[English] 日本語
 Yorodumi
Yorodumi- EMDB-21112: VRC03 and 10-1074 Bound BG505 F14 HIV-1 SOSIP Envelope Trimer Str... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-21112 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | VRC03 and 10-1074 Bound BG505 F14 HIV-1 SOSIP Envelope Trimer Structure | |||||||||
 Map data Map data | VRC03 and 10-1074 Bound BG505 F14/Vt8 HIV-1 SOSIP Envelope Trimer | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Trimer / Complex / Immunogen / HIV-1 / VIRAL PROTEIN-IMMUNE SYSTEM complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated perturbation of host defense response / immunoglobulin complex / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / viral protein processing / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane ...symbiont-mediated perturbation of host defense response / immunoglobulin complex / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / viral protein processing / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity / extracellular region / identical protein binding / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |   Human immunodeficiency virus 1 / Human immunodeficiency virus 1 /  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.9 Å | |||||||||
 Authors Authors | Henderson R / Acharya P / Bartesaghi A / Zhou Y | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2020 Journal: Nat Commun / Year: 2020Title: Disruption of the HIV-1 Envelope allosteric network blocks CD4-induced rearrangements. Authors: Rory Henderson / Maolin Lu / Ye Zhou / Zekun Mu / Robert Parks / Qifeng Han / Allen L Hsu / Elizabeth Carter / Scott C Blanchard / R J Edwards / Kevin Wiehe / Kevin O Saunders / Mario J ...Authors: Rory Henderson / Maolin Lu / Ye Zhou / Zekun Mu / Robert Parks / Qifeng Han / Allen L Hsu / Elizabeth Carter / Scott C Blanchard / R J Edwards / Kevin Wiehe / Kevin O Saunders / Mario J Borgnia / Alberto Bartesaghi / Walther Mothes / Barton F Haynes / Priyamvada Acharya / S Munir Alam /  Abstract: The trimeric HIV-1 Envelope protein (Env) mediates viral-host cell fusion via a network of conformational transitions, with allosteric elements in each protomer orchestrating host receptor-induced ...The trimeric HIV-1 Envelope protein (Env) mediates viral-host cell fusion via a network of conformational transitions, with allosteric elements in each protomer orchestrating host receptor-induced exposure of the co-receptor binding site and fusion elements. To understand the molecular details of this allostery, here, we introduce Env mutations aimed to prevent CD4-induced rearrangements in the HIV-1 BG505 Env trimer. Binding analysis and single-molecule Förster Resonance Energy Transfer confirm that these mutations prevent CD4-induced transitions of the HIV-1 Env. Structural analysis by single-particle cryo-electron microscopy performed on the BG505 SOSIP mutant Env proteins shows rearrangements in the gp120 topological layer contacts with gp41. Displacement of a conserved tryptophan (W571) from its typical pocket in these Env mutants renders the Env insensitive to CD4 binding. These results reveal the critical function of W571 as a conformational switch in Env allostery and receptor-mediated viral entry and provide insights on Env conformation that are relevant for vaccine design. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_21112.map.gz emd_21112.map.gz | 43 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-21112-v30.xml emd-21112-v30.xml emd-21112.xml emd-21112.xml | 18.9 KB 18.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_21112.png emd_21112.png | 44.3 KB | ||
| Filedesc metadata |  emd-21112.cif.gz emd-21112.cif.gz | 6.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21112 http://ftp.pdbj.org/pub/emdb/structures/EMD-21112 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21112 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21112 | HTTPS FTP |
-Related structure data
| Related structure data |  6v8zMC  6v8xC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_21112.map.gz / Format: CCP4 / Size: 46.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_21112.map.gz / Format: CCP4 / Size: 46.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
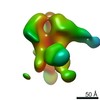
| Annotation | VRC03 and 10-1074 Bound BG505 F14/Vt8 HIV-1 SOSIP Envelope Trimer | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08312 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Complex between VRC03, 10-1074, and BG505 F14 HIV-1 SOSIP Env Trimer
+Supramolecule #1: Complex between VRC03, 10-1074, and BG505 F14 HIV-1 SOSIP Env Trimer
+Supramolecule #2: BG505 F14/Vt8 HIV-1 SOSIP Env Trimer
+Supramolecule #3: VRC03
+Supramolecule #4: 10-1074
+Macromolecule #1: Envelope glycoprotein gp120
+Macromolecule #2: envelope glycoprotein gp41
+Macromolecule #3: 10-1074 Fab Heavy Chain
+Macromolecule #4: 10-1074 Fab Light Chain
+Macromolecule #5: VRC03 Fab Heavy Chain
+Macromolecule #6: VRC03 Fab Light Chain
+Macromolecule #11: 2-acetamido-2-deoxy-beta-D-glucopyranose
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Grid | Details: unspecified |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 54.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C3 (3 fold cyclic) / Resolution.type: BY AUTHOR / Resolution: 2.9 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 84378 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)