[English] 日本語
 Yorodumi
Yorodumi- EMDB-32324: Cryo-EM structure of SoxS-dependent transcription activation comp... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of SoxS-dependent transcription activation complex with fpr promoter DNA | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | bacterial RNA polymerase / TRANSCRIPTION-DNA COMPLEX | |||||||||
| Function / homology |  Function and homology information Function and homology informationbacterial-type RNA polymerase holo enzyme binding / sigma factor antagonist complex / RNA polymerase complex / submerged biofilm formation / cellular response to cell envelope stress / regulation of DNA-templated transcription initiation / sigma factor activity / bacterial-type flagellum assembly / bacterial-type RNA polymerase core enzyme binding / cytosolic DNA-directed RNA polymerase complex ...bacterial-type RNA polymerase holo enzyme binding / sigma factor antagonist complex / RNA polymerase complex / submerged biofilm formation / cellular response to cell envelope stress / regulation of DNA-templated transcription initiation / sigma factor activity / bacterial-type flagellum assembly / bacterial-type RNA polymerase core enzyme binding / cytosolic DNA-directed RNA polymerase complex / bacterial-type flagellum-dependent cell motility / nitrate assimilation / cis-regulatory region sequence-specific DNA binding / regulation of DNA-templated transcription elongation / transcription elongation factor complex / transcription antitermination / cell motility / DNA-templated transcription initiation / ribonucleoside binding / DNA-directed RNA polymerase / DNA-directed RNA polymerase activity / response to heat / protein-containing complex assembly / sequence-specific DNA binding / intracellular iron ion homeostasis / protein dimerization activity / DNA-binding transcription factor activity / response to antibiotic / negative regulation of DNA-templated transcription / regulation of DNA-templated transcription / magnesium ion binding / DNA binding / zinc ion binding / membrane / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |   | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.2 Å | |||||||||
 Authors Authors | Lin W / Feng Y | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nucleic Acids Res / Year: 2022 Journal: Nucleic Acids Res / Year: 2022Title: Structural basis of three different transcription activation strategies adopted by a single regulator SoxS. Authors: Jing Shi / Lu Wang / Aijia Wen / Fulin Wang / Yuqiong Zhang / Libing Yu / Fangfang Li / Yuanling Jin / Zhenzhen Feng / Jiacong Li / Yujiao Yang / Fei Gao / Yu Zhang / Yu Feng / Shuang Wang / ...Authors: Jing Shi / Lu Wang / Aijia Wen / Fulin Wang / Yuqiong Zhang / Libing Yu / Fangfang Li / Yuanling Jin / Zhenzhen Feng / Jiacong Li / Yujiao Yang / Fei Gao / Yu Zhang / Yu Feng / Shuang Wang / Wei Zhao / Wei Lin /  Abstract: Transcription activation is established through extensive protein-protein and protein-DNA interactions that allow an activator to engage and remodel RNA polymerase. SoxS, a global transcription ...Transcription activation is established through extensive protein-protein and protein-DNA interactions that allow an activator to engage and remodel RNA polymerase. SoxS, a global transcription activator, diversely regulates subsets of stress response genes with different promoters, but the detailed SoxS-dependent transcription initiation mechanisms remain obscure. Here, we report cryo-EM structures of three SoxS-dependent transcription activation complexes (SoxS-TACI, SoxS-TACII and SoxS-TACIII) comprising of Escherichia coli RNA polymerase (RNAP), SoxS protein and three representative classes of SoxS-regulated promoters. The structures reveal that SoxS monomer orchestrates transcription initiation through specific interactions with the promoter DNA and different conserved domains of RNAP. In particular, SoxS is positioned in the opposite orientation in SoxS-TACIII to that in SoxS-TACI and SoxS-TACII, unveiling a novel mode of transcription activation. Strikingly, two universally conserved C-terminal domains of alpha subunit (αCTD) of RNAP associate with each other, bridging SoxS and region 4 of σ70. We show that SoxS interacts with RNAP directly and independently from DNA, remodeling the enzyme to activate transcription from cognate SoxS promoters while repressing transcription from UP-element containing promoters. Our data provide a comprehensive summary of SoxS-dependent promoter architectures and offer new insights into the αCTD contribution to transcription control in bacteria. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_32324.map.gz emd_32324.map.gz | 37.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-32324-v30.xml emd-32324-v30.xml emd-32324.xml emd-32324.xml | 23.2 KB 23.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_32324.png emd_32324.png | 72.6 KB | ||
| Filedesc metadata |  emd-32324.cif.gz emd-32324.cif.gz | 8.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32324 http://ftp.pdbj.org/pub/emdb/structures/EMD-32324 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32324 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32324 | HTTPS FTP |
-Related structure data
| Related structure data |  7w5yMC  7w5wC  7w5xC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_32324.map.gz / Format: CCP4 / Size: 40.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_32324.map.gz / Format: CCP4 / Size: 40.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.307 Å | ||||||||||||||||||||||||||||||||||||
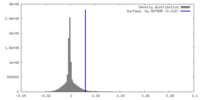
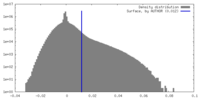
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
+Entire : SoxS-dependent transcription activation complex with fpr promoter DNA
+Supramolecule #1: SoxS-dependent transcription activation complex with fpr promoter DNA
+Supramolecule #2: DNA-directed RNA polymerase subunits
+Supramolecule #3: DNA
+Macromolecule #1: DNA-directed RNA polymerase subunit alpha
+Macromolecule #2: DNA-directed RNA polymerase subunit beta
+Macromolecule #3: DNA-directed RNA polymerase subunit beta'
+Macromolecule #4: DNA-directed RNA polymerase subunit omega
+Macromolecule #5: RNA polymerase sigma factor RpoD
+Macromolecule #8: Regulatory protein SoxS
+Macromolecule #6: fpr promoter DNA forward strand
+Macromolecule #7: fpr promoter DNA reverse strand
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 52.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.2 µm / Nominal defocus min: 1.4000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 4.2 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 74601 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)