[English] 日本語
 Yorodumi
Yorodumi- EMDB-15784: CryoEM structure of bacterial RapZ.GlmZ complex central to the co... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
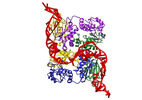
| Title | CryoEM structure of bacterial RapZ.GlmZ complex central to the control of cell envelope biogenesis | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | endonuclease RNase E / adaptor protein RapZ / small regulatory RNA GlmZ / RNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationRNA destabilization / carbohydrate derivative binding / protein homotetramerization / molecular adaptor activity / GTP binding / protein-containing complex / RNA binding / ATP binding / identical protein binding Similarity search - Function | |||||||||
| Biological species |   | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.28 Å | |||||||||
 Authors Authors | Islam MS / Hardwick HW / Chirgadze DY / Luisi BF | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: EMBO J / Year: 2023 Journal: EMBO J / Year: 2023Title: Structure of a bacterial ribonucleoprotein complex central to the control of cell envelope biogenesis. Authors: Md Saiful Islam / Steven W Hardwick / Laura Quell / Svetlana Durica-Mitic / Dimitri Y Chirgadze / Boris Görke / Ben F Luisi /   Abstract: Biogenesis of the essential precursor of the bacterial cell envelope, glucosamine-6-phosphate (GlcN6P), is controlled by intricate post-transcriptional networks mediated by GlmZ, a small regulatory ...Biogenesis of the essential precursor of the bacterial cell envelope, glucosamine-6-phosphate (GlcN6P), is controlled by intricate post-transcriptional networks mediated by GlmZ, a small regulatory RNA (sRNA). GlmZ stimulates translation of the mRNA encoding GlcN6P synthtase in Escherichia coli, but when bound by RapZ protein, the sRNA becomes inactivated through cleavage by the endoribonuclease RNase E. Here, we report the cryoEM structure of the RapZ:GlmZ complex, revealing a complementary match of the RapZ tetrameric quaternary structure to structural repeats in the sRNA. The nucleic acid is contacted by RapZ mostly through a highly conserved domain that shares an evolutionary relationship with phosphofructokinase and suggests links between metabolism and riboregulation. We also present the structure of a precleavage intermediate formed between the binary RapZ:GlmZ complex and RNase E that reveals how GlmZ is presented and recognised by the enzyme. The structures provide a framework for understanding how other encounter complexes might guide recognition and action of endoribonucleases on target transcripts, and how structured substrates in polycistronic precursors may be recognised for processing by RNase E. #1:  Journal: Biorxiv / Year: 2022 Journal: Biorxiv / Year: 2022Title: Structure of a bacterial ribonucleoprotein complex central to the control of cell envelope biogenesis Authors: Islam MS / Hardwick SW / Quell L / Chirgadze DY / Gorke B / Luisi BF | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15784.map.gz emd_15784.map.gz | 344.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15784-v30.xml emd-15784-v30.xml emd-15784.xml emd-15784.xml | 21.6 KB 21.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_15784.png emd_15784.png | 73.4 KB | ||
| Filedesc metadata |  emd-15784.cif.gz emd-15784.cif.gz | 6.6 KB | ||
| Others |  emd_15784_half_map_1.map.gz emd_15784_half_map_1.map.gz emd_15784_half_map_2.map.gz emd_15784_half_map_2.map.gz | 344.6 MB 344.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15784 http://ftp.pdbj.org/pub/emdb/structures/EMD-15784 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15784 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15784 | HTTPS FTP |
-Validation report
| Summary document |  emd_15784_validation.pdf.gz emd_15784_validation.pdf.gz | 1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_15784_full_validation.pdf.gz emd_15784_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  emd_15784_validation.xml.gz emd_15784_validation.xml.gz | 16.7 KB | Display | |
| Data in CIF |  emd_15784_validation.cif.gz emd_15784_validation.cif.gz | 19.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15784 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15784 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15784 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15784 | HTTPS FTP |
-Related structure data
| Related structure data |  8b0iMC  8b0jC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_15784.map.gz / Format: CCP4 / Size: 371.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15784.map.gz / Format: CCP4 / Size: 371.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.652 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_15784_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
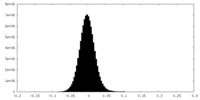
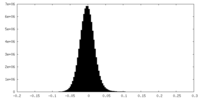
| Density Histograms |
-Half map: #1
| File | emd_15784_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
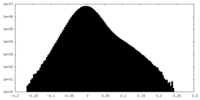
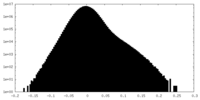
| Density Histograms |
- Sample components
Sample components
-Entire : RapZ tetramer, GlmZ stem loops I & II
| Entire | Name: RapZ tetramer, GlmZ stem loops I & II |
|---|---|
| Components |
|
-Supramolecule #1: RapZ tetramer, GlmZ stem loops I & II
| Supramolecule | Name: RapZ tetramer, GlmZ stem loops I & II / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all / Details: In vitro reconstituted RapZ and GlmZ complex |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: RNase adapter protein RapZ
| Macromolecule | Name: RNase adapter protein RapZ / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 32.53832 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MVLMIVSGRS GSGKSVALRA LEDMGFYCVD NLPVVLLPDL ARTLADREIS AAVSIDVRNM PESPEIFEQA MSNLPDAFSP QLLFLDADR NTLIRRYSDT RRLHPLSSKN LSLESAIDKE SDLLEPLRSR ADLIVDTSEM SVHELAEMLR TRLLGKRERE L TMVFESFG ...String: MVLMIVSGRS GSGKSVALRA LEDMGFYCVD NLPVVLLPDL ARTLADREIS AAVSIDVRNM PESPEIFEQA MSNLPDAFSP QLLFLDADR NTLIRRYSDT RRLHPLSSKN LSLESAIDKE SDLLEPLRSR ADLIVDTSEM SVHELAEMLR TRLLGKRERE L TMVFESFG FKHGIPIDAD YVFDVRFLPN PHWDPKLRPM TGLDKPVAAF LDRHTEVHNF IYQTRSYLEL WLPMLETNNR SY LTVAIGC TGGKHRSVYI AEQLADYFRS RGKNVQSRHR TLEKRKP UniProtKB: RNase adapter protein RapZ |
-Macromolecule #2: GlmZ small regulatory RNA
| Macromolecule | Name: GlmZ small regulatory RNA / type: rna / ID: 2 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 66.061859 KDa |
| Sequence | String: GUAGAUGCUC AUUCCAUCUC UUAUGUUCGC CUUAGUGCCU CAUAAACUCC GGAAUGACGC AGAGCCGUUU ACGGUGCUUA UCGUCCACU GACAGAUGUC GCUUAUGCCU CAUCAGACAC CAUGGACACA ACGUUGAGUG AAGCACCCAC UUGUUGUCAU A CAGACCUG ...String: GUAGAUGCUC AUUCCAUCUC UUAUGUUCGC CUUAGUGCCU CAUAAACUCC GGAAUGACGC AGAGCCGUUU ACGGUGCUUA UCGUCCACU GACAGAUGUC GCUUAUGCCU CAUCAGACAC CAUGGACACA ACGUUGAGUG AAGCACCCAC UUGUUGUCAU A CAGACCUG UUUUAACGCC UGCUCCGUUA AUAAGAGCAG GCGUUUUUU |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | 3D array |
- Sample preparation
Sample preparation
| Concentration | 5 mg/mL |
|---|---|
| Buffer | pH: 7.5 / Details: 25 mM HEPES pH 7.5, 300 KCl, and 1 mM MgCl2 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 120 sec. |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 47.3 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: DIFFRACTION / Nominal defocus max: 0.3 µm / Nominal defocus min: 0.1 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)