登録情報 データベース : EMDB / ID : EMD-30566タイトル Human SECR in complex with an engineered Gs heterotrimer 複合体 : SECR-Gs complex複合体 : Secretin複合体 : Secretin receptorタンパク質・ペプチド : Secretin receptor複合体 : Guanine nucleotide-binding protein G(s) subunit alpha isoforms shortタンパク質・ペプチド : Guanine nucleotide-binding protein G(s) subunit alpha isoforms short複合体 : Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1タンパク質・ペプチド : Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1複合体 : Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2タンパク質・ペプチド : Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2複合体 : nanobody Nb35タンパク質・ペプチド : nanobody Nb35機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト) / Rattus norvegicus (ドブネズミ) / Bos taurus (ウシ) / unidentified (未定義) 手法 / / 解像度 : 2.9 Å Fukuhara S / Kobayashi K / Kusakizako T / Shihoya W / Nureki O ジャーナル : Biochem Biophys Res Commun / 年 : 2020タイトル : Structure of the human secretin receptor coupled to an engineered heterotrimeric G protein.著者 : Satoshi Fukuhara / Kazuhiro Kobayashi / Tsukasa Kusakizako / Wataru Iida / Masahiko Kato / Wataru Shihoya / Osamu Nureki / 要旨 : Secretin is a gastrointestinal hormone that exerts multiple physiological functions via activation of the secretin receptor (SECR). SECR belongs to the class B G-protein-coupled receptors and is ... Secretin is a gastrointestinal hormone that exerts multiple physiological functions via activation of the secretin receptor (SECR). SECR belongs to the class B G-protein-coupled receptors and is involved in various processes, such as regulation of the pH of the duodenal content, food intake, and water homeostasis. Here, we report a cryo-electron microscopy structure of human SECR bound to secretin and an engineered Gs heterotrimer. The structure revealed the basic architecture of SECR and the secretin binding mode. A structural comparison of the SECR and PAC1R transmembrane domains revealed that transmembrane helices 1 and 2 play a prominent role in secretin recognition. Moreover, the extracellular domain of SECR is perpendicular to the TMD, unlike that of PAC1R. This comparison revealed the diverged peptide recognition mechanisms of these receptors, which belong to the same subgroup. Our structural information will facilitate drug discovery research for clinical applications. 履歴 登録 2020年9月20日 - ヘッダ(付随情報) 公開 2020年11月4日 - マップ公開 2020年11月4日 - 更新 2020年12月30日 - 現状 2020年12月30日 処理サイト : PDBj / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト) /
Homo sapiens (ヒト) / 

 データ登録者
データ登録者 引用
引用 ジャーナル: Biochem Biophys Res Commun / 年: 2020
ジャーナル: Biochem Biophys Res Commun / 年: 2020
 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_30566.map.gz
emd_30566.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-30566-v30.xml
emd-30566-v30.xml emd-30566.xml
emd-30566.xml EMDBヘッダ
EMDBヘッダ emd_30566_fsc.xml
emd_30566_fsc.xml FSCデータファイル
FSCデータファイル emd_30566.png
emd_30566.png emd_30566_half_map_1.map.gz
emd_30566_half_map_1.map.gz emd_30566_half_map_2.map.gz
emd_30566_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-30566
http://ftp.pdbj.org/pub/emdb/structures/EMD-30566 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30566
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30566 emd_30566_validation.pdf.gz
emd_30566_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_30566_full_validation.pdf.gz
emd_30566_full_validation.pdf.gz emd_30566_validation.xml.gz
emd_30566_validation.xml.gz emd_30566_validation.cif.gz
emd_30566_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30566
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30566 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30566
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30566 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_30566.map.gz / 形式: CCP4 / 大きさ: 35.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_30566.map.gz / 形式: CCP4 / 大きさ: 35.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 画像解析
画像解析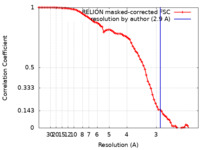
 ムービー
ムービー コントローラー
コントローラー




























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)