[English] 日本語
 Yorodumi
Yorodumi- EMDB-31421: Cryo-EM structure of the GDP-bound dopamine receptor 1 and mini-G... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the GDP-bound dopamine receptor 1 and mini-Gs complex with Nb35 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | GPCR / dopamine receptor / mini-Gs / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationdopamine neurotransmitter receptor activity, coupled via Gs / dopamine neurotransmitter receptor activity / cerebral cortex GABAergic interneuron migration / Dopamine receptors / regulation of dopamine uptake involved in synaptic transmission / dopamine binding / operant conditioning / phospholipase C-activating dopamine receptor signaling pathway / heterotrimeric G-protein binding / peristalsis ...dopamine neurotransmitter receptor activity, coupled via Gs / dopamine neurotransmitter receptor activity / cerebral cortex GABAergic interneuron migration / Dopamine receptors / regulation of dopamine uptake involved in synaptic transmission / dopamine binding / operant conditioning / phospholipase C-activating dopamine receptor signaling pathway / heterotrimeric G-protein binding / peristalsis / G protein-coupled receptor complex / modification of postsynaptic structure / positive regulation of neuron migration / habituation / grooming behavior / regulation of dopamine metabolic process / astrocyte development / dopamine transport / sensitization / striatum development / dentate gyrus development / positive regulation of potassium ion transport / conditioned taste aversion / maternal behavior / arrestin family protein binding / non-motile cilium / long-term synaptic depression / mating behavior / G protein-coupled dopamine receptor signaling pathway / adult walking behavior / : / ciliary membrane / temperature homeostasis / dopamine metabolic process / adenylate cyclase-activating G protein-coupled bile acid receptor signaling pathway / adenylate cyclase-activating serotonin receptor signaling pathway / transmission of nerve impulse / regulation of skeletal muscle contraction / PKA activation in glucagon signalling / hair follicle placode formation / developmental growth / intracellular transport / G-protein alpha-subunit binding / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / D1 dopamine receptor binding / vascular endothelial cell response to laminar fluid shear stress / behavioral fear response / renal water homeostasis / activation of adenylate cyclase activity / neuronal action potential / Hedgehog 'off' state / cellular response to acidic pH / positive regulation of synaptic transmission, glutamatergic / adenylate cyclase-activating adrenergic receptor signaling pathway / behavioral response to cocaine / synapse assembly / cellular response to glucagon stimulus / intracellular glucose homeostasis / presynaptic modulation of chemical synaptic transmission / adenylate cyclase activator activity / positive regulation of release of sequestered calcium ion into cytosol / positive regulation of insulin secretion involved in cellular response to glucose stimulus / trans-Golgi network membrane / response to amphetamine / synaptic transmission, glutamatergic / negative regulation of inflammatory response to antigenic stimulus / response to prostaglandin E / bone development / visual learning / platelet aggregation / G protein-coupled receptor activity / GABA-ergic synapse / vasodilation / cognition / G-protein beta/gamma-subunit complex binding / memory / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G protein-coupled acetylcholine receptor signaling pathway / protein import into nucleus / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / positive regulation of insulin secretion / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / sensory perception of smell / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / long-term synaptic potentiation Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.13 Å | |||||||||
 Authors Authors | Xiao T / Zheng S | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2022 Journal: Sci Adv / Year: 2022Title: Structural insights into G protein activation by D1 dopamine receptor. Authors: Xiao Teng / Sijia Chen / Qing Wang / Zhao Chen / Xiaoying Wang / Niu Huang / Sanduo Zheng /  Abstract: G protein-coupled receptors (GPCRs) comprise the largest family of membrane receptors and are the most important drug targets. An agonist-bound GPCR engages heterotrimeric G proteins and triggers the ...G protein-coupled receptors (GPCRs) comprise the largest family of membrane receptors and are the most important drug targets. An agonist-bound GPCR engages heterotrimeric G proteins and triggers the exchange of guanosine diphosphate (GDP) with guanosine triphosphate (GTP) to promote G protein activation. A complete understanding of molecular mechanisms of G protein activation has been hindered by a lack of structural information of GPCR-G protein complex in nucleotide-bound states. Here, we report the cryo-EM structures of the D1 dopamine receptor and mini-G complex in the nucleotide-free and nucleotide-bound states. These structures reveal major conformational changes in Gα such as structural rearrangements of the carboxyl- and amino-terminal α helices that account for the release of GDP and the GTP-dependent dissociation of Gα from Gβγ subunits. As validated by biochemical and cellular signaling studies, our structures shed light into the molecular basis of the entire signaling events of GPCR-mediated G protein activation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_31421.map.gz emd_31421.map.gz | 20.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-31421-v30.xml emd-31421-v30.xml emd-31421.xml emd-31421.xml | 20.4 KB 20.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_31421.png emd_31421.png | 23.9 KB | ||
| Filedesc metadata |  emd-31421.cif.gz emd-31421.cif.gz | 7.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-31421 http://ftp.pdbj.org/pub/emdb/structures/EMD-31421 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31421 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31421 | HTTPS FTP |
-Related structure data
| Related structure data |  7f1oMC  7f0tC  7f1zC  7f23C  7f24C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_31421.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_31421.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.087 Å | ||||||||||||||||||||||||||||||||||||
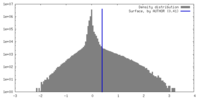
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Cryo-EM structure of the GDP-bound dopamine receptor 1 and mini-G...
| Entire | Name: Cryo-EM structure of the GDP-bound dopamine receptor 1 and mini-Gs complex with Nb35 |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of the GDP-bound dopamine receptor 1 and mini-G...
| Supramolecule | Name: Cryo-EM structure of the GDP-bound dopamine receptor 1 and mini-Gs complex with Nb35 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: D(1A) dopamine receptor
| Macromolecule | Name: D(1A) dopamine receptor / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 52.409656 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDASIDMRT LNTSAMDGTG LVVERDFSVR ILTACFLSLL ILSTLLGNTL VCAAVIRFRH LRSKVTNFF VISLAVSDLL VAVLVMPWKA VAEIAGFWPF GSFCNIWVAF DIMCSTASIL NLCVISVDRY WAISSPFRYE R KMTPKAAF ...String: MKTIIALSYI FCLVFADYKD DDDASIDMRT LNTSAMDGTG LVVERDFSVR ILTACFLSLL ILSTLLGNTL VCAAVIRFRH LRSKVTNFF VISLAVSDLL VAVLVMPWKA VAEIAGFWPF GSFCNIWVAF DIMCSTASIL NLCVISVDRY WAISSPFRYE R KMTPKAAF ILISVAWTLS VLISFIPVQL SWHKAKPTSP SDGNATSLAE TIDNCDSSLS RTYAISSSVI SFYIPVAIMI VT YTRIYRI AQKQIRRIAA LERAAVHAKN CQTTTGNGKP VECSQPESSF KMSFKRETKV LKTLSVIMGV FVCCWLPFFI LNC ILPFCG SGETQPFCID SNTFDVFVWF GWANSSLNPI IYAFNADFRK AFSTLLGCYR LCPATNNAIE TVSINNNGAA MFSS HHEPR GSISKECNLV YLIPHAVGSS EDLKKEEAAG IARPLEKLSP ALSVILDYDT DVSLEKIQPI TQNGQHPT UniProtKB: D(1A) dopamine receptor |
-Macromolecule #2: Guanine nucleotide-binding protein G(s) subunit alpha isoforms sh...
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short,Isoform Gnas-2 of Guanine nucleotide-binding protein G(s) subunit alpha isoforms short type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 28.907684 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: NSKTEDQRNE EKAQREANKK IEKQLQKDKQ VYRATHRLLL LGADNSGKST IVKQMRILHG GSGGSGGTSG IFETKFQVDK VNFHMFDVG GQRDERRKWI QCFNDVTAII FVVDSSDYNR LQEALNLFKS IWNNRWLRTI SVILFLNKQD LLAEKVLAGK S KIEDYFPE ...String: NSKTEDQRNE EKAQREANKK IEKQLQKDKQ VYRATHRLLL LGADNSGKST IVKQMRILHG GSGGSGGTSG IFETKFQVDK VNFHMFDVG GQRDERRKWI QCFNDVTAII FVVDSSDYNR LQEALNLFKS IWNNRWLRTI SVILFLNKQD LLAEKVLAGK S KIEDYFPE FARYTTPEDA TPEPGEDPRV TRAKYFIRDE FLRISTASGD GRHYCYPHFT CAVDTENARR IFNDCRDIIQ RM HLRQYEL L UniProtKB: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short, Guanine nucleotide-binding protein G(s) subunit alpha isoforms short |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.286891 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: HHHHHHLEVL FQGPGSSGSE LDQLRQEAEQ LKNQIRDARK ACADATLSQI TNNIDPVGRI QMRTRRTLRG HLAKIYAMHW GTDSRLLVS ASQDGKLIIW DSYTTNKVHA IPLRSSWVMT CAYAPSGNYV ACGGLDNICS IYNLKTREGN VRVSRELAGH T GYLSCCRF ...String: HHHHHHLEVL FQGPGSSGSE LDQLRQEAEQ LKNQIRDARK ACADATLSQI TNNIDPVGRI QMRTRRTLRG HLAKIYAMHW GTDSRLLVS ASQDGKLIIW DSYTTNKVHA IPLRSSWVMT CAYAPSGNYV ACGGLDNICS IYNLKTREGN VRVSRELAGH T GYLSCCRF LDDNQIVTSS GDTTCALWDI ETGQQTTTFT GHTGDVMSLS LAPDTRLFVS GACDASAKLW DVREGMCRQT FT GHESDIN AICFFPNGNA FATGSDDATC RLFDLRADQE LMTYSHDNII CGITSVSFSK SGRLLLAGYD DFNCNVWDAL KAD RAGVLA GHDNRVSCLG VTDDGMAVAT GSWDSFLKIW N UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.845078 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFSAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #5: Nanobody 35
| Macromolecule | Name: Nanobody 35 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 17.352498 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKYLLPTAAA GLLLLAAQPA MAQVQLQESG GGLVQPGGSL RLSCAASGFT FSNYKMNWVR QAPGKGLEWV SDISQSGASI SYTGSVKGR FTISRDNAKN TLYLQMNSLK PEDTAVYYCA RCPAPFTRDC FDVTSTTYAY RGQGTQVTVS SHHHHHHEPE A |
-Macromolecule #6: L-DOPAMINE
| Macromolecule | Name: L-DOPAMINE / type: ligand / ID: 6 / Number of copies: 1 / Formula: LDP |
|---|---|
| Molecular weight | Theoretical: 153.178 Da |
| Chemical component information |  ChemComp-LDP: |
-Macromolecule #7: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 7 / Number of copies: 1 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #8: GUANOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: GUANOSINE-5'-DIPHOSPHATE / type: ligand / ID: 8 / Number of copies: 1 / Formula: GDP |
|---|---|
| Molecular weight | Theoretical: 443.201 Da |
| Chemical component information |  ChemComp-GDP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 4.0 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 35 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 281 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number real images: 601 / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal magnification: 64000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)