+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Native eisosome lattice bound to plasma membrane microdomain | |||||||||||||||
 Map data Map data | Native eisosome lattice - sharpened map | |||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | BAR domain / plasma membrane microdomain / membrane curvature / native biochemistry / LIPID BINDING PROTEIN | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationprotein localization to eisosome filament / eisosome filament / eisosome assembly / eisosome / membrane organization / lipid droplet / cell periphery / endocytosis / intracellular protein localization / response to heat ...protein localization to eisosome filament / eisosome filament / eisosome assembly / eisosome / membrane organization / lipid droplet / cell periphery / endocytosis / intracellular protein localization / response to heat / mitochondrial outer membrane / lipid binding / mitochondrion / plasma membrane / cytoplasm Similarity search - Function | |||||||||||||||
| Biological species |  | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||||||||
 Authors Authors | Kefauver JM / Zou L / Loewith RJ / Desfosses A | |||||||||||||||
| Funding support | European Union,  Switzerland, 4 items Switzerland, 4 items
| |||||||||||||||
 Citation Citation | Journal: Acta Crystallogr D Struct Biol / Year: 2018 Title: Real-space refinement in PHENIX for cryo-EM and crystallography. Authors: Pavel V Afonine / Billy K Poon / Randy J Read / Oleg V Sobolev / Thomas C Terwilliger / Alexandre Urzhumtsev / Paul D Adams /    Abstract: This article describes the implementation of real-space refinement in the phenix.real_space_refine program from the PHENIX suite. The use of a simplified refinement target function enables very fast ...This article describes the implementation of real-space refinement in the phenix.real_space_refine program from the PHENIX suite. The use of a simplified refinement target function enables very fast calculation, which in turn makes it possible to identify optimal data-restraint weights as part of routine refinements with little runtime cost. Refinement of atomic models against low-resolution data benefits from the inclusion of as much additional information as is available. In addition to standard restraints on covalent geometry, phenix.real_space_refine makes use of extra information such as secondary-structure and rotamer-specific restraints, as well as restraints or constraints on internal molecular symmetry. The re-refinement of 385 cryo-EM-derived models available in the Protein Data Bank at resolutions of 6 Å or better shows significant improvement of the models and of the fit of these models to the target maps. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_18307.map.gz emd_18307.map.gz | 59.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-18307-v30.xml emd-18307-v30.xml emd-18307.xml emd-18307.xml | 41 KB 41 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_18307.png emd_18307.png | 134.8 KB | ||
| Filedesc metadata |  emd-18307.cif.gz emd-18307.cif.gz | 6.7 KB | ||
| Others |  emd_18307_additional_1.map.gz emd_18307_additional_1.map.gz emd_18307_additional_10.map.gz emd_18307_additional_10.map.gz emd_18307_additional_11.map.gz emd_18307_additional_11.map.gz emd_18307_additional_2.map.gz emd_18307_additional_2.map.gz emd_18307_additional_3.map.gz emd_18307_additional_3.map.gz emd_18307_additional_4.map.gz emd_18307_additional_4.map.gz emd_18307_additional_5.map.gz emd_18307_additional_5.map.gz emd_18307_additional_6.map.gz emd_18307_additional_6.map.gz emd_18307_additional_7.map.gz emd_18307_additional_7.map.gz emd_18307_additional_8.map.gz emd_18307_additional_8.map.gz emd_18307_additional_9.map.gz emd_18307_additional_9.map.gz emd_18307_half_map_1.map.gz emd_18307_half_map_1.map.gz emd_18307_half_map_2.map.gz emd_18307_half_map_2.map.gz | 31.8 MB 271.1 MB 59.8 MB 267.4 MB 272.9 MB 273.9 MB 274.1 MB 272.7 MB 271.2 MB 272 MB 266.6 MB 59.3 MB 59.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-18307 http://ftp.pdbj.org/pub/emdb/structures/EMD-18307 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18307 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18307 | HTTPS FTP |
-Related structure data
| Related structure data |  8qb7MC  8qb8MC  8qb9C  8qbbC  8qbdC  8qbeC  8qbfC  8qbgC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_18307.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_18307.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Native eisosome lattice - sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.327 Å | ||||||||||||||||||||||||||||||||||||
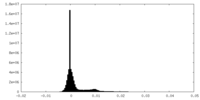
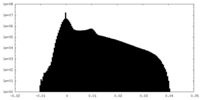
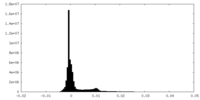
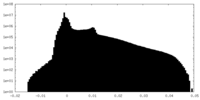
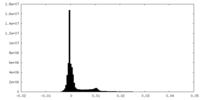
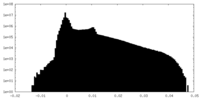
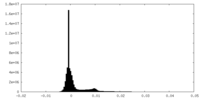
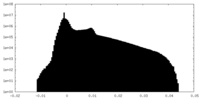
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
+Additional map: Native eisosome lattice - unsharpened map
+Additional map: Native eisosome helical map (C2, rise=11.00, twist=54.02)
+Additional map: Native eisosome lattice - deepEMhancer sharpened map
+Additional map: Native eisosome helical map (C4, rise=21.27, twist=50.99)
+Additional map: Native eisosome helical map (C7, rise=37.28, twist=36.72)
+Additional map: Native eisosome helical map (C1, rise=5.58, twist=53.51)
+Additional map: Native eisosome helical map (C1, rise=5.13, twist=49.32)
+Additional map: Native eisosome helical map (C1, rise=5.2, twist=136.56)
+Additional map: Native eisosome helical map (C2, rise=10.02, twist=47.98)
+Additional map: Native eisosome helical map (C1, rise=4.89, twist=46.59)
+Additional map: Native eisosome helical map (C8, rise=37.41, twist=57.83)
+Half map: Native eisosome lattice - half map B
+Half map: Native eisosome lattice - half map A
- Sample components
Sample components
-Entire : Helical lattice of native Pil1/Lsp1 protein bound to plasma membr...
| Entire | Name: Helical lattice of native Pil1/Lsp1 protein bound to plasma membrane microdomain |
|---|---|
| Components |
|
-Supramolecule #1: Helical lattice of native Pil1/Lsp1 protein bound to plasma membr...
| Supramolecule | Name: Helical lattice of native Pil1/Lsp1 protein bound to plasma membrane microdomain type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Sphingolipid long chain base-responsive protein PIL1
| Macromolecule | Name: Sphingolipid long chain base-responsive protein PIL1 / type: protein_or_peptide / ID: 1 / Number of copies: 14 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 38.393043 KDa |
| Sequence | String: MHRTYSLRNS RAPTASQLQN PPPPPSTTKG RFFGKGGLAY SFRRSAAGAF GPELSRKLSQ LVKIEKNVLR SMELTANERR DAAKQLSIW GLENDDDVSD ITDKLGVLIY EVSELDDQFI DRYDQYRLTL KSIRDIEGSV QPSRDRKDKI TDKIAYLKYK D PQSPKIEV ...String: MHRTYSLRNS RAPTASQLQN PPPPPSTTKG RFFGKGGLAY SFRRSAAGAF GPELSRKLSQ LVKIEKNVLR SMELTANERR DAAKQLSIW GLENDDDVSD ITDKLGVLIY EVSELDDQFI DRYDQYRLTL KSIRDIEGSV QPSRDRKDKI TDKIAYLKYK D PQSPKIEV LEQELVRAEA ESLVAEAQLS NITRSKLRAA FNYQFDSIIE HSEKIALIAG YGKALLELLD DSPVTPGETR PA YDGYEAS KQIIIDAESA LNEWTLDSAQ VKPTLSFKQD YEDFEPEEGE EEEEEDGQGR WSEDEQEDGQ IEEPEQEEEG AVE EHEQVG HQQSESLPQQ TTA UniProtKB: Sphingolipid long chain base-responsive protein PIL1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Buffer | pH: 7 / Details: 50mM PIPES pH 7, 300mM NaCl, 1mM CHAPS, 0.5mM DTT |
|---|---|
| Grid | Model: EMS Lacey Carbon / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: LACEY |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER / Details: Cylindrical map |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.2 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC (ver. 4.1.2) / Number images used: 2423944 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.0.8) |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC (ver. 4.1.2) |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)