+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM structure of the bi-functional malonyl-CoA reductase from Roseiflexus castenholzii | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | The 3-hydroxypropionate cycle / Bifunctional enzyme / Short chain dehydrogenase / OXIDOREDUCTASE | |||||||||
| 機能・相同性 | oxidoreductase activity, acting on the CH-OH group of donors, NAD or NADP as acceptor / short chain dehydrogenase / PKS_KR / Short-chain dehydrogenase/reductase SDR / NAD(P)-binding domain superfamily / nucleotide binding / Short-chain dehydrogenase/reductase SDR 機能・相同性情報 機能・相同性情報 | |||||||||
| 生物種 |  Roseiflexus castenholzii DSM 13941 (バクテリア) Roseiflexus castenholzii DSM 13941 (バクテリア) | |||||||||
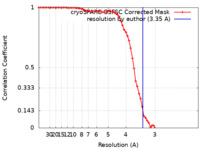
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / ネガティブ染色法 / 解像度: 3.35 Å | |||||||||
 データ登録者 データ登録者 | Zhang X / Xu X / Xin J | |||||||||
| 資金援助 |  中国, 1件 中国, 1件
| |||||||||
 引用 引用 |  ジャーナル: mBio / 年: 2023 ジャーナル: mBio / 年: 2023タイトル: Structural basis of a bi-functional malonyl-CoA reductase (MCR) from the photosynthetic green non-sulfur bacterium . 著者: Xin Zhang / Jiyu Xin / Zhiguo Wang / Wenping Wu / Yutong Liu / Zhenzhen Min / Yueyong Xin / Bing Liu / Jun He / Xingwei Zhang / Xiaoling Xu /  要旨: Malonyl-CoA reductase (MCR) is a NADPH-dependent bi-functional enzyme that performs alcohol dehydrogenase and aldehyde dehydrogenase (CoA-acylating) activities in the N- and C-terminal fragments, ...Malonyl-CoA reductase (MCR) is a NADPH-dependent bi-functional enzyme that performs alcohol dehydrogenase and aldehyde dehydrogenase (CoA-acylating) activities in the N- and C-terminal fragments, respectively. It catalyzes the two-step reduction of malonyl-CoA to 3-hydroxypropionate (3-HP), a key reaction in the autotrophic CO fixation cycles of green non-sulfur bacteria and the archaea . However, the structural basis underlying substrate selection, coordination, and the subsequent catalytic reactions of full-length MCR is largely unknown. For the first time, we here determined the structure of full-length MCR from the photosynthetic green non-sulfur bacterium (MCR) at 3.35 Å resolution. Furthermore, we determined the crystal structures of the N- and C-terminal fragments bound with reaction intermediates NADP and malonate semialdehyde (MSA) at 2.0 Å and 2.3 Å, respectively, and elucidated the catalytic mechanisms using a combination of molecular dynamics simulations and enzymatic analyses. Full-length MCR was a homodimer of two cross-interlocked subunits, each containing four tandemly arranged short-chain dehydrogenase/reductase (SDR) domains. Only the catalytic domains SDR1 and SDR3 incorporated additional secondary structures that changed with NADP-MSA binding. The substrate, malonyl-CoA, was immobilized in the substrate-binding pocket of SDR3 through coordination with Arg1164 and Arg799 of SDR4 and the extra domain, respectively. Malonyl-CoA was successively reduced through protonation by the Tyr743-Arg746 pair in SDR3 and the catalytic triad (Thr165-Tyr178-Lys182) in SDR1 after nucleophilic attack from NADPH hydrides. IMPORTANCE The bi-functional MCR catalyzes NADPH-dependent reduction of malonyl-CoA to 3-HP, an important metabolic intermediate and platform chemical, from biomass. The individual MCR-N and MCR-C fragments, which contain the alcohol dehydrogenase and aldehyde dehydrogenase (CoA-acylating) activities, respectively, have previously been structurally investigated and reconstructed into a malonyl-CoA pathway for the biosynthetic production of 3-HP. However, no structural information for full-length MCR has been available to illustrate the catalytic mechanism of this enzyme, which greatly limits our capacity to increase the 3-HP yield of recombinant strains. Here, we report the cryo-electron microscopy structure of full-length MCR for the first time and elucidate the mechanisms underlying substrate selection, coordination, and catalysis in the bi-functional MCR. These findings provide a structural and mechanistic basis for enzyme engineering and biosynthetic applications of the 3-HP carbon fixation pathways. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_34812.map.gz emd_34812.map.gz | 28.6 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-34812-v30.xml emd-34812-v30.xml emd-34812.xml emd-34812.xml | 15 KB 15 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_34812_fsc.xml emd_34812_fsc.xml | 6.6 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_34812.png emd_34812.png | 126.5 KB | ||
| その他 |  emd_34812_half_map_1.map.gz emd_34812_half_map_1.map.gz emd_34812_half_map_2.map.gz emd_34812_half_map_2.map.gz | 28 MB 28.1 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34812 http://ftp.pdbj.org/pub/emdb/structures/EMD-34812 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34812 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34812 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_34812_validation.pdf.gz emd_34812_validation.pdf.gz | 730.2 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_34812_full_validation.pdf.gz emd_34812_full_validation.pdf.gz | 729.7 KB | 表示 | |
| XML形式データ |  emd_34812_validation.xml.gz emd_34812_validation.xml.gz | 13.9 KB | 表示 | |
| CIF形式データ |  emd_34812_validation.cif.gz emd_34812_validation.cif.gz | 17.6 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34812 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34812 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34812 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34812 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8hi4MC  8hi5C  8hi6C M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_34812.map.gz / 形式: CCP4 / 大きさ: 30.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_34812.map.gz / 形式: CCP4 / 大きさ: 30.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ | X=Y=Z: 1.07 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: #2
| ファイル | emd_34812_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
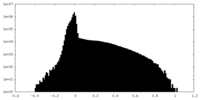
| 投影像・断面図 |
| ||||||||||||
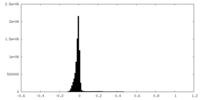
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_34812_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
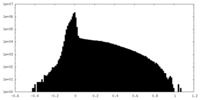
| 投影像・断面図 |
| ||||||||||||
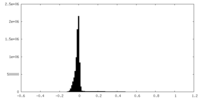
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Malonyl-coenzyme A reductase/Malonate semialdehyde reductase/Shor...
| 全体 | 名称: Malonyl-coenzyme A reductase/Malonate semialdehyde reductase/Short-chain dehydrogenase |
|---|---|
| 要素 |
|
-超分子 #1: Malonyl-coenzyme A reductase/Malonate semialdehyde reductase/Shor...
| 超分子 | 名称: Malonyl-coenzyme A reductase/Malonate semialdehyde reductase/Short-chain dehydrogenase タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all 詳細: The full-length MCR is a homodimer both in solution and cryo-EM structure. |
|---|---|
| 由来(天然) | 生物種:  Roseiflexus castenholzii DSM 13941 (バクテリア) Roseiflexus castenholzii DSM 13941 (バクテリア) |
-分子 #1: Short-chain dehydrogenase/reductase SDR
| 分子 | 名称: Short-chain dehydrogenase/reductase SDR / タイプ: protein_or_peptide / ID: 1 詳細: F269L is a natural mutation occurred during recombinant expression of the protein コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Roseiflexus castenholzii DSM 13941 (バクテリア) Roseiflexus castenholzii DSM 13941 (バクテリア) |
| 分子量 | 理論値: 134.268375 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MSTVRRLEGK VALITGGAGN IGEVITRRFL AEGATVVITG RNAEKLAVYR RRLIDEERVA PERVVALRMD GSDIAQVRAG VAQIVHGGT DVPIPLHRID ILVNNAGSAG PRRRLVDIPL EPSEVQPPDS ETLAQAVGNL VGITWNLTRA AAPHMPSGSS V INISTIFS ...文字列: MSTVRRLEGK VALITGGAGN IGEVITRRFL AEGATVVITG RNAEKLAVYR RRLIDEERVA PERVVALRMD GSDIAQVRAG VAQIVHGGT DVPIPLHRID ILVNNAGSAG PRRRLVDIPL EPSEVQPPDS ETLAQAVGNL VGITWNLTRA AAPHMPSGSS V INISTIFS RTDYYGRIAY VAPKAALNAL SDGLARELGV RGIRVNTIYP GPIESERIYT MFQAMDALKG QPEGDTASGF LR MMRLSRI DQNGEVVKRF PSPVDVANTA VLLASDESAA FTGHAFEVTH GMEVPTESRT TFVSRPGLRS VDATGKVILI CAG DQVDDA VALADTLRSC RATVVIGFRD PRALEKASVL LREPRHALAA DMYGRPTMTA EARLVRLDPL DPRAAAQTLE QIHA ELGAI HHAVVLPGQS RHAPSASLIE VDDQVVERFL HQELVGTIAL ARELARFWEE YPSGSSMHRV LFVSNPDDQQ GNQYS HILR AAVEQLVRVW RHESEYDSVN PAHQQEGQSS AAVWANQLIR YVNNEMANLD FTCAWVAKLL GSDRRIAEIN LYLPEE IVG TIGVHNPGFG WAESLFGLHM GKVALITGGS AGIGGQIGRL LALSGAHVML AARNADQLEQ MRASIVREVR DASYPDA ES RVAIFPGSDV SDIDGLERLV NHTVRVFGKV DYLINNAGIA GAEEMVIDMP VDAWRHTLRA NLISNYALLR RLAPQMKA A GGAYVLNVSS YFGGEKYVAI PYPNRSDYAV SKAGQRAMVE SLARFLGPEI QINAIAPGPV EGERLKGAGS RPGLFMRRA RLILENKRLN EVFAALLAAR HEGATIADLL PDLFANDIQS IANSAAMPAP LRRLATMLRE TSDAGGSAQS YLMNATIARK LLNRLENGG YITLHDRRAL TVEPPEPFFT EAQIEREAIK VRDGILGMLH LQRMPTEFDV ALATVFYLAD RNVTGETFHP S GGLRFERT VTEGELFGKP GQQRLERLKG SVVYLIGEHL RQHLVLLART FLDEIHVARV VLLTETTQAA TDLAAELSDY EA AGRFVVI PTCGDIEGGI DRAMAEYGRP GPVISTPFRP LPDRALSARN GDWSSVLTTA EFEELVEQQI THHFRVARKA GLI EGANVT LVTPPTSARS TSEEFALANF VKTTLHALTA TAGAESERTV PHVPVNQVDL TRRARSEEPR TPSEEEEELQ RFVN AVLLT SAPLPTPLES RYRARIYRGN AITV UniProtKB: Short-chain dehydrogenase/reductase SDR |
-実験情報
-構造解析
| 手法 | ネガティブ染色法, クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 0.30 mg/mL |
|---|---|
| 緩衝液 | pH: 8 |
| 染色 | タイプ: NEGATIVE / 材質: Uranyl Acetate |
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 平均電子線量: 60.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 1.8 µm / 最小 デフォーカス(公称値): 1.3 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー






 Z
Z Y
Y X
X