+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
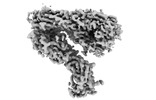
| Title | Integrin alphaM/beta2 ectodomain | |||||||||
 Map data Map data | Composite map of sharpened global and local reconstructions | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Integrin / Complement / Receptor / CELL ADHESION | |||||||||
| Function / homology |  Function and homology information Function and homology informationectodermal cell differentiation / positive regulation of neutrophil degranulation / cellular extravasation / integrin alphaM-beta2 complex / response to Gram-positive bacterium / response to curcumin / positive regulation of microglial cell mediated cytotoxicity / cell-cell adhesion via plasma-membrane adhesion molecules / complement component C3b binding / vertebrate eye-specific patterning ...ectodermal cell differentiation / positive regulation of neutrophil degranulation / cellular extravasation / integrin alphaM-beta2 complex / response to Gram-positive bacterium / response to curcumin / positive regulation of microglial cell mediated cytotoxicity / cell-cell adhesion via plasma-membrane adhesion molecules / complement component C3b binding / vertebrate eye-specific patterning / complement-mediated synapse pruning / Toll Like Receptor 4 (TLR4) Cascade / leukocyte migration involved in inflammatory response / complement receptor mediated signaling pathway / integrin complex / heterotypic cell-cell adhesion / cargo receptor activity / leukocyte cell-cell adhesion / phagocytosis, engulfment / negative regulation of dopamine metabolic process / cell adhesion mediated by integrin / forebrain development / amyloid-beta clearance / tertiary granule membrane / plasma membrane raft / positive regulation of protein targeting to membrane / Integrin cell surface interactions / response to mechanical stimulus / phagocytosis / endothelial cell migration / specific granule membrane / cell adhesion molecule binding / positive regulation of superoxide anion generation / heat shock protein binding / receptor-mediated endocytosis / response to ischemia / cell-matrix adhesion / integrin-mediated signaling pathway / Cell surface interactions at the vascular wall / microglial cell activation / cell-cell adhesion / integrin binding / positive regulation of angiogenesis / positive regulation of nitric oxide biosynthetic process / response to estradiol / amyloid-beta binding / Interleukin-4 and Interleukin-13 signaling / cell adhesion / membrane raft / innate immune response / external side of plasma membrane / Neutrophil degranulation / protein-containing complex binding / cell surface / extracellular space / extracellular exosome / metal ion binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.7 Å | |||||||||
 Authors Authors | Goldsmith JA / McLellan JS | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2022 Journal: Cell Rep / Year: 2022Title: Structural basis for non-canonical integrin engagement by Bordetella adenylate cyclase toxin. Authors: Jory A Goldsmith / Andrea M DiVenere / Jennifer A Maynard / Jason S McLellan /  Abstract: Integrins are ubiquitous cell-surface heterodimers that are exploited by pathogens and toxins, including leukotoxins that target β integrins on phagocytes. The Bordetella adenylate cyclase toxin ...Integrins are ubiquitous cell-surface heterodimers that are exploited by pathogens and toxins, including leukotoxins that target β integrins on phagocytes. The Bordetella adenylate cyclase toxin (ACT) uses the αβ integrin as a receptor, but the structural basis for integrin binding and neutralization by antibodies is poorly understood. Here, we use cryoelectron microscopy to determine a 2.7 Å resolution structure of an ACT fragment bound to αβ. This structure reveals that ACT interacts with the headpiece and calf-2 of the α subunit in a non-canonical manner specific to bent, inactive αβ. Neutralizing antibody epitopes map to ACT residues involved in α binding, providing the basis for antibody-mediated attachment inhibition. Furthermore, binding to αβ positions the essential ACT acylation sites, which are conserved among toxins exported by type I secretion systems, at the cell membrane. These findings reveal a structural mechanism for integrin-mediated attachment and explain antibody-mediated neutralization of ACT intoxication. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26739.map.gz emd_26739.map.gz | 119 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26739-v30.xml emd-26739-v30.xml emd-26739.xml emd-26739.xml | 12.3 KB 12.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_26739.png emd_26739.png | 53.3 KB | ||
| Filedesc metadata |  emd-26739.cif.gz emd-26739.cif.gz | 6.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26739 http://ftp.pdbj.org/pub/emdb/structures/EMD-26739 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26739 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26739 | HTTPS FTP |
-Validation report
| Summary document |  emd_26739_validation.pdf.gz emd_26739_validation.pdf.gz | 363.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_26739_full_validation.pdf.gz emd_26739_full_validation.pdf.gz | 363.3 KB | Display | |
| Data in XML |  emd_26739_validation.xml.gz emd_26739_validation.xml.gz | 6.8 KB | Display | |
| Data in CIF |  emd_26739_validation.cif.gz emd_26739_validation.cif.gz | 7.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26739 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26739 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26739 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26739 | HTTPS FTP |
-Related structure data
| Related structure data |  7usmMC  7uslC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26739.map.gz / Format: CCP4 / Size: 147.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26739.map.gz / Format: CCP4 / Size: 147.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite map of sharpened global and local reconstructions | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.073 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Integrin alphaM/beta2 ectodomain
| Entire | Name: Integrin alphaM/beta2 ectodomain |
|---|---|
| Components |
|
-Supramolecule #1: Integrin alphaM/beta2 ectodomain
| Supramolecule | Name: Integrin alphaM/beta2 ectodomain / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Integrin alpha-M
| Macromolecule | Name: Integrin alpha-M / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 128.455289 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: FNLDTENAMT FQENARGFGQ SVVQLQGSRV VVGAPQEIVA ANQRGSLYQC DYSTGSCEPI RLQVPVEAVN MSLGLSLAAT TSPPQLLAC GPTVHQTCSE NTYVKGLCFL FGSNLRQQPQ KFPEALRGCP QEDSDIAFLI DGSGSIIPHD FRRMKEFVST V MEQLKKSK ...String: FNLDTENAMT FQENARGFGQ SVVQLQGSRV VVGAPQEIVA ANQRGSLYQC DYSTGSCEPI RLQVPVEAVN MSLGLSLAAT TSPPQLLAC GPTVHQTCSE NTYVKGLCFL FGSNLRQQPQ KFPEALRGCP QEDSDIAFLI DGSGSIIPHD FRRMKEFVST V MEQLKKSK TLFSLMQYSE EFRIHFTFKE FQNNPNPRSL VKPITQLLGR THTATGIRKV VRELFNITNG ARKNAFKILV VI TDGEKFG DPLGYEDVIP EADREGVIRY VIGVGDAFRS EKSRQELNTI ASKPPRDHVF QVNNFEALKT IQNQLREKIF AIE GTQTGS SSSFEHEMSQ EGFSAAITSN GPLLSTVGSY DWAGGVFLYT SKEKSTFINM TRVDSDMNDA YLGYAAAIIL RNRV QSLVL GAPRYQHIGL VAMFRQNTGM WESNANVKGT QIGAYFGASL CSVDVDSNGS TDLVLIGAPH YYEQTRGGQV SVCPL PRGR ARWQCDAVLY GEQGQPWGRF GAALTVLGDV NGDKLTDVAI GAPGEEDNRG AVYLFHGTSG SGISPSHSQR IAGSKL SPR LQYFGQSLSG GQDLTMDGLV DLTVGAQGHV LLLRSQPVLR VKAIMEFNPR EVARNVFECN DQVVKGKEAG EVRVCLH VQ KSTRDRLREG QIQSVVTYDL ALDSGRPHSR AVFNETKNST RRQTQVLGLT QTCETLKLQL PNCIEDPVSP IVLRLNFS L VGTPLSAFGN LRPVLAEDAQ RLFTALFPFE KNCGNDNICQ DDLSITFSFM SLDCLVVGGP REFNVTVTVR NDGEDSYRT QVTFFFPLDL SYRKVSTLQN QRSQRSWRLA CESASSTEVS GALKSTSCSI NHPIFPENSE VTFNITFDVD SKASLGNKLL LKANVTSEN NMPRTNKTEF QLELPVKYAV YMVVTSHGVS TKYLNFTASE NTSRVMQHQY QVSNLGQRSL PISLVFLVPV R LNQTVIWD RPQVTFSENL SSTCHTKERL PSHSDFLAEL RKAPVVNCSI AVCQRIQCDI PFFGIQEEFN ATLKGNLSFD WY IKTSHNH LLIVSTAEIL FNDSVFTLLP GQGAFVRSQT ETKVEPFEVP NGCGGLEVLF QGPGENAQCE KELQALEKEN AQL EWELQA LEKELAQWSH PQFEKGGGSG GGGSGGSAWS HPQFEK UniProtKB: Integrin alpha-M |
-Macromolecule #2: Integrin beta
| Macromolecule | Name: Integrin beta / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 80.784938 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QECTKFKVSS CRECIESGPG CTWCQKLNFT GPGDPDSIRC DTRPQLLMRG CAADDIMDPT SLAETQEDHN GGQKQLSPQK VTLYLRPGQ AAAFNVTFRR AKGYPIDLYY LMDLSYSMLD DLRNVKKLGG DLLRALNEIT ESGRIGFGSF VDKTVLPFVN T HPDKLRNP ...String: QECTKFKVSS CRECIESGPG CTWCQKLNFT GPGDPDSIRC DTRPQLLMRG CAADDIMDPT SLAETQEDHN GGQKQLSPQK VTLYLRPGQ AAAFNVTFRR AKGYPIDLYY LMDLSYSMLD DLRNVKKLGG DLLRALNEIT ESGRIGFGSF VDKTVLPFVN T HPDKLRNP CPNKEKECQP PFAFRHVLKL TNNSNQFQTE VGKQLISGNL DAPEGGLDAM MQVAACPEEI GWRNVTRLLV FA TDDGFHF AGDGKLGAIL TPNDGRCHLE DNLYKRSNEF DYPSVGQLAH KLAENNIQPI FAVTSRMVKT YEKLTEIIPK SAV GELSED SSNVVHLIKN AYNKLSSRVF LDHNALPDTL KVTYDSFCSN GVTHRNQPRG DCDGVQINVP ITFQVKVTAT ECIQ EQSFV IRALGFTDIV TVQVLPQCEC RCRDQSRDRS LCHGKGFLEC GICRCDTGYI GKNCECQTQG RSSQELEGSC RKDNN SIIC SGLGDCVCGQ CLCHTSDVPG KLIYGQYCEC DTINCERYNG QVCGGPGRGL CFCGKCRCHP GFEGSACQCE RTTEGC LNP RRVECSGRGR CRCNVCECHS GYQLPLCQEC PGCPSPCGKY ISCAECLKFE KGPFGKNCSA ACPGLQLSNN PVKGRTC KE RDSEGCWVAY TLEQQDGMDR YLIYVDESRE CVAGPDGCGL EVLFQGPGKN AQCKKKLQAL KKKNAQLKWK LQALKKKL A QGGHHHHHH UniProtKB: Integrin beta |
-Macromolecule #4: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 4 / Number of copies: 4 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Macromolecule #5: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 5 / Number of copies: 9 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 80.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.2 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.7 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 1114678 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller





















 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)





















