+ Open data
Open data
- Basic information
Basic information
| Entry | 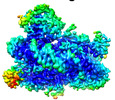 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | histone methyltransferase | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | histone methyltransferase / GENE REGULATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationhistone H4K20me methyltransferase activity / [histone H4]-N-methyl-L-lysine20 N-methyltransferase / histone H4K20 monomethyltransferase activity / [histone H4]-lysine20 N-methyltransferase / muscle organ development / RNA polymerase II core promoter sequence-specific DNA binding / heterochromatin / nucleosomal DNA binding / cellular response to estradiol stimulus / euchromatin ...histone H4K20me methyltransferase activity / [histone H4]-N-methyl-L-lysine20 N-methyltransferase / histone H4K20 monomethyltransferase activity / [histone H4]-lysine20 N-methyltransferase / muscle organ development / RNA polymerase II core promoter sequence-specific DNA binding / heterochromatin / nucleosomal DNA binding / cellular response to estradiol stimulus / euchromatin / heterochromatin formation / chromatin DNA binding / structural constituent of chromatin / nucleosome / nucleosome assembly / chromatin organization / methylation / RNA polymerase II cis-regulatory region sequence-specific DNA binding / protein heterodimerization activity / Amyloid fiber formation / positive regulation of transcription by RNA polymerase II / DNA binding / extracellular exosome / nucleoplasm / nucleus Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) / | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | Li H / Wang WY | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2023 Journal: Mol Cell / Year: 2023Title: Structural insight into H4K20 methylation on H2A.Z-nucleosome by SUV420H1. Authors: Li Huang / Youwang Wang / Haizhen Long / Haoqiang Zhu / Zengqi Wen / Liwei Zhang / Wenhao Zhang / Zhenqian Guo / Longge Wang / Fangyi Tang / Jie Hu / Keyan Bao / Ping Zhu / Guohong Li / Zheng Zhou /  Abstract: DNA replication ensures the accurate transmission of genetic information during the cell cycle. Histone variant H2A.Z is crucial for early replication origins licensing and activation in which ...DNA replication ensures the accurate transmission of genetic information during the cell cycle. Histone variant H2A.Z is crucial for early replication origins licensing and activation in which SUV420H1 preferentially recognizes H2A.Z-nucleosome and deposits H4 lysine 20 dimethylation (H4K20me2) on replication origins. Here, we report the cryo-EM structures of SUV420H1 bound to H2A.Z-nucleosome or H2A-nucleosome and demonstrate that SUV420H1 directly interacts with H4 N-terminal tail, the DNA, and the acidic patch in the nucleosome. The H4 (1-24) forms a lasso-shaped structure that stabilizes the SUV420H1-nucleosome complex and precisely projects the H4K20 residue into the SUV420H1 catalytic center. In vitro and in vivo analyses reveal a crucial role of the SUV420H1 KR loop (residues 214-223), which lies close to the H2A.Z-specific residues D97/S98, in H2A.Z-nucleosome preferential recognition. Together, our findings elucidate how SUV420H1 recognizes nucleosomes to ensure site-specific H4K20me2 modification and provide insights into how SUV420H1 preferentially recognizes H2A.Z nucleosome. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_34053.map.gz emd_34053.map.gz | 1.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-34053-v30.xml emd-34053-v30.xml emd-34053.xml emd-34053.xml | 19.9 KB 19.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_34053.png emd_34053.png | 168.9 KB | ||
| Filedesc metadata |  emd-34053.cif.gz emd-34053.cif.gz | 6.3 KB | ||
| Others |  emd_34053_half_map_1.map.gz emd_34053_half_map_1.map.gz emd_34053_half_map_2.map.gz emd_34053_half_map_2.map.gz | 23.4 MB 23.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34053 http://ftp.pdbj.org/pub/emdb/structures/EMD-34053 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34053 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34053 | HTTPS FTP |
-Validation report
| Summary document |  emd_34053_validation.pdf.gz emd_34053_validation.pdf.gz | 676.6 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_34053_full_validation.pdf.gz emd_34053_full_validation.pdf.gz | 676.2 KB | Display | |
| Data in XML |  emd_34053_validation.xml.gz emd_34053_validation.xml.gz | 9.4 KB | Display | |
| Data in CIF |  emd_34053_validation.cif.gz emd_34053_validation.cif.gz | 11 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34053 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34053 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34053 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34053 | HTTPS FTP |
-Related structure data
| Related structure data |  7yrdMC  7yrgC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_34053.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_34053.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_34053_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
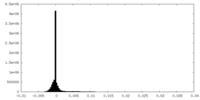
| Density Histograms |
-Half map: #1
| File | emd_34053_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
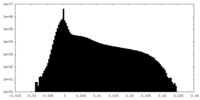
| Density Histograms |
- Sample components
Sample components
-Entire : histone methyltransferase and nucleosome complex
| Entire | Name: histone methyltransferase and nucleosome complex |
|---|---|
| Components |
|
-Supramolecule #1: histone methyltransferase and nucleosome complex
| Supramolecule | Name: histone methyltransferase and nucleosome complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Histone H3.2
| Macromolecule | Name: Histone H3.2 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 12.046091 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GEVKKPHRYR PGTVALREIR RYQKSTELLI RKLPFQRLVR EIAQDFKTDL RFQSSAVMAL QEASEAYLVA LFEDTNLCAI HAKRVTIMP KDIQLARRIR GERA UniProtKB: Histone H3.2 |
-Macromolecule #2: Histone H4
| Macromolecule | Name: Histone H4 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 11.265247 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGRGKGGKGL GKGGAKRHR(ECX) VLRDNIQGIT KPAIRRLARR GGVKRISGLI YEETRGVLKV FLENVIRDAV TYTEHA KRK TVTAMDVVYA LKRQGRTLYG FGG UniProtKB: Histone H4 |
-Macromolecule #4: Histone H2A.Z
| Macromolecule | Name: Histone H2A.Z / type: protein_or_peptide / ID: 4 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 12.091093 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GKAKTKAVSR SQRAGLQFPV GRIHRHLKSR TTSHGRVGAT AAVYSAAILE YLTAEVLELA GNASKDLKVK RITPRHLQLA IRGDEELDS LIKATIAGGG VIPHIHKSLI GKKG UniProtKB: Histone H2A.Z |
-Macromolecule #5: Histone H2B 1.1
| Macromolecule | Name: Histone H2B 1.1 / type: protein_or_peptide / ID: 5 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 10.607212 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: KTRKESYAIY VYKVLKQVHP DTGISSKAMS IMNSFVNDVF ERIAGEASRL AHYNKRSTIT SREIQTAVRL LLPGELAKHA VSEGTKAVT KYTSAK UniProtKB: Histone H2B 1.1 |
-Macromolecule #6: [histone H4]-N-methyl-L-lysine20 N-methyltransferase KMT5B
| Macromolecule | Name: [histone H4]-N-methyl-L-lysine20 N-methyltransferase KMT5B type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO / EC number: [histone H4]-lysine20 N-methyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 32.214787 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GQSRYVPSSG MSAKELCEND DLATSLVLDP YLGFQTHKMN TRFRPIKGRQ EELKEVIERF KKDEHLEKAF KCLTSGEWAR HYFLNKNKM QEKLFKEHVF IYLRMFATDS GFEILPCNRY SSEQNGAKIV ATKEWKRNDK IELLVGCIAE LSEIEENMLL R HGENDFSV ...String: GQSRYVPSSG MSAKELCEND DLATSLVLDP YLGFQTHKMN TRFRPIKGRQ EELKEVIERF KKDEHLEKAF KCLTSGEWAR HYFLNKNKM QEKLFKEHVF IYLRMFATDS GFEILPCNRY SSEQNGAKIV ATKEWKRNDK IELLVGCIAE LSEIEENMLL R HGENDFSV MYSTRKNCAQ LWLGPAAFIN HDCRPNCKFV STGRDTACVK ALRDIEPGEE ISCYYGDGFF GENNEFCECY TC ERRGTGA FKSRVGLPAP APVINSKYGL RETDKRLNRL KK UniProtKB: [histone H4]-N-methyl-L-lysine20 N-methyltransferase KMT5B |
-Macromolecule #3: DNA (146-MER)
| Macromolecule | Name: DNA (146-MER) / type: dna / ID: 3 / Number of copies: 2 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 45.054844 KDa |
| Sequence | String: (DA)(DT)(DC)(DA)(DA)(DT)(DA)(DT)(DC)(DC) (DA)(DC)(DC)(DT)(DG)(DC)(DA)(DG)(DA)(DT) (DT)(DC)(DT)(DA)(DC)(DC)(DA)(DA)(DA) (DA)(DG)(DT)(DG)(DT)(DA)(DT)(DT)(DT)(DG) (DG) (DA)(DA)(DA)(DC)(DT)(DG) ...String: (DA)(DT)(DC)(DA)(DA)(DT)(DA)(DT)(DC)(DC) (DA)(DC)(DC)(DT)(DG)(DC)(DA)(DG)(DA)(DT) (DT)(DC)(DT)(DA)(DC)(DC)(DA)(DA)(DA) (DA)(DG)(DT)(DG)(DT)(DA)(DT)(DT)(DT)(DG) (DG) (DA)(DA)(DA)(DC)(DT)(DG)(DC)(DT) (DC)(DC)(DA)(DT)(DC)(DA)(DA)(DA)(DA)(DG) (DG)(DC) (DA)(DT)(DG)(DT)(DT)(DC)(DA) (DG)(DC)(DG)(DG)(DA)(DA)(DT)(DT)(DC)(DC) (DG)(DC)(DT) (DG)(DA)(DA)(DC)(DA)(DT) (DG)(DC)(DC)(DT)(DT)(DT)(DT)(DG)(DA)(DT) (DG)(DG)(DA)(DG) (DC)(DA)(DG)(DT)(DT) (DT)(DC)(DC)(DA)(DA)(DA)(DT)(DA)(DC)(DA) (DC)(DT)(DT)(DT)(DT) (DG)(DG)(DT)(DA) (DG)(DA)(DA)(DT)(DC)(DT)(DG)(DC)(DA)(DG) (DG)(DT)(DG)(DG)(DA)(DT) (DA)(DT)(DT) (DG)(DA)(DT) |
-Macromolecule #7: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 7 / Number of copies: 1 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #8: S-ADENOSYLMETHIONINE
| Macromolecule | Name: S-ADENOSYLMETHIONINE / type: ligand / ID: 8 / Number of copies: 1 / Formula: SAM |
|---|---|
| Molecular weight | Theoretical: 398.437 Da |
| Chemical component information |  ChemComp-SAM: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.3000000000000003 µm / Nominal defocus min: 1.8 µm |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.2 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 223000 |
| Initial angle assignment | Type: RANDOM ASSIGNMENT |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller








 Z
Z Y
Y X
X