+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of an open form of CHIKV nsP1 capping pores | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Alphavirus Replication complex / capping pores / membrane pore / Methyltransferase / Guanylyl transferase / VIRAL PROT / VIRAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationADP-ribose 1''-phosphate phosphatase / host cell filopodium / mRNA methyltransferase activity / mRNA 5'-phosphatase / mRNA 5'-triphosphate monophosphatase activity / polynucleotide adenylyltransferase / poly(A) RNA polymerase activity / mRNA modification / regulation of cytoskeleton organization / symbiont-mediated suppression of host mRNA transcription via inhibition of RNA polymerase II activity ...ADP-ribose 1''-phosphate phosphatase / host cell filopodium / mRNA methyltransferase activity / mRNA 5'-phosphatase / mRNA 5'-triphosphate monophosphatase activity / polynucleotide adenylyltransferase / poly(A) RNA polymerase activity / mRNA modification / regulation of cytoskeleton organization / symbiont-mediated suppression of host mRNA transcription via inhibition of RNA polymerase II activity / 7-methylguanosine mRNA capping / symbiont-mediated suppression of host JAK-STAT cascade via inhibition of STAT1 activity / cysteine-type peptidase activity / Transferases; Transferring one-carbon groups; Methyltransferases / host cell cytoplasmic vesicle membrane / Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases / nucleoside-triphosphate phosphatase / methylation / Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases / RNA helicase activity / symbiont-mediated suppression of host innate immune response / RNA helicase / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / symbiont-mediated suppression of host gene expression / RNA-directed RNA polymerase / viral RNA genome replication / RNA-directed RNA polymerase activity / DNA-templated transcription / GTP binding / host cell plasma membrane / host cell nucleus / ATP hydrolysis activity / proteolysis / RNA binding / ATP binding / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Chikungunya virus strain S27-African prototype Chikungunya virus strain S27-African prototype | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.8 Å | |||||||||
 Authors Authors | Reguera J / Jones R / Hons M | |||||||||
| Funding support |  France, 1 items France, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2023 Journal: Proc Natl Acad Sci U S A / Year: 2023Title: Structural basis and dynamics of Chikungunya alphavirus RNA capping by nsP1 capping pores. Authors: Rhian Jones / Michael Hons / Nadia Rabah / Noelia Zamarreño / Rocío Arranz / Juan Reguera /   Abstract: Alphaviruses are emerging positive-stranded RNA viruses which replicate and transcribe their genomes in membranous organelles formed in the cell cytoplasm. The nonstructural protein 1 (nsP1) is ...Alphaviruses are emerging positive-stranded RNA viruses which replicate and transcribe their genomes in membranous organelles formed in the cell cytoplasm. The nonstructural protein 1 (nsP1) is responsible for viral RNA capping and gates the replication organelles by assembling into monotopic membrane-associated dodecameric pores. The capping pathway is unique to Alphaviruses; beginning with the N methylation of a guanosine triphosphate (GTP) molecule, followed by the covalent linkage of an mGMP group to a conserved histidine in nsP1 and the transfer of this cap structure to a diphosphate RNA. Here, we provide structural snapshots of different stages of the reaction pathway showing how nsP1 pores recognize the substrates of the methyl-transfer reaction, GTP and S-adenosyl methionine (SAM), how the enzyme reaches a metastable postmethylation state with SAH and mGTP in the active site, and the subsequent covalent transfer of mGMP to nsP1 triggered by the presence of RNA and postdecapping reaction conformational changes inducing the opening of the pore. In addition, we biochemically characterize the capping reaction, demonstrating specificity for the RNA substrate and the reversibility of the cap transfer resulting in decapping activity and the release of reaction intermediates. Our data identify the molecular determinants allowing each pathway transition, providing an explanation for the need for the SAM methyl donor all along the pathway and clues about the conformational rearrangements associated to the enzymatic activity of nsP1. Together, our results set ground for the structural and functional understanding of alphavirus RNA-capping and the design of antivirals. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15704.map.gz emd_15704.map.gz | 449.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15704-v30.xml emd-15704-v30.xml emd-15704.xml emd-15704.xml | 22.5 KB 22.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_15704.png emd_15704.png | 66.7 KB | ||
| Filedesc metadata |  emd-15704.cif.gz emd-15704.cif.gz | 6.8 KB | ||
| Others |  emd_15704_half_map_1.map.gz emd_15704_half_map_1.map.gz emd_15704_half_map_2.map.gz emd_15704_half_map_2.map.gz | 441.5 MB 441.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15704 http://ftp.pdbj.org/pub/emdb/structures/EMD-15704 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15704 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15704 | HTTPS FTP |
-Related structure data
| Related structure data |  8axvMC  8aovC  8aowC  8aoxC  8apxC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_15704.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15704.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.839 Å | ||||||||||||||||||||||||||||||||||||
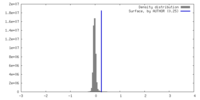
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_15704_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
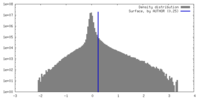
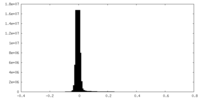
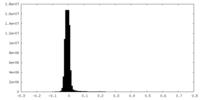
| Density Histograms |
-Half map: #1
| File | emd_15704_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
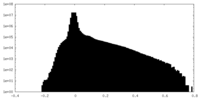
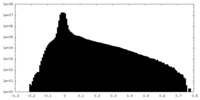
| Density Histograms |
- Sample components
Sample components
-Entire : Chikungunya nsP1 in an open form
| Entire | Name: Chikungunya nsP1 in an open form |
|---|---|
| Components |
|
-Supramolecule #1: Chikungunya nsP1 in an open form
| Supramolecule | Name: Chikungunya nsP1 in an open form / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Chikungunya virus strain S27-African prototype Chikungunya virus strain S27-African prototype |
| Molecular weight | Theoretical: 720 KDa |
-Macromolecule #1: mRNA-capping enzyme nsP1
| Macromolecule | Name: mRNA-capping enzyme nsP1 / type: protein_or_peptide / ID: 1 / Number of copies: 12 / Enantiomer: LEVO EC number: Transferases; Transferring one-carbon groups; Methyltransferases |
|---|---|
| Source (natural) | Organism:  Chikungunya virus strain S27-African prototype / Strain: S27-African prototype Chikungunya virus strain S27-African prototype / Strain: S27-African prototype |
| Molecular weight | Theoretical: 59.975422 KDa |
| Recombinant expression | Organism:  Baculovirus expression vector pFastBac1-HM Baculovirus expression vector pFastBac1-HM |
| Sequence | String: MDPVYVDIDA DSAFLKALQR AYPMFEVEPR QVTPNDHANA RAFSHLAIKL IEQEIDPDST ILDIGSAPAR RMMSDRKYHC VCPMRSAED PERLANYARK LASAAGKVLD RNISGKIGDL QAVMAVPDTE TPTFCLHTDV SCRQRADVAI YQDVYAVHAP T SLYHQAIK ...String: MDPVYVDIDA DSAFLKALQR AYPMFEVEPR QVTPNDHANA RAFSHLAIKL IEQEIDPDST ILDIGSAPAR RMMSDRKYHC VCPMRSAED PERLANYARK LASAAGKVLD RNISGKIGDL QAVMAVPDTE TPTFCLHTDV SCRQRADVAI YQDVYAVHAP T SLYHQAIK GVRLAYWVGF DTTPFMYNAM AGAYPSYSTN WADEQVLKAK NIGLCSTDLT EGRRGKLSIM RGKKLEPCDR VL FSVGSTL YPESRKLLKS WHLPSVFHLK GKLSFTCRCD TVVSCEGYVV KRITMSPGLY GKTTGYAVTH HADGFLMCKT TDT VDGERV SFSVCTYVPA TICDQMTGIL ATEVTPEDAQ KLLVGLNQRI VVNGRTQRNT NTMKNYMIPV VAQAFSKWAK ECRK DMEDE KLLGVRERTL TCCCLWAFKK QKTHTVYKRP DTQSIQKVQA EFDSFVVPSL WSSGLSIPLR TRIKWLLSKV PKTDL TPYS GDAQEARDAE KEAEEEREAE LTLEALPPLQ AAQEDVQVEI DVEQLEDRAG A UniProtKB: Polyprotein P1234 |
-Macromolecule #2: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 2 / Number of copies: 12 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #3: 2-amino-7-methyl-1,7-dihydro-6H-purin-6-one
| Macromolecule | Name: 2-amino-7-methyl-1,7-dihydro-6H-purin-6-one / type: ligand / ID: 3 / Number of copies: 12 / Formula: MY6 |
|---|---|
| Molecular weight | Theoretical: 165.153 Da |
| Chemical component information | 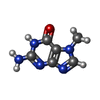 ChemComp-MY6: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.15 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.6 Component:
| |||||||||||||||
| Grid | Model: Quantifoil R2/2 / Material: GOLD / Mesh: 300 / Support film - Material: GRAPHENE OXIDE / Support film - topology: CONTINUOUS / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 10 sec. / Pretreatment - Atmosphere: AIR | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 98 % / Chamber temperature: 25 K / Instrument: FEI VITROBOT MARK IV Details: Sample was incubated with an RNA with a 5' methyl guanosine cap prior to vitrification. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 44.7 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 105000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)