+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
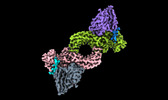
| タイトル | Structure of beta-arrestin1 in complex with a phosphopeptide corresponding to the human C5a anaphylatoxin chemotactic receptor 1, C5aR1 (Local refine) | |||||||||
 マップデータ マップデータ | Full map | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | GPCR / Arrestin / SIGNALING PROTEIN / SIGNALING PROTEIN-IMMUNE SYSTEM complex | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報V2 vasopressin receptor binding / alpha-1A adrenergic receptor binding / follicle-stimulating hormone receptor binding / complement component C5a signaling pathway / sensory perception of touch / presynapse organization / regulation of tau-protein kinase activity / G alpha (s) signalling events / alpha-1B adrenergic receptor binding / complement component C5a receptor activity ...V2 vasopressin receptor binding / alpha-1A adrenergic receptor binding / follicle-stimulating hormone receptor binding / complement component C5a signaling pathway / sensory perception of touch / presynapse organization / regulation of tau-protein kinase activity / G alpha (s) signalling events / alpha-1B adrenergic receptor binding / complement component C5a receptor activity / follicle-stimulating hormone signaling pathway / protein phosphorylated amino acid binding / angiotensin receptor binding / Lysosome Vesicle Biogenesis / response to peptidoglycan / AP-2 adaptor complex binding / Golgi Associated Vesicle Biogenesis / MAP2K and MAPK activation / Ub-specific processing proteases / positive regulation of smooth muscle cell apoptotic process / sensory perception of chemical stimulus / negative regulation of interleukin-8 production / Cargo recognition for clathrin-mediated endocytosis / Clathrin-mediated endocytosis / clathrin adaptor activity / complement receptor mediated signaling pathway / regulation of G protein-coupled receptor signaling pathway / arrestin family protein binding / G protein-coupled receptor internalization / response to morphine / positive regulation of neutrophil chemotaxis / Thrombin signalling through proteinase activated receptors (PARs) / mitogen-activated protein kinase kinase binding / clathrin binding / positive regulation of Rho protein signal transduction / positive regulation of macrophage chemotaxis / stress fiber assembly / negative regulation of Notch signaling pathway / amyloid-beta clearance / pseudopodium / positive regulation of insulin secretion involved in cellular response to glucose stimulus / negative regulation of interleukin-6 production / cysteine-type endopeptidase inhibitor activity involved in apoptotic process / positive regulation of receptor internalization / phototransduction / : / positive regulation of vascular endothelial growth factor production / cellular defense response / clathrin-coated pit / negative regulation of protein ubiquitination / visual perception / Peptide ligand-binding receptors / neutrophil chemotaxis / GTPase activator activity / secretory granule membrane / negative regulation of protein phosphorylation / positive regulation of protein ubiquitination / Regulation of Complement cascade / positive regulation of epithelial cell proliferation / G protein-coupled receptor binding / G protein-coupled receptor activity / astrocyte activation / nuclear estrogen receptor binding / phosphoprotein binding / insulin-like growth factor receptor binding / microglial cell activation / mRNA transcription by RNA polymerase II / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / negative regulation of ERK1 and ERK2 cascade / cognition / endocytosis / positive regulation of angiogenesis / chemotaxis / protein transport / apical part of cell / positive regulation of peptidyl-serine phosphorylation / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events / positive regulation of cytosolic calcium ion concentration / cytoplasmic vesicle / ubiquitin-dependent protein catabolic process / basolateral plasma membrane / postsynaptic membrane / regulation of apoptotic process / proteasome-mediated ubiquitin-dependent protein catabolic process / transmembrane transporter binding / negative regulation of neuron apoptotic process / positive regulation of MAPK cascade / dendritic spine / transcription coactivator activity / positive regulation of ERK1 and ERK2 cascade / defense response to Gram-positive bacterium / protein ubiquitination / endosome / response to xenobiotic stimulus / inflammatory response / positive regulation of protein phosphorylation / immune response / G protein-coupled receptor signaling pathway / signaling receptor binding 類似検索 - 分子機能 | |||||||||
| 生物種 |   Homo sapiens (ヒト) / Homo sapiens (ヒト) /  | |||||||||
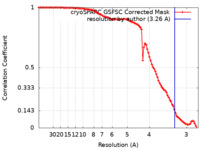
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.26 Å | |||||||||
 データ登録者 データ登録者 | Maharana J / Sarma P / Yadav MK / Banerjee R / Shukla AK | |||||||||
| 資金援助 |  インド, 1件 インド, 1件
| |||||||||
 引用 引用 |  ジャーナル: Mol Cell / 年: 2023 ジャーナル: Mol Cell / 年: 2023タイトル: Structural snapshots uncover a key phosphorylation motif in GPCRs driving β-arrestin activation. 著者: Jagannath Maharana / Parishmita Sarma / Manish K Yadav / Sayantan Saha / Vinay Singh / Shirsha Saha / Mohamed Chami / Ramanuj Banerjee / Arun K Shukla /   要旨: Agonist-induced GPCR phosphorylation is a key determinant for the binding and activation of β-arrestins (βarrs). However, it is not entirely clear how different GPCRs harboring divergent ...Agonist-induced GPCR phosphorylation is a key determinant for the binding and activation of β-arrestins (βarrs). However, it is not entirely clear how different GPCRs harboring divergent phosphorylation patterns impart converging active conformation on βarrs leading to broadly conserved functional responses such as desensitization, endocytosis, and signaling. Here, we present multiple cryo-EM structures of activated βarrs in complex with distinct phosphorylation patterns derived from the carboxyl terminus of different GPCRs. These structures help identify a P-X-P-P type phosphorylation motif in GPCRs that interacts with a spatially organized K-K-R-R-K-K sequence in the N-domain of βarrs. Sequence analysis of the human GPCRome reveals the presence of this phosphorylation pattern in a large number of receptors, and its contribution in βarr activation is demonstrated by targeted mutagenesis experiments combined with an intrabody-based conformational sensor. Taken together, our findings provide important structural insights into the ability of distinct GPCRs to activate βarrs through a significantly conserved mechanism. #1:  ジャーナル: Mol.Cell / 年: 2023 ジャーナル: Mol.Cell / 年: 2023タイトル: Structure of beta-arrestin in complex with a phosphopeptide 著者: Maharana J / Sarma P / Yadav MK / Banerjee R / Shukla AK | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_35104.map.gz emd_35104.map.gz | 86 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-35104-v30.xml emd-35104-v30.xml emd-35104.xml emd-35104.xml | 22.8 KB 22.8 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_35104_fsc.xml emd_35104_fsc.xml | 9.4 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_35104.png emd_35104.png | 46 KB | ||
| Filedesc metadata |  emd-35104.cif.gz emd-35104.cif.gz | 6.8 KB | ||
| その他 |  emd_35104_half_map_1.map.gz emd_35104_half_map_1.map.gz emd_35104_half_map_2.map.gz emd_35104_half_map_2.map.gz | 84.3 MB 84.3 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35104 http://ftp.pdbj.org/pub/emdb/structures/EMD-35104 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35104 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35104 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_35104_validation.pdf.gz emd_35104_validation.pdf.gz | 956 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_35104_full_validation.pdf.gz emd_35104_full_validation.pdf.gz | 955.5 KB | 表示 | |
| XML形式データ |  emd_35104_validation.xml.gz emd_35104_validation.xml.gz | 17.7 KB | 表示 | |
| CIF形式データ |  emd_35104_validation.cif.gz emd_35104_validation.cif.gz | 22.9 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35104 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35104 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35104 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35104 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8i0nMC  8go8C  8gocC  8gooC  8gp3C  8i0qC  8i0zC  8i10C M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_35104.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_35104.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Full map | ||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.3667 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: Half map B
| ファイル | emd_35104_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
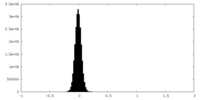
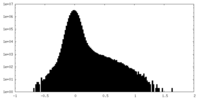
| 注釈 | Half map B | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: Half map A
| ファイル | emd_35104_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
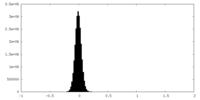
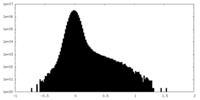
| 注釈 | Half map A | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Peptide 1 bound beta-arrestin1 in complex with Fab30 - Local refine
| 全体 | 名称: Peptide 1 bound beta-arrestin1 in complex with Fab30 - Local refine |
|---|---|
| 要素 |
|
-超分子 #1: Peptide 1 bound beta-arrestin1 in complex with Fab30 - Local refine
| 超分子 | 名称: Peptide 1 bound beta-arrestin1 in complex with Fab30 - Local refine タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 分子量 | 理論値: 190 KDa |
-超分子 #2: beta-arrestin1
| 超分子 | 名称: beta-arrestin1 / タイプ: complex / ID: 2 / 親要素: 1 / 含まれる分子: #1 |
|---|---|
| 由来(天然) | 生物種:  |
-超分子 #3: C5a anaphylatoxin chemotactic receptor 1
| 超分子 | 名称: C5a anaphylatoxin chemotactic receptor 1 / タイプ: complex / ID: 3 / 親要素: 1 / 含まれる分子: #4 / 詳細: Chemically synthesized |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) / Synthetically produced: Yes Homo sapiens (ヒト) / Synthetically produced: Yes |
-超分子 #4: Fab30
| 超分子 | 名称: Fab30 / タイプ: complex / ID: 4 / 親要素: 1 / 含まれる分子: #2-#3 |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: Beta-arrestin-1
| 分子 | 名称: Beta-arrestin-1 / タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 47.088508 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MGDKGTRVFK KASPNGKLTV YLGKRDFVDH IDLVDPVDGV VLVDPEYLKE RRVYVTLTCA FRYGREDLDV LGLTFRKDLF VANVQSFPP APEDKKPLTR LQERLIKKLG EHAYPFTFEI PPNLPCSVTL QPGPEDTGKA CGVDYEVKAF CAENLEEKIH K RNSVRLVI ...文字列: MGDKGTRVFK KASPNGKLTV YLGKRDFVDH IDLVDPVDGV VLVDPEYLKE RRVYVTLTCA FRYGREDLDV LGLTFRKDLF VANVQSFPP APEDKKPLTR LQERLIKKLG EHAYPFTFEI PPNLPCSVTL QPGPEDTGKA CGVDYEVKAF CAENLEEKIH K RNSVRLVI RKVQYAPERP GPQPTAETTR QFLMSDKPLH LEASLDKEIY YHGEPISVNV HVTNNTNKTV KKIKISVRQY AD ICLFNTA QYKCPVAMEE ADDTVAPSST FCKVYTLTPF LANNREKRGL ALDGKLKHED TNLASSTLLR EGANREILGI IVS YKVKVK LVVSRGGLLG DLASSDVAVE LPFTLMHPKP KEEPPHREVP ESETPVDTNL IELDTNDDDI VFEDFARQRL KGMK DDKDE EDDGTGSPHL NNR UniProtKB: Beta-arrestin-1 |
-分子 #2: Fab30 heavy chain
| 分子 | 名称: Fab30 heavy chain / タイプ: protein_or_peptide / ID: 2 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 25.512354 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: EISEVQLVES GGGLVQPGGS LRLSCAASGF NVYSSSIHWV RQAPGKGLEW VASISSYYGY TYYADSVKGR FTISADTSKN TAYLQMNSL RAEDTAVYYC ARSRQFWYSG LDYWGQGTLV TVSSASTKGP SVFPLAPSSK STSGGTAALG CLVKDYFPEP V TVSWNSGA ...文字列: EISEVQLVES GGGLVQPGGS LRLSCAASGF NVYSSSIHWV RQAPGKGLEW VASISSYYGY TYYADSVKGR FTISADTSKN TAYLQMNSL RAEDTAVYYC ARSRQFWYSG LDYWGQGTLV TVSSASTKGP SVFPLAPSSK STSGGTAALG CLVKDYFPEP V TVSWNSGA LTSGVHTFPA VLQSSGLYSL SSVVTVPSSS LGTQTYICNV NHKPSNTKVD KKVEPKSCDK THHHHHHHH |
-分子 #3: Fab30 light chain
| 分子 | 名称: Fab30 light chain / タイプ: protein_or_peptide / ID: 3 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 23.435064 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: SDIQMTQSPS SLSASVGDRV TITCRASQSV SSAVAWYQQK PGKAPKLLIY SASSLYSGVP SRFSGSRSGT DFTLTISSLQ PEDFATYYC QQYKYVPVTF GQGTKVEIKR TVAAPSVFIF PPSDSQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN S QESVTEQD ...文字列: SDIQMTQSPS SLSASVGDRV TITCRASQSV SSAVAWYQQK PGKAPKLLIY SASSLYSGVP SRFSGSRSGT DFTLTISSLQ PEDFATYYC QQYKYVPVTF GQGTKVEIKR TVAAPSVFIF PPSDSQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN S QESVTEQD SKDSTYSLSS TLTLSKADYE KHKVYACEVT HQGLSSPVTK SFNRGEC |
-分子 #4: C5a anaphylatoxin chemotactic receptor 1
| 分子 | 名称: C5a anaphylatoxin chemotactic receptor 1 / タイプ: protein_or_peptide / ID: 4 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 2.69834 KDa |
| 配列 | 文字列: E(SEP)K(SEP)F(TPO)R(SEP)(TPO)V D(TPO)MAQKTQAV UniProtKB: C5a anaphylatoxin chemotactic receptor 1 |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 構成要素:
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 90 % / チャンバー内温度: 283.15 K / 装置: LEICA EM GP / 詳細: Blotted for 3 seconds before plunging.. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: COUNTING / 実像数: 6212 / 平均電子線量: 56.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 0.5 µm / 倍率(公称値): 165000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー

























 Z
Z Y
Y X
X