+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | CLC-ec1 E202Y at pH 4.5 100mM Cl Turn | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | CLC-ec1 / ecCLC / eriC / CLC transporter / chloride proton antiporter / TRANSPORT PROTEIN | |||||||||
| 機能・相同性 | Chloride channel, ClcA / Chloride channel, voltage gated / Chloride channel, core / Voltage gated chloride channel / voltage-gated chloride channel activity / antiporter activity / plasma membrane / H(+)/Cl(-) exchange transporter ClcA 機能・相同性情報 機能・相同性情報 | |||||||||
| 生物種 |  | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.5 Å | |||||||||
 データ登録者 データ登録者 | Fortea E / Lee S / Argyos Y / Chadda R / Ciftci D / Huysmans G / Robertson JL / Boudker O / Accardi A | |||||||||
| 資金援助 |  米国, 1件 米国, 1件
| |||||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2024 ジャーナル: Nat Struct Mol Biol / 年: 2024タイトル: Structural basis of pH-dependent activation in a CLC transporter. 著者: Eva Fortea / Sangyun Lee / Rahul Chadda / Yiorgos Argyros / Priyanka Sandal / Robyn Mahoney-Kruszka / Hatice Didar Ciftci / Maria E Falzone / Gerard Huysmans / Janice L Robertson / Olga ...著者: Eva Fortea / Sangyun Lee / Rahul Chadda / Yiorgos Argyros / Priyanka Sandal / Robyn Mahoney-Kruszka / Hatice Didar Ciftci / Maria E Falzone / Gerard Huysmans / Janice L Robertson / Olga Boudker / Alessio Accardi /   要旨: CLCs are dimeric chloride channels and anion/proton exchangers that regulate processes such as muscle contraction and endo-lysosome acidification. Common gating controls their activity; its closure ...CLCs are dimeric chloride channels and anion/proton exchangers that regulate processes such as muscle contraction and endo-lysosome acidification. Common gating controls their activity; its closure simultaneously silences both protomers, and its opening allows them to independently transport ions. Mutations affecting common gating in human CLCs cause dominant genetic disorders. The structural rearrangements underlying common gating are unknown. Here, using single-particle cryo-electron microscopy, we show that the prototypical Escherichia coli CLC-ec1 undergoes large-scale rearrangements in activating conditions. The slow, pH-dependent remodeling of the dimer interface leads to the concerted opening of the intracellular H pathways and is required for transport. The more frequent formation of short water wires in the open H pathway enables Cl pore openings. Mutations at disease-causing sites favor CLC-ec1 activation and accelerate common gate opening in the human CLC-7 exchanger. We suggest that the pH activation mechanism of CLC-ec1 is related to the common gating of CLC-7. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
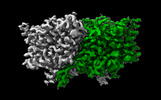
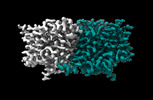
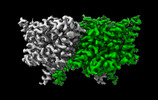
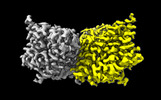
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_29883.map.gz emd_29883.map.gz | 2 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-29883-v30.xml emd-29883-v30.xml emd-29883.xml emd-29883.xml | 13.9 KB 13.9 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_29883.png emd_29883.png | 82.8 KB | ||
| Filedesc metadata |  emd-29883.cif.gz emd-29883.cif.gz | 5.3 KB | ||
| その他 |  emd_29883_half_map_1.map.gz emd_29883_half_map_1.map.gz emd_29883_half_map_2.map.gz emd_29883_half_map_2.map.gz | 26.7 MB 26.7 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29883 http://ftp.pdbj.org/pub/emdb/structures/EMD-29883 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29883 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29883 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_29883_validation.pdf.gz emd_29883_validation.pdf.gz | 761.9 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_29883_full_validation.pdf.gz emd_29883_full_validation.pdf.gz | 761.5 KB | 表示 | |
| XML形式データ |  emd_29883_validation.xml.gz emd_29883_validation.xml.gz | 10.7 KB | 表示 | |
| CIF形式データ |  emd_29883_validation.cif.gz emd_29883_validation.cif.gz | 12.4 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29883 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29883 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29883 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29883 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_29883.map.gz / 形式: CCP4 / 大きさ: 28.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_29883.map.gz / 形式: CCP4 / 大きさ: 28.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ | X=Y=Z: 1.076 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: #1
| ファイル | emd_29883_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
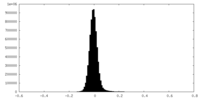
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_29883_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
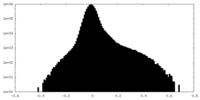
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Structure of ecCLC-E202Y mutant at pH 4.5 in 100mM Cl
| 全体 | 名称: Structure of ecCLC-E202Y mutant at pH 4.5 in 100mM Cl |
|---|---|
| 要素 |
|
-超分子 #1: Structure of ecCLC-E202Y mutant at pH 4.5 in 100mM Cl
| 超分子 | 名称: Structure of ecCLC-E202Y mutant at pH 4.5 in 100mM Cl タイプ: cell / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: H(+)/Cl(-) exchange transporter ClcA
| 分子 | 名称: H(+)/Cl(-) exchange transporter ClcA / タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 50.424461 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MKTDTPSLET PQAARLRRRQ LIRQLLERDK TPLAILFMAA VVGTLVGLAA VAFDKGVAWL QNQRMGALVH TADNYPLLLT VAFLCSAVL AMFGYFLVRK YAPEAGGSGI PEIEGALEDQ RPVRWWRVLP VKFFGGLGTL GGGMVLGREG PTVQIGGNIG R MVLDIFRL ...文字列: MKTDTPSLET PQAARLRRRQ LIRQLLERDK TPLAILFMAA VVGTLVGLAA VAFDKGVAWL QNQRMGALVH TADNYPLLLT VAFLCSAVL AMFGYFLVRK YAPEAGGSGI PEIEGALEDQ RPVRWWRVLP VKFFGGLGTL GGGMVLGREG PTVQIGGNIG R MVLDIFRL KGDEARHTLL ATGAAAGLAA AFNAPLAGIL FIIYEMRPQF RYTLISIKAV FIGVIMSTIM YRIFNHEVAL ID VGKLSDA PLNTLWLYLI LGIIFGIFGP IFNKWVLGMQ DLLHRVHGGN ITKWVLMGGA IGGLCGLLGF VAPATSGGGF NLI PIATAG NFSMGMLVFI FVARVITTLL CFSSGAPGGI FAPMLALGTV LGTAFGMVAV ELFPQYHLEA GTFAIAGMGA LLAA SIRAP LTGIILVLEM TDNYQLILPM IITGLGATLL AQFTGGKPLY SAILARTLAK QEAEQLARSK AASASENT UniProtKB: H(+)/Cl(-) exchange transporter ClcA |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 4.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 平均電子線量: 51.791 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.0 µm 最小 デフォーカス(公称値): 0.7000000000000001 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 初期モデル | モデルのタイプ: NONE |
|---|---|
| 最終 再構成 | 想定した対称性 - 点群: C1 (非対称) / 解像度のタイプ: BY AUTHOR / 解像度: 3.5 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 使用した粒子像数: 71031 |
| 初期 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
 ムービー
ムービー コントローラー
コントローラー








 Z
Z Y
Y X
X