+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Nucleocapsid monomer structure from SARS-CoV-2 | ||||||||||||
 Map data Map data | Nucleocapsid monomer structure from SARS-CoV-2 | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | SARS-CoV-2 / N protein / COVID-19 / RNA binding protein / VIRAL PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationcytoplasmic capsid assembly / response to host immune response / viral RNA genome packaging / negative regulation of interferon-beta production / Maturation of nucleoprotein / intracellular non-membrane-bounded organelle / positive regulation of NLRP3 inflammasome complex assembly / MHC class I protein binding / CD28 dependent PI3K/Akt signaling / SARS-CoV-2 targets host intracellular signalling and regulatory pathways ...cytoplasmic capsid assembly / response to host immune response / viral RNA genome packaging / negative regulation of interferon-beta production / Maturation of nucleoprotein / intracellular non-membrane-bounded organelle / positive regulation of NLRP3 inflammasome complex assembly / MHC class I protein binding / CD28 dependent PI3K/Akt signaling / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / protein sequestering activity / VEGFR2 mediated vascular permeability / molecular condensate scaffold activity / NOD1/2 Signaling Pathway / TAK1-dependent IKK and NF-kappa-B activation / DDX58/IFIH1-mediated induction of interferon-alpha/beta / MHC class I protein complex / Interleukin-1 signaling / RNA stem-loop binding / Interferon alpha/beta signaling / PIP3 activates AKT signaling / viral capsid / Transcription of SARS-CoV-2 sgRNAs / host cell endoplasmic reticulum-Golgi intermediate compartment / viral nucleocapsid / host cell Golgi apparatus / Translation of Structural Proteins / Virion Assembly and Release / host extracellular space / Induction of Cell-Cell Fusion / Attachment and Entry / host cell perinuclear region of cytoplasm / ribonucleoprotein complex / SARS-CoV-2 activates/modulates innate and adaptive immune responses / protein homodimerization activity / RNA binding / extracellular region / identical protein binding / cytoplasm Similarity search - Function | ||||||||||||
| Biological species |  | ||||||||||||
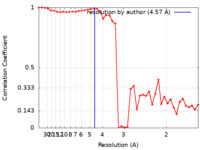
| Method | single particle reconstruction / cryo EM / Resolution: 4.57 Å | ||||||||||||
 Authors Authors | Casasanta M / Jonaid GM / Kaylor L / Luqiu W / DiCecco L / Solares M / Berry S / Kelly DF | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation | Journal: Nanoscale / Year: 2021 Title: Microchip-based structure determination of low-molecular weight proteins using cryo-electron microscopy. Authors: Michael A Casasanta / G M Jonaid / Liam Kaylor / William Y Luqiu / Maria J Solares / Mariah L Schroen / William J Dearnaley / Jarad Wilson / Madeline J Dukes / Deborah F Kelly /  Abstract: Interest in cryo-Electron Microscopy (EM) imaging has skyrocketed in recent years due to its pristine views of macromolecules and materials. As advances in instrumentation and computing algorithms ...Interest in cryo-Electron Microscopy (EM) imaging has skyrocketed in recent years due to its pristine views of macromolecules and materials. As advances in instrumentation and computing algorithms spurred this progress, there is renewed focus to address specimen-related challenges. Here we contribute a microchip-based toolkit to perform complementary structural and biochemical analysis on low-molecular weight proteins. As a model system, we used the SARS-CoV-2 nucleocapsid (N) protein (48 kDa) due to its stability and important role in therapeutic development. Cryo-EM structures of the N protein monomer revealed a flexible N-terminal "top hat" motif and a helical-rich C-terminal domain. To complement our structural findings, we engineered microchip-based immunoprecipitation assays that led to the discovery of the first antibody binding site on the N protein. The data also facilitated molecular modeling of a variety of pandemic and common cold-related coronavirus proteins. Such insights may guide future pandemic-preparedness protocols through immuno-engineering strategies to mitigate viral outbreaks. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29002.map.gz emd_29002.map.gz | 3.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29002-v30.xml emd-29002-v30.xml emd-29002.xml emd-29002.xml | 19.2 KB 19.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_29002_fsc.xml emd_29002_fsc.xml | 4.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_29002.png emd_29002.png | 187.7 KB | ||
| Filedesc metadata |  emd-29002.cif.gz emd-29002.cif.gz | 6.4 KB | ||
| Others |  emd_29002_half_map_1.map.gz emd_29002_half_map_1.map.gz emd_29002_half_map_2.map.gz emd_29002_half_map_2.map.gz | 3.5 MB 3.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29002 http://ftp.pdbj.org/pub/emdb/structures/EMD-29002 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29002 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29002 | HTTPS FTP |
-Validation report
| Summary document |  emd_29002_validation.pdf.gz emd_29002_validation.pdf.gz | 690.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_29002_full_validation.pdf.gz emd_29002_full_validation.pdf.gz | 690 KB | Display | |
| Data in XML |  emd_29002_validation.xml.gz emd_29002_validation.xml.gz | 8.7 KB | Display | |
| Data in CIF |  emd_29002_validation.cif.gz emd_29002_validation.cif.gz | 11.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29002 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29002 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29002 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29002 | HTTPS FTP |
-Related structure data
| Related structure data |  8fd5MC  8fg2C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_29002.map.gz / Format: CCP4 / Size: 4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29002.map.gz / Format: CCP4 / Size: 4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Nucleocapsid monomer structure from SARS-CoV-2 | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.8 Å | ||||||||||||||||||||||||||||||||||||
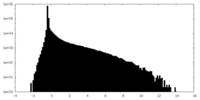
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half Map 1
| File | emd_29002_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Map 2
| File | emd_29002_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : A chain contains the full-length N protein
| Entire | Name: A chain contains the full-length N protein |
|---|---|
| Components |
|
-Supramolecule #1: A chain contains the full-length N protein
| Supramolecule | Name: A chain contains the full-length N protein / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Residues 1-49 were fit into the map separately from residues 50 - 419. The 2 fragments were fit as individual rigid bodies, then refined and rebuilt. There is a missing peptide linkage ...Details: Residues 1-49 were fit into the map separately from residues 50 - 419. The 2 fragments were fit as individual rigid bodies, then refined and rebuilt. There is a missing peptide linkage between residues 49 and 50. |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Nucleoprotein
| Macromolecule | Name: Nucleoprotein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 45.689645 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSDNGPQNQR NAPRITFGGP SDSTGSNQNG ERSGARSKQR RPQGLPNNTA SWFTALTQHG KEDLKFPRGQ GVPINTNSSP DDQIGYYRR ATRRIRGGDG KMKDLSPRWY FYYLGTGPEA GLPYGANKDG IIWVATEGAL NTPKDHIGTR NPANNAAIVL Q LPQGTTLP ...String: MSDNGPQNQR NAPRITFGGP SDSTGSNQNG ERSGARSKQR RPQGLPNNTA SWFTALTQHG KEDLKFPRGQ GVPINTNSSP DDQIGYYRR ATRRIRGGDG KMKDLSPRWY FYYLGTGPEA GLPYGANKDG IIWVATEGAL NTPKDHIGTR NPANNAAIVL Q LPQGTTLP KGFYAEGSRG GSQASSRSSS RSRNSSRNST PGSSRGTSPA RMAGNGGDAA LALLLLDRLN QLESKMSGKG QQ QQGQTVT KKSAAEASKK PRQKRTATKA YNVTQAFGRR GPEQTQGNFG DQELIRQGTD YKHWPQIAQF APSASAFFGM SRI GMEVTP SGTWLTYTGA IKLDDKDPNF KDQVILLNKH IDAYKTFPPT EPKKDKKKKA DETQALPQRQ KKQQTVTLLP AADL DDFSK QLQQSMSSAD STQA UniProtKB: Nucleoprotein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.1 mg/mL |
|---|---|
| Buffer | pH: 7.5 Details: 20 mM Tris (pH 7.5), 150 mM NaCl, 10 mM MgCl2, 10 mM CaCl2 |
| Grid | Model: Homemade / Material: SILICON NITRIDE Details: Protein samples were added to the Ni-NTA-coated microchips and incubated for 1 minute at room temperature prior to vitrification. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK III Details: A Mark III Vitrobot was used to plunge samples into liquid ethane, operating at room temperature and 100% humidity with 3 - 4 seconds blot time. |
| Details | Sample was enriched using Ni-NTA coated silicon nitride microchips |
- Electron microscopy
Electron microscopy
| Microscope | TFS TALOS F200C |
|---|---|
| Image recording | Film or detector model: DIRECT ELECTRON DE-12 (4k x 3k) / Detector mode: INTEGRATING / Number grids imaged: 10 / Number real images: 200 / Average exposure time: 1.0 sec. / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 5.0 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 45000 |
| Sample stage | Specimen holder model: GATAN 626 SINGLE TILT LIQUID NITROGEN CRYO TRANSFER HOLDER Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: RIGID BODY FIT / Overall B value: 100 |
|---|---|
| Output model |  PDB-8fd5: |
 Movie
Movie Controller
Controller









 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)