[English] 日本語
 Yorodumi
Yorodumi- EMDB-15272: CRYO-EM STRUCTURE OF LEISHMANIA MAJOR 80S RIBOSOME : snoRNA MUTANT -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CRYO-EM STRUCTURE OF LEISHMANIA MAJOR 80S RIBOSOME : snoRNA MUTANT | |||||||||
 Map data Map data | Merged Map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CRYO-EM / LEISHMANIA MAJOR / 80S RIBOSOME snoRNA MUTANT / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationnuclear lumen / ciliary plasm / ciliary transition zone / negative regulation of translational frameshifting / endonucleolytic cleavage to generate mature 3'-end of SSU-rRNA from (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / protein-RNA complex assembly / maturation of LSU-rRNA / endonucleolytic cleavage in ITS1 to separate SSU-rRNA from 5.8S rRNA and LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / translation regulator activity / rescue of stalled cytosolic ribosome ...nuclear lumen / ciliary plasm / ciliary transition zone / negative regulation of translational frameshifting / endonucleolytic cleavage to generate mature 3'-end of SSU-rRNA from (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / protein-RNA complex assembly / maturation of LSU-rRNA / endonucleolytic cleavage in ITS1 to separate SSU-rRNA from 5.8S rRNA and LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / translation regulator activity / rescue of stalled cytosolic ribosome / protein kinase C binding / ribosomal large subunit biogenesis / maturation of LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / maturation of SSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / maturation of SSU-rRNA / small-subunit processome / maintenance of translational fidelity / rRNA processing / kinase activity / ribosomal small subunit assembly / ribosome biogenesis / ribosome binding / ribosomal small subunit biogenesis / 5S rRNA binding / small ribosomal subunit / ribosomal large subunit assembly / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / negative regulation of translation / rRNA binding / structural constituent of ribosome / ribosome / translation / ribonucleoprotein complex / mRNA binding / nucleolus / RNA binding / zinc ion binding / nucleoplasm / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Leishmania major strain Friedlin (eukaryote) Leishmania major strain Friedlin (eukaryote) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.46 Å | |||||||||
 Authors Authors | Rajan KS / Yonath A / Bashan A | |||||||||
| Funding support |  Israel, 1 items Israel, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2024 Journal: Cell Rep / Year: 2024Title: Structural and mechanistic insights into the function of Leishmania ribosome lacking a single pseudouridine modification. Authors: K Shanmugha Rajan / Saurav Aryal / Disha-Gajanan Hiregange / Anat Bashan / Hava Madmoni / Mika Olami / Tirza Doniger / Smadar Cohen-Chalamish / Pascal Pescher / Masato Taoka / Yuko Nobe / ...Authors: K Shanmugha Rajan / Saurav Aryal / Disha-Gajanan Hiregange / Anat Bashan / Hava Madmoni / Mika Olami / Tirza Doniger / Smadar Cohen-Chalamish / Pascal Pescher / Masato Taoka / Yuko Nobe / Aliza Fedorenko / Tanaya Bose / Ella Zimermann / Eric Prina / Noa Aharon-Hefetz / Yitzhak Pilpel / Toshiaki Isobe / Ron Unger / Gerald F Späth / Ada Yonath / Shulamit Michaeli /    Abstract: Leishmania is the causative agent of cutaneous and visceral diseases affecting millions of individuals worldwide. Pseudouridine (Ψ), the most abundant modification on rRNA, changes during the ...Leishmania is the causative agent of cutaneous and visceral diseases affecting millions of individuals worldwide. Pseudouridine (Ψ), the most abundant modification on rRNA, changes during the parasite life cycle. Alterations in the level of a specific Ψ in helix 69 (H69) affected ribosome function. To decipher the molecular mechanism of this phenotype, we determine the structure of ribosomes lacking the single Ψ and its parental strain at ∼2.4-3 Å resolution using cryo-EM. Our findings demonstrate the significance of a single Ψ on H69 to its structure and the importance for its interactions with helix 44 and specific tRNAs. Our study suggests that rRNA modification affects translation of mRNAs carrying codon bias due to selective accommodation of tRNAs by the ribosome. Based on the high-resolution structures, we propose a mechanism explaining how the ribosome selects specific tRNAs. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15272.map.gz emd_15272.map.gz | 56.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15272-v30.xml emd-15272-v30.xml emd-15272.xml emd-15272.xml | 109.1 KB 109.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_15272.png emd_15272.png | 77.2 KB | ||
| Filedesc metadata |  emd-15272.cif.gz emd-15272.cif.gz | 20.5 KB | ||
| Others |  emd_15272_additional_1.map.gz emd_15272_additional_1.map.gz emd_15272_half_map_1.map.gz emd_15272_half_map_1.map.gz emd_15272_half_map_2.map.gz emd_15272_half_map_2.map.gz | 66.2 MB 259.2 MB 259.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15272 http://ftp.pdbj.org/pub/emdb/structures/EMD-15272 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15272 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15272 | HTTPS FTP |
-Related structure data
| Related structure data |  8a98MC  8ovjC  8rxhC  8rxxC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_15272.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15272.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Merged Map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||||||||||||||||||
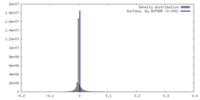
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Post processed consensus map
| File | emd_15272_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post processed consensus map | ||||||||||||
| Projections & Slices |
| ||||||||||||
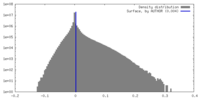
| Density Histograms |
-Half map: Half Map 1
| File | emd_15272_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
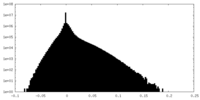
| Density Histograms |
-Half map: Half Map 2
| File | emd_15272_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
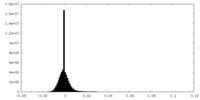
| Density Histograms |
- Sample components
Sample components
+Entire : CRYO-EM STRUCTURE OF LEISHMANIA MAJOR 80S RIBOSOME snoRNA MUTANT
+Supramolecule #1: CRYO-EM STRUCTURE OF LEISHMANIA MAJOR 80S RIBOSOME snoRNA MUTANT
+Macromolecule #1: Putative 40S ribosomal protein S15
+Macromolecule #2: 40S ribosomal protein S24
+Macromolecule #3: Putative 40S ribosomal protein S21
+Macromolecule #4: 40S ribosomal protein S19-like protein
+Macromolecule #5: Putative 40S ribosomal protein S17
+Macromolecule #6: Putative 40S ribosomal protein S11
+Macromolecule #7: Putative 40S ribosomal protein S13
+Macromolecule #8: Putative ribosomal protein S29
+Macromolecule #9: Putative 40S ribosomal protein S18
+Macromolecule #10: 40S ribosomal protein SA
+Macromolecule #11: Putative 40S ribosomal protein S3
+Macromolecule #12: Putative 40S ribosomal protein S9
+Macromolecule #13: 40S ribosomal protein S4
+Macromolecule #14: 40S ribosomal protein S8
+Macromolecule #16: Small ribosomal subunit protein RACK1
+Macromolecule #17: 40S ribosomal protein S25
+Macromolecule #18: Putative 40S ribosomal protein S27-1
+Macromolecule #19: 40S ribosomal protein S30
+Macromolecule #21: 40S ribosomal protein S12
+Macromolecule #22: Putative 40S ribosomal protein S23
+Macromolecule #23: 40S ribosomal protein S14
+Macromolecule #24: Putative 40S ribosomal protein S10
+Macromolecule #25: Putative 40S ribosomal protein S33
+Macromolecule #26: Putative ribosomal protein S20
+Macromolecule #27: Putative 40S ribosomal protein S16
+Macromolecule #28: Putative 40S ribosomal protein S15A
+Macromolecule #29: 40S ribosomal protein S7
+Macromolecule #30: 40S ribosomal protein S5
+Macromolecule #31: 40S ribosomal protein S6
+Macromolecule #32: 40S ribosomal protein S2
+Macromolecule #33: 40S ribosomal protein S3a
+Macromolecule #34: 40S ribosomal protein S26
+Macromolecule #35: Putative RNA binding protein
+Macromolecule #36: 60S ribosomal protein L32
+Macromolecule #37: 60S ribosomal protein L30
+Macromolecule #38: Putative 60S ribosomal protein L7
+Macromolecule #39: Putative 60S ribosomal protein L35
+Macromolecule #40: 60S ribosomal protein L29
+Macromolecule #45: Putative ribosomal protein L3
+Macromolecule #48: Putative 60S ribosomal protein L2
+Macromolecule #49: Putative 60S ribosomal protein L5
+Macromolecule #50: 60S ribosomal protein L7a
+Macromolecule #51: Putative 60S ribosomal protein L13a
+Macromolecule #52: Ribosomal protein L15
+Macromolecule #53: eL13_chain_I
+Macromolecule #54: 60S ribosomal protein L18
+Macromolecule #55: Putative 60S ribosomal protein L19
+Macromolecule #56: 60S ribosomal protein L18a
+Macromolecule #57: Putative 60S ribosomal protein L9
+Macromolecule #59: Putative 60S ribosomal protein L17
+Macromolecule #60: Putative 60S ribosomal protein L21
+Macromolecule #61: Putative 40S ribosomal protein L14
+Macromolecule #62: 60S ribosomal protein L11
+Macromolecule #63: Putative 60S ribosomal protein L27A/L29
+Macromolecule #64: 60S ribosomal protein L27
+Macromolecule #65: Putative 60S ribosomal protein L28
+Macromolecule #66: Putative 60S ribosomal protein L6
+Macromolecule #67: Putative 60S ribosomal protein L34
+Macromolecule #68: Putative ribosomal protein l35a
+Macromolecule #69: Putative 60S ribosomal protein L23
+Macromolecule #70: Putative 60S ribosomal protein L23a
+Macromolecule #71: Putative 60S ribosomal protein L26
+Macromolecule #72: Putative 60S ribosomal protein L22
+Macromolecule #73: Putative 60S Ribosomal protein L36
+Macromolecule #74: Putative 60S ribosomal protein L44
+Macromolecule #75: 60S ribosomal protein L37a
+Macromolecule #76: Putative ribosomal protein L24
+Macromolecule #77: Putative ribosomal protein L38
+Macromolecule #78: Putative 60S ribosomal protein L39
+Macromolecule #79: 60S ribosomal protein L41
+Macromolecule #80: Putative ribosomal protein L1a
+Macromolecule #82: Putative 60S ribosomal subunit protein L31
+Macromolecule #83: Ribosomal protein L37
+Macromolecule #15: E-site_tRNA_chain_S4
+Macromolecule #20: SSU_rRNA_chain_S1
+Macromolecule #41: LSUa_rRNA_chain_1
+Macromolecule #42: LSUb_rRNA_chain_2
+Macromolecule #43: SR2_chain_4
+Macromolecule #44: 5.8S_rRNA_chain_7
+Macromolecule #46: 5S_rRNA_chain_8
+Macromolecule #47: SR4_chain_5
+Macromolecule #58: SR6_chain_6
+Macromolecule #81: SR1_chain_3
+Macromolecule #84: MAGNESIUM ION
+Macromolecule #85: ZINC ION
+Macromolecule #86: SODIUM ION
+Macromolecule #87: POTASSIUM ION
+Macromolecule #88: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.6 Component:
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 1.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.46 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 345376 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)