+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the outward-facing FLVCR2 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | MFS / choline transport / human transporter / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationheme export / heme transmembrane transporter activity / mitochondrial membrane / heme binding / endoplasmic reticulum membrane / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
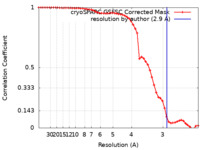
| Method | single particle reconstruction / cryo EM / Resolution: 2.9 Å | |||||||||
 Authors Authors | Weng T-H / Wu D / Safarian S | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Molecular mechanism of choline and ethanolamine transport in humans. Authors: Keiken Ri / Tsai-Hsuan Weng / Ainara Claveras Cabezudo / Wiebke Jösting / Yu Zhang / Andre Bazzone / Nancy C P Leong / Sonja Welsch / Raymond T Doty / Gonca Gursu / Tiffany Jia Ying Lim / ...Authors: Keiken Ri / Tsai-Hsuan Weng / Ainara Claveras Cabezudo / Wiebke Jösting / Yu Zhang / Andre Bazzone / Nancy C P Leong / Sonja Welsch / Raymond T Doty / Gonca Gursu / Tiffany Jia Ying Lim / Sarah Luise Schmidt / Janis L Abkowitz / Gerhard Hummer / Di Wu / Long N Nguyen / Schara Safarian /    Abstract: Human feline leukaemia virus subgroup C receptor-related proteins 1 and 2 (FLVCR1 and FLVCR2) are members of the major facilitator superfamily. Their dysfunction is linked to several clinical ...Human feline leukaemia virus subgroup C receptor-related proteins 1 and 2 (FLVCR1 and FLVCR2) are members of the major facilitator superfamily. Their dysfunction is linked to several clinical disorders, including PCARP, HSAN and Fowler syndrome. Earlier studies concluded that FLVCR1 may function as a haem exporter, whereas FLVCR2 was suggested to act as a haem importer, yet conclusive biochemical and detailed molecular evidence remained elusive for the function of both transporters. Here, we show that FLVCR1 and FLVCR2 facilitate the transport of choline and ethanolamine across the plasma membrane, using a concentration-driven substrate translocation process. Through structural and computational analyses, we have identified distinct conformational states of FLVCRs and unravelled the coordination chemistry underlying their substrate interactions. Fully conserved tryptophan and tyrosine residues form the binding pocket of both transporters and confer selectivity for choline and ethanolamine through cation-π interactions. Our findings clarify the mechanisms of choline and ethanolamine transport by FLVCR1 and FLVCR2, enhance our comprehension of disease-associated mutations that interfere with these vital processes and shed light on the conformational dynamics of these major facilitator superfamily proteins during the transport cycle. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_18337.map.gz emd_18337.map.gz | 7.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-18337-v30.xml emd-18337-v30.xml emd-18337.xml emd-18337.xml | 16.4 KB 16.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_18337_fsc.xml emd_18337_fsc.xml | 5.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_18337.png emd_18337.png | 60.1 KB | ||
| Filedesc metadata |  emd-18337.cif.gz emd-18337.cif.gz | 6 KB | ||
| Others |  emd_18337_half_map_1.map.gz emd_18337_half_map_1.map.gz emd_18337_half_map_2.map.gz emd_18337_half_map_2.map.gz | 13.5 MB 13.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-18337 http://ftp.pdbj.org/pub/emdb/structures/EMD-18337 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18337 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18337 | HTTPS FTP |
-Validation report
| Summary document |  emd_18337_validation.pdf.gz emd_18337_validation.pdf.gz | 866.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_18337_full_validation.pdf.gz emd_18337_full_validation.pdf.gz | 865.8 KB | Display | |
| Data in XML |  emd_18337_validation.xml.gz emd_18337_validation.xml.gz | 12.3 KB | Display | |
| Data in CIF |  emd_18337_validation.cif.gz emd_18337_validation.cif.gz | 15.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18337 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18337 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18337 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18337 | HTTPS FTP |
-Related structure data
| Related structure data |  8qcyMC  8qcsC  8qctC  8qcxC  8qd0C  8r8tC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_18337.map.gz / Format: CCP4 / Size: 14.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_18337.map.gz / Format: CCP4 / Size: 14.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.146 Å | ||||||||||||||||||||||||||||||||||||
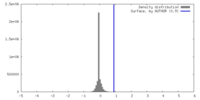
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_18337_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
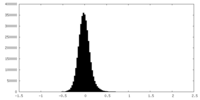
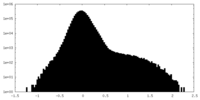
| Density Histograms |
-Half map: #1
| File | emd_18337_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
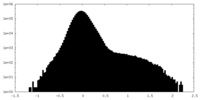
| Density Histograms |
- Sample components
Sample components
-Entire : FLVCR2 monomer
| Entire | Name: FLVCR2 monomer |
|---|---|
| Components |
|
-Supramolecule #1: FLVCR2 monomer
| Supramolecule | Name: FLVCR2 monomer / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 58 KDa |
-Macromolecule #1: Heme transporter FLVCR2
| Macromolecule | Name: Heme transporter FLVCR2 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 58.277094 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MVNEGPNQEE SDDTPVPESA LQADPSVSVH PSVSVHPSVS INPSVSVHPS SSAHPSALAQ PSGLAHPSSS GPEDLSVIKV SRRRWAVVL VFSCYSMCNS FQWIQYGSIN NIFMHFYGVS AFAIDWLSMC YMLTYIPLLL PVAWLLEKFG LRTIALTGSA L NCLGAWVK ...String: MVNEGPNQEE SDDTPVPESA LQADPSVSVH PSVSVHPSVS INPSVSVHPS SSAHPSALAQ PSGLAHPSSS GPEDLSVIKV SRRRWAVVL VFSCYSMCNS FQWIQYGSIN NIFMHFYGVS AFAIDWLSMC YMLTYIPLLL PVAWLLEKFG LRTIALTGSA L NCLGAWVK LGSLKPHLFP VTVVGQLICS VAQVFILGMP SRIASVWFGA NEVSTACSVA VFGNQLGIAI GFLVPPVLVP NI EDRDELA YHISIMFYII GGVATLLLIL VIIVFKEKPK YPPSRAQSLS YALTSPDASY LGSIARLFKN LNFVLLVITY GLN AGAFYA LSTLLNRMVI WHYPGEEVNA GRIGLTIVIA GMLGAVISGI WLDRSKTYKE TTLVVYIMTL VGMVVYTFTL NLGH LWVVF ITAGTMGFFM TGYLPLGFEF AVELTYPESE GISSGLLNIS AQVFGIIFTI SQGQIIDNYG TKPGNIFLCV FLTLG AALT AFIKADLRRQ KANKETLENK LQEEEEESNT SKVPTAVSED HLDYKDDDDK UniProtKB: Heme transporter FLVCR2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.5 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 90 sec. |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 80.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.1 µm / Nominal defocus min: 1.1 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)