[English] 日本語
 Yorodumi
Yorodumi- EMDB-33832: CryoEM structure of Arabidopsis ROS1 in complex with TG mismatch ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of Arabidopsis ROS1 in complex with TG mismatch dsDNA at 3.3 Angstroms resolution | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ROS1 / DNA glycosylase / DNA demethylation / DNA BINDING PROTEIN / DNA BINDING PROTEIN-DNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationDNA demethylase activity / chromosomal 5-methylcytosine DNA demethylation pathway / positive regulation of male gonad development / Transcriptional regulation of testis differentiation / sex differentiation / DNA N-glycosylase activity / male sex determination / defense response to fungus / DNA-(apurinic or apyrimidinic site) endonuclease activity / class I DNA-(apurinic or apyrimidinic site) endonuclease activity ...DNA demethylase activity / chromosomal 5-methylcytosine DNA demethylation pathway / positive regulation of male gonad development / Transcriptional regulation of testis differentiation / sex differentiation / DNA N-glycosylase activity / male sex determination / defense response to fungus / DNA-(apurinic or apyrimidinic site) endonuclease activity / class I DNA-(apurinic or apyrimidinic site) endonuclease activity / DNA-(apurinic or apyrimidinic site) lyase / Deactivation of the beta-catenin transactivating complex / base-excision repair / 4 iron, 4 sulfur cluster binding / DNA-binding transcription activator activity, RNA polymerase II-specific / DNA-binding transcription factor binding / DNA-binding transcription factor activity, RNA polymerase II-specific / calmodulin binding / cell differentiation / nuclear speck / RNA polymerase II cis-regulatory region sequence-specific DNA binding / DNA repair / positive regulation of gene expression / positive regulation of DNA-templated transcription / chromatin / nucleolus / positive regulation of transcription by RNA polymerase II / DNA binding / nucleoplasm / metal ion binding / nucleus / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | |||||||||
 Authors Authors | Du X / Du J | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Nat Plants / Year: 2023 Journal: Nat Plants / Year: 2023Title: Molecular basis of the plant ROS1-mediated active DNA demethylation. Authors: Xuan Du / Zhenlin Yang / Guohui Xie / Changshi Wang / Laixing Zhang / Kaige Yan / Maojun Yang / Sisi Li / Jian-Kang Zhu / Jiamu Du /   Abstract: Active DNA demethylation plays a crucial role in eukaryotic gene imprinting and antagonizing DNA methylation. The plant-specific REPRESSOR OF SILENCING 1/DEMETER (ROS1/DME) family of enzymes directly ...Active DNA demethylation plays a crucial role in eukaryotic gene imprinting and antagonizing DNA methylation. The plant-specific REPRESSOR OF SILENCING 1/DEMETER (ROS1/DME) family of enzymes directly excise 5-methyl-cytosine (5mC), representing an efficient DNA demethylation pathway distinct from that of animals. Here, we report the cryo-electron microscopy structures of an Arabidopsis ROS1 catalytic fragment in complex with substrate DNA, mismatch DNA and reaction intermediate, respectively. The substrate 5mC is flipped-out from the DNA duplex and subsequently recognized by the ROS1 base-binding pocket through hydrophobic and hydrogen-bonding interactions towards the 5-methyl group and Watson-Crick edge respectively, while the different protonation states of the bases determine the substrate preference for 5mC over T:G mismatch. Together with the structure of the reaction intermediate complex, our structural and biochemical studies revealed the molecular basis for substrate specificity, as well as the reaction mechanism underlying 5mC demethylation by the ROS1/DME family of plant-specific DNA demethylases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33832.map.gz emd_33832.map.gz | 12.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33832-v30.xml emd-33832-v30.xml emd-33832.xml emd-33832.xml | 20.7 KB 20.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_33832.png emd_33832.png | 83.2 KB | ||
| Filedesc metadata |  emd-33832.cif.gz emd-33832.cif.gz | 6.8 KB | ||
| Others |  emd_33832_half_map_1.map.gz emd_33832_half_map_1.map.gz emd_33832_half_map_2.map.gz emd_33832_half_map_2.map.gz | 195 MB 195.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33832 http://ftp.pdbj.org/pub/emdb/structures/EMD-33832 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33832 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33832 | HTTPS FTP |
-Related structure data
| Related structure data |  7yhoMC  7yhpC  7yhqC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_33832.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33832.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.67 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_33832_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
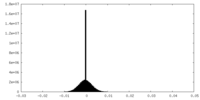
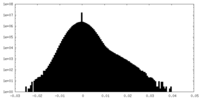
| Density Histograms |
-Half map: #1
| File | emd_33832_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
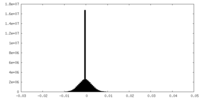
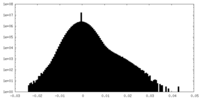
| Density Histograms |
- Sample components
Sample components
-Entire : ROS1-TG mismatch dsDNA complex
| Entire | Name: ROS1-TG mismatch dsDNA complex |
|---|---|
| Components |
|
-Supramolecule #1: ROS1-TG mismatch dsDNA complex
| Supramolecule | Name: ROS1-TG mismatch dsDNA complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|
-Supramolecule #2: ROS1
| Supramolecule | Name: ROS1 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: TG mismatch dsDNA
| Supramolecule | Name: TG mismatch dsDNA / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2-#3 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Sex-determining region Y protein,DNA glycosylase/AP lyase ROS1
| Macromolecule | Name: Sex-determining region Y protein,DNA glycosylase/AP lyase ROS1 type: protein_or_peptide / ID: 1 Details: Fusion protein of residues 56 to 130 of database UNP Q05066, Linker, residues 511 to 1393 of database UNP Q9SJQ6, with deletion of residues 665-835 and 1074-1109. Number of copies: 1 / Enantiomer: LEVO / EC number: DNA-(apurinic or apyrimidinic site) lyase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 86.260859 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SVQDRVKRPM NAFIVWSRDQ RRKMALENPR MRNSEISKQL GYQWKMLTEA EKWPFFQEAQ KLQAMHREKY PNYKYRKGGS SAGAIVPVT PVKKPRPRPK VDLDDETDRV WKLLLENINS EGVDGSDEQK AKWWEEERNV FRGRADSFIA RMHLVQGDRR F TPWKGSVV ...String: SVQDRVKRPM NAFIVWSRDQ RRKMALENPR MRNSEISKQL GYQWKMLTEA EKWPFFQEAQ KLQAMHREKY PNYKYRKGGS SAGAIVPVT PVKKPRPRPK VDLDDETDRV WKLLLENINS EGVDGSDEQK AKWWEEERNV FRGRADSFIA RMHLVQGDRR F TPWKGSVV DSVVGVFLTQ NVSDHLSSSA FMSLASQFPV PFVPSSNFDA GTSSMPSIQI TYLDSEETMS SPPDHNHSCQ KP TLKEKGK KVLKEEKKAF DWDCLRREAQ ARAGIREKTR STMDTVDWKA IRAADVKEVA ETIKSRGMNH KLAERIQGFL DRL VNDHGS IDLEWLRDVP PDKAKEYLLS FNGLGLKSVE CVRLLTLHHL AFPVNTNVGR IAVRLGWVPL QPLPESLQLH LLEM YPMLE SIQKYLWPRL CKLDQKTLYE LHYQMITFGK VFCTKSKPNC NACPMKGECR HFASAFASAR LALPSTEKTC CEPII EEPA SPEPETAEVS IADIEEAFFE DPEEIPTIRL NMDAFTSNLK KIMEHNKELQ DGNMSSALVA LTAETASLPM PKLKNI SQL RTEHRVYELP DEHPLLAQLE KREPDDPCSY LLAIWTPGET ADSIQPSVST CIFQANGMLC DEETCFSCNS IKETRSQ IV RGTILIPCRT AMRGSFPLNG TYFQVNEVFA DHASSLNPIN VPRELIWELP RRTVYFGTSV PTIFKGLSTE KIQACFWK G YVCVRGFDRK TRGPKPLIAR LHFPASKLKG QQANLA UniProtKB: Sex-determining region Y protein, DNA glycosylase/AP lyase ROS1, DNA glycosylase/AP lyase ROS1, DNA glycosylase/AP lyase ROS1 |
-Macromolecule #2: DNA (40-MER)
| Macromolecule | Name: DNA (40-MER) / type: dna / ID: 2 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 12.39499 KDa |
| Sequence | String: (DT)(DA)(DG)(DC)(DT)(DG)(DA)(DG)(DT)(DG) (DA)(DT)(DC)(DC)(DG)(DG)(DA)(DG)(DT)(DC) (DT)(DG)(DA)(DA)(DA)(DC)(DA)(DA)(DT) (DG)(DC)(DA)(DT)(DC)(DA)(DC)(DA)(DG)(DA) (DG) |
-Macromolecule #3: DNA (40-MER)
| Macromolecule | Name: DNA (40-MER) / type: dna / ID: 3 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 12.244859 KDa |
| Sequence | String: (DC)(DT)(DC)(DT)(DG)(DT)(DG)(DA)(DT)(DG) (DC)(DA)(DT)(DT)(DG)(DT)(DT)(DT)(DC)(DA) (DG)(DA)(DC)(DT)(DC)(DT)(DG)(DG)(DA) (DT)(DC)(DA)(DC)(DT)(DC)(DA)(DG)(DC)(DT) (DA) |
-Macromolecule #4: IRON/SULFUR CLUSTER
| Macromolecule | Name: IRON/SULFUR CLUSTER / type: ligand / ID: 4 / Number of copies: 1 / Formula: SF4 |
|---|---|
| Molecular weight | Theoretical: 351.64 Da |
| Chemical component information |  ChemComp-FS1: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.7 mg/mL |
|---|---|
| Buffer | pH: 8 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average exposure time: 1.3467 sec. / Average electron dose: 1.2 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source: OTHER |
| Electron optics | Illumination mode: OTHER / Imaging mode: OTHER / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)