride channel complex 類似検索 - 分子機能 | |||||||||
| 生物種 |  | |||||||||
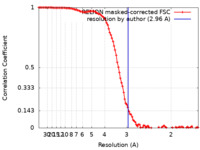
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.96 Å | |||||||||
 データ登録者 データ登録者 | Yang Z / Zhang X / Zhang P | |||||||||
| 資金援助 |  中国, 2件 中国, 2件
| |||||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2023 ジャーナル: Nat Commun / 年: 2023タイトル: Molecular mechanism underlying regulation of Arabidopsis CLCa transporter by nucleotides and phospholipids. 著者: Zhao Yang / Xue Zhang / Shiwei Ye / Jingtao Zheng / Xiaowei Huang / Fang Yu / Zhenguo Chen / Shiqing Cai / Peng Zhang /  要旨: Chloride channels (CLCs) transport anion across membrane to regulate ion homeostasis and acidification of intracellular organelles, and are divided into anion channels and anion/proton antiporters. ...Chloride channels (CLCs) transport anion across membrane to regulate ion homeostasis and acidification of intracellular organelles, and are divided into anion channels and anion/proton antiporters. Arabidopsis thaliana CLCa (AtCLCa) transporter localizes to the tonoplast which imports NO and to a less extent Cl from cytoplasm. The activity of AtCLCa and many other CLCs is regulated by nucleotides and phospholipids, however, the molecular mechanism remains unclear. Here we determine the cryo-EM structures of AtCLCa bound with NO and Cl, respectively. Both structures are captured in ATP and PI(4,5)P bound conformation. Structural and electrophysiological analyses reveal a previously unidentified N-terminal β-hairpin that is stabilized by ATP binding to block the anion transport pathway, thereby inhibiting the AtCLCa activity. While AMP loses the inhibition capacity due to lack of the β/γ- phosphates required for β-hairpin stabilization. This well explains how AtCLCa senses the ATP/AMP status to regulate the physiological nitrogen-carbon balance. Our data further show that PI(4,5)P or PI(3,5)P binds to the AtCLCa dimer interface and occupies the proton-exit pathway, which may help to understand the inhibition of AtCLCa by phospholipids to facilitate guard cell vacuole acidification and stomatal closure. In a word, our work suggests the regulatory mechanism of AtCLCa by nucleotides and phospholipids under certain physiological scenarios and provides new insights for future study of CLCs. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_35299.map.gz emd_35299.map.gz | 12 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-35299-v30.xml emd-35299-v30.xml emd-35299.xml emd-35299.xml | 17.4 KB 17.4 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_35299_fsc.xml emd_35299_fsc.xml | 10.7 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_35299.png emd_35299.png | 125.5 KB | ||
| その他 |  emd_35299_additional_1.map.gz emd_35299_additional_1.map.gz emd_35299_half_map_1.map.gz emd_35299_half_map_1.map.gz emd_35299_half_map_2.map.gz emd_35299_half_map_2.map.gz | 96.4 MB 79.6 MB 79.6 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35299 http://ftp.pdbj.org/pub/emdb/structures/EMD-35299 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35299 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35299 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_35299_validation.pdf.gz emd_35299_validation.pdf.gz | 748.3 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_35299_full_validation.pdf.gz emd_35299_full_validation.pdf.gz | 747.9 KB | 表示 | |
| XML形式データ |  emd_35299_validation.xml.gz emd_35299_validation.xml.gz | 17.6 KB | 表示 | |
| CIF形式データ |  emd_35299_validation.cif.gz emd_35299_validation.cif.gz | 22.7 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35299 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35299 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35299 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35299 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8iabMC  8iadC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_35299.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_35299.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ | X=Y=Z: 0.83 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-追加マップ: unmasked map
| ファイル | emd_35299_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | unmasked map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_35299_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_35299_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : The Arabidopsis CLCa transporter
| 全体 | 名称: The Arabidopsis CLCa transporter |
|---|---|
| 要素 |
|
-超分子 #1: The Arabidopsis CLCa transporter
| 超分子 | 名称: The Arabidopsis CLCa transporter / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1 |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: Chloride channel protein CLC-a
| 分子 | 名称: Chloride channel protein CLC-a / タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 85.488906 KDa |
| 組換発現 | 生物種:  Komagataella pastoris (菌類) Komagataella pastoris (菌類) |
| 配列 | 文字列: MDEDGNLQIS NSNYNGEEEG EDPENNTLNQ PLLKRHRTLS STPLALVGAK VSHIESLDYE INENDLFKHD WRSRSKAQVF QYIFLKWTL ACLVGLFTGL IATLINLAVE NIAGYKLLAV GYYIAQDRFW TGLMVFTGAN LGLTLVATVL VVYFAPTAAG P GIPEIKAY ...文字列: MDEDGNLQIS NSNYNGEEEG EDPENNTLNQ PLLKRHRTLS STPLALVGAK VSHIESLDYE INENDLFKHD WRSRSKAQVF QYIFLKWTL ACLVGLFTGL IATLINLAVE NIAGYKLLAV GYYIAQDRFW TGLMVFTGAN LGLTLVATVL VVYFAPTAAG P GIPEIKAY LNGIDTPNMF GFTTMMVKIV GSIGAVAAGL DLGKEGPLVH IGSCIASLLG QGGPDNHRIK WRWLRYFNND RD RRDLITC GSASGVCAAF RSPVGGVLFA LEEVATWWRS ALLWRTFFST AVVVVVLRAF IEICNSGKCG LFGSGGLIMF DVS HVEVRY HAADIIPVTL IGVFGGILGS LYNHLLHKVL RLYNLINQKG KIHKVLLSLG VSLFTSVCLF GLPFLAECKP CDPS IDEIC PTNGRSGNFK QFNCPNGYYN DLSTLLLTTN DDAVRNIFSS NTPNEFGMVS LWIFFGLYCI LGLITFGIAT PSGLF LPII LMGSAYGRML GTAMGSYTNI DQGLYAVLGA ASLMAGSMRM TVSLCVIFLE LTNNLLLLPI TMFVLLIAKT VGDSFN LSI YEIILHLKGL PFLEANPEPW MRNLTVGELN DAKPPVVTLN GVEKVANIVD VLRNTTHNAF PVLDGADQNT GTELHGL IL RAHLVKVLKK RWFLNEKRRT EEWEVREKFT PVELAEREDN FDDVAITSSE MQLYVDLHPL TNTTPYTVVQ SMSVAKAL V LFRSVGLRHL LVVPKIQASG MSPVIGILTR QDLRAYNILQ AFPHLDKHKS GKAR UniProtKB: Chloride channel protein CLC-a |
-分子 #2: ADENOSINE-5'-TRIPHOSPHATE
| 分子 | 名称: ADENOSINE-5'-TRIPHOSPHATE / タイプ: ligand / ID: 2 / コピー数: 2 / 式: ATP |
|---|---|
| 分子量 | 理論値: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-分子 #3: MAGNESIUM ION
| 分子 | 名称: MAGNESIUM ION / タイプ: ligand / ID: 3 / コピー数: 2 / 式: MG |
|---|---|
| 分子量 | 理論値: 24.305 Da |
-分子 #4: [(2R)-2-octanoyloxy-3-[oxidanyl-[(1R,2R,3S,4R,5R,6S)-2,3,6-tris(o...
| 分子 | 名称: [(2R)-2-octanoyloxy-3-[oxidanyl-[(1R,2R,3S,4R,5R,6S)-2,3,6-tris(oxidanyl)-4,5-diphosphonooxy-cyclohexyl]oxy-phosphoryl]oxy-propyl] octanoate タイプ: ligand / ID: 4 / コピー数: 2 / 式: PIO |
|---|---|
| 分子量 | 理論値: 746.566 Da |
| Chemical component information |  ChemComp-PIO: |
-分子 #5: CHLORIDE ION
| 分子 | 名称: CHLORIDE ION / タイプ: ligand / ID: 5 / コピー数: 6 / 式: CL |
|---|---|
| 分子量 | 理論値: 35.453 Da |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 8 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 平均電子線量: 56.3 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.0 µm / 最小 デフォーカス(公称値): 1.0 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー






 Z
Z Y
Y X
X