+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | MthK-A90L mutant in closed state with 0 Ca2+ | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | MTHK / TRANSPORT PROTEIN / ION CHANNEL | |||||||||
| Function / homology |  Function and homology information Function and homology informationmonoatomic cation transmembrane transporter activity / potassium ion transport / metal ion binding / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |   Methanothermobacter thermautotrophicus (archaea) Methanothermobacter thermautotrophicus (archaea) | |||||||||
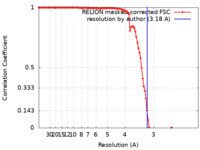
| Method | single particle reconstruction / cryo EM / Resolution: 3.18 Å | |||||||||
 Authors Authors | Agarwal S / Nimigean CM | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Chem Biol / Year: 2024 Journal: Nat Chem Biol / Year: 2024Title: Calcium-gated potassium channel blockade via membrane-facing fenestrations. Authors: Chen Fan / Emelie Flood / Nattakan Sukomon / Shubhangi Agarwal / Toby W Allen / Crina M Nimigean /    Abstract: Quaternary ammonium blockers were previously shown to bind in the pore to block both open and closed conformations of large-conductance calcium-activated potassium (BK and MthK) channels. Because ...Quaternary ammonium blockers were previously shown to bind in the pore to block both open and closed conformations of large-conductance calcium-activated potassium (BK and MthK) channels. Because blocker entry was assumed through the intracellular entryway (bundle crossing), closed-pore access suggested that the gate was not at the bundle crossing. Structures of closed MthK, a Methanobacterium thermoautotrophicum homolog of BK channels, revealed a tightly constricted intracellular gate, leading us to investigate the membrane-facing fenestrations as alternative pathways for blocker access directly from the membrane. Atomistic free energy simulations showed that intracellular blockers indeed access the pore through the fenestrations, and a mutant channel with narrower fenestrations displayed no closed-state TPeA block at concentrations that blocked the wild-type channel. Apo BK channels display similar fenestrations, suggesting that blockers may use them as access paths into closed channels. Thus, membrane fenestrations represent a non-canonical pathway for selective targeting of specific channel conformations, opening novel ways to selectively drug BK channels. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27459.map.gz emd_27459.map.gz | 8.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27459-v30.xml emd-27459-v30.xml emd-27459.xml emd-27459.xml | 19.3 KB 19.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_27459_fsc.xml emd_27459_fsc.xml | 10.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_27459.png emd_27459.png | 45.4 KB | ||
| Filedesc metadata |  emd-27459.cif.gz emd-27459.cif.gz | 6.6 KB | ||
| Others |  emd_27459_half_map_1.map.gz emd_27459_half_map_1.map.gz emd_27459_half_map_2.map.gz emd_27459_half_map_2.map.gz | 76.2 MB 76.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27459 http://ftp.pdbj.org/pub/emdb/structures/EMD-27459 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27459 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27459 | HTTPS FTP |
-Related structure data
| Related structure data |  8djbMC  9405C  9406C  9407C  5bkiC  5bkjC  5bkkC  8fz7C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_27459.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27459.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.083 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_27459_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_27459_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : MthK A90L mutant in 0 Ca2+
| Entire | Name: MthK A90L mutant in 0 Ca2+ |
|---|---|
| Components |
|
-Supramolecule #1: MthK A90L mutant in 0 Ca2+
| Supramolecule | Name: MthK A90L mutant in 0 Ca2+ / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:   Methanothermobacter thermautotrophicus (archaea) Methanothermobacter thermautotrophicus (archaea) |
| Molecular weight | Theoretical: 220 KDa |
-Macromolecule #1: Calcium-gated potassium channel MthK
| Macromolecule | Name: Calcium-gated potassium channel MthK / type: protein_or_peptide / ID: 1 / Number of copies: 8 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Methanothermobacter thermautotrophicus (archaea) Methanothermobacter thermautotrophicus (archaea) |
| Molecular weight | Theoretical: 37.398242 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MVLVIEIIRK HLPRVLKVPA TRILLLVLAV IIYGTAGFHF IEGESWTVSL YWTFVTIATV GYGDYSPSTP LGMYFTVTLI VLGIGTFAV LVERLLEFLI NREQMKLMGL IDVAKSRHVV ICGWSESTLE CLRELRGSEV FVLAEDENVR KKVLRSGANF V HGDPTRVS ...String: MVLVIEIIRK HLPRVLKVPA TRILLLVLAV IIYGTAGFHF IEGESWTVSL YWTFVTIATV GYGDYSPSTP LGMYFTVTLI VLGIGTFAV LVERLLEFLI NREQMKLMGL IDVAKSRHVV ICGWSESTLE CLRELRGSEV FVLAEDENVR KKVLRSGANF V HGDPTRVS DLEKANVRGA RAVIVDLESD SETIHCILGI RKIDESVRII AEAERYENIE QLRMAGADQV ISPFVISGRL MS RSIDDGY EAMFVQDVLA EESTRRMVEV PIPEGSKLEG VSVLDADIHD VTGVIIIGVG RGDELIIDPP RDYSFRAGDI ILG IGKPEE IERLKNYISA UniProtKB: Calcium-gated potassium channel MthK |
-Macromolecule #2: POTASSIUM ION
| Macromolecule | Name: POTASSIUM ION / type: ligand / ID: 2 / Number of copies: 1 / Formula: K |
|---|---|
| Molecular weight | Theoretical: 39.098 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 7 mg/mL |
|---|---|
| Buffer | pH: 8.5 |
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 80 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK IV / Details: BLOTTING FOR 2 S (BLOT FORCE 1). |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 65.19 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)