+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Single Particle Cryo-EM structure of photosynthetic A2B2 glyceraldehyde 3-phosphate dehydrogenase from Spinacia oleracia | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Photosynthesis / Calvin-Benson cycle / redox regulation / glyceraldehyde-3-phosphate dehydrogenase / OXIDOREDUCTASE | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報glyceraldehyde-3-phosphate dehydrogenase (NADP+) (phosphorylating) / glyceraldehyde-3-phosphate dehydrogenase (NADP+) (phosphorylating) activity / reductive pentose-phosphate cycle / apoplast / glyceraldehyde-3-phosphate dehydrogenase (NAD+) (phosphorylating) activity / chloroplast / glucose metabolic process / NAD binding / NADP binding 類似検索 - 分子機能 | |||||||||
| 生物種 |  Spinacia oleracea (ホウレンソウ) Spinacia oleracea (ホウレンソウ) | |||||||||
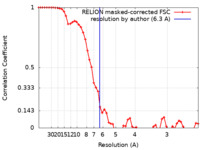
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 6.3 Å | |||||||||
 データ登録者 データ登録者 | Marotta R / Fermani S / Sparla F / Trost P / Del Giudice A | |||||||||
| 資金援助 |  フランス, 1件 フランス, 1件
| |||||||||
 引用 引用 |  ジャーナル: Acta Crystallogr D Struct Biol / 年: 2022 ジャーナル: Acta Crystallogr D Struct Biol / 年: 2022タイトル: Unravelling the regulation pathway of photosynthetic AB-GAPDH. 著者: Roberto Marotta / Alessandra Del Giudice / Libero Gurrieri / Silvia Fanti / Paolo Swuec / Luciano Galantini / Giuseppe Falini / Paolo Trost / Simona Fermani / Francesca Sparla /  要旨: Oxygenic phototrophs perform carbon fixation through the Calvin-Benson cycle. Different mechanisms adjust the cycle and the light-harvesting reactions to rapid environmental changes. Photosynthetic ...Oxygenic phototrophs perform carbon fixation through the Calvin-Benson cycle. Different mechanisms adjust the cycle and the light-harvesting reactions to rapid environmental changes. Photosynthetic glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is a key enzyme in the cycle. In land plants, different photosynthetic GAPDHs exist: the most abundant isoform is formed by AB heterotetramers and the least abundant by A homotetramers. Regardless of the subunit composition, GAPDH is the major consumer of photosynthetic NADPH and its activity is strictly regulated. While A-GAPDH is regulated by CP12, AB-GAPDH is autonomously regulated through the C-terminal extension (CTE) of its B subunits. Reversible inhibition of AB-GAPDH occurs via the oxidation of a cysteine pair located in the CTE and the substitution of NADP(H) with NAD(H) in the cofactor-binding site. These combined conditions lead to a change in the oligomerization state and enzyme inhibition. SEC-SAXS and single-particle cryo-EM analysis were applied to reveal the structural basis of this regulatory mechanism. Both approaches revealed that spinach (AB)-GAPDH oligomers with n = 1, 2, 4 and 5 co-exist in a dynamic system. B subunits mediate the contacts between adjacent tetramers in AB and AB oligomers. The CTE of each B subunit penetrates into the active site of a B subunit of the adjacent tetramer, which in turn moves its CTE in the opposite direction, effectively preventing the binding of the substrate 1,3-bisphosphoglycerate in the B subunits. The whole mechanism is made possible, and eventually controlled, by pyridine nucleotides. In fact, NAD(H), by removing NADP(H) from A subunits, allows the entrance of the CTE into the active site of the B subunit, hence stabilizing inhibited oligomers. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_13824.map.gz emd_13824.map.gz | 2.3 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-13824-v30.xml emd-13824-v30.xml emd-13824.xml emd-13824.xml | 12.3 KB 12.3 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_13824_fsc.xml emd_13824_fsc.xml | 5.5 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_13824.png emd_13824.png | 66.3 KB | ||
| Filedesc metadata |  emd-13824.cif.gz emd-13824.cif.gz | 5.6 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13824 http://ftp.pdbj.org/pub/emdb/structures/EMD-13824 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13824 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13824 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_13824_validation.pdf.gz emd_13824_validation.pdf.gz | 418.4 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_13824_full_validation.pdf.gz emd_13824_full_validation.pdf.gz | 417.9 KB | 表示 | |
| XML形式データ |  emd_13824_validation.xml.gz emd_13824_validation.xml.gz | 8.5 KB | 表示 | |
| CIF形式データ |  emd_13824_validation.cif.gz emd_13824_validation.cif.gz | 10.7 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13824 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13824 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13824 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13824 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_13824.map.gz / 形式: CCP4 / 大きさ: 12.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_13824.map.gz / 形式: CCP4 / 大きさ: 12.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ | X=Y=Z: 1.21 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : Photosynthetic A2B2 glyceraldehyde-3-phosphate dehydrogenase hete...
| 全体 | 名称: Photosynthetic A2B2 glyceraldehyde-3-phosphate dehydrogenase hetero-tetramer complexed with NAD. |
|---|---|
| 要素 |
|
-超分子 #1: Photosynthetic A2B2 glyceraldehyde-3-phosphate dehydrogenase hete...
| 超分子 | 名称: Photosynthetic A2B2 glyceraldehyde-3-phosphate dehydrogenase hetero-tetramer complexed with NAD. タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#2 |
|---|---|
| 由来(天然) | 生物種:  Spinacia oleracea (ホウレンソウ) Spinacia oleracea (ホウレンソウ) |
-分子 #1: Glyceraldehyde-3-phosphate dehydrogenase B, chloroplastic
| 分子 | 名称: Glyceraldehyde-3-phosphate dehydrogenase B, chloroplastic タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO EC番号: glyceraldehyde-3-phosphate dehydrogenase (NADP+) (phosphorylating) |
|---|---|
| 由来(天然) | 生物種:  Spinacia oleracea (ホウレンソウ) / 器官: chloroplast Spinacia oleracea (ホウレンソウ) / 器官: chloroplast |
| 分子量 | 理論値: 36.28866 KDa |
| 配列 | 文字列: KLKVAINGFG RIGRNFLRCW HGRKDSPLDV VVVNDSGGVK SATHLLKYDS ILGTFKADVK IIDNETFSID GKPIKVVSNR DPLKLPWAE LGIDIVIEGT GVFVDGPGAG KHIQAGAKKV IITAPAKGSD IPTYVVGVNE KDYGHDVANI ISNASCTTNC L APFVKVLD ...文字列: KLKVAINGFG RIGRNFLRCW HGRKDSPLDV VVVNDSGGVK SATHLLKYDS ILGTFKADVK IIDNETFSID GKPIKVVSNR DPLKLPWAE LGIDIVIEGT GVFVDGPGAG KHIQAGAKKV IITAPAKGSD IPTYVVGVNE KDYGHDVANI ISNASCTTNC L APFVKVLD EELGIVKGTM TTTHSYTGDQ RLLDASHRDL RRARAAALNI VPTSTGAAKA VSLVLPQLKG KLNGIALRVP TP NVSVVDL VVNIEKVGVT AEDVNNAFRK AAAGPLKGVL DVCDIPLVSV DFRCSDFSST IDSSLTMVMG GDMVKVVAWY DNE WGYSQR VVDLADLVAN KWP UniProtKB: Glyceraldehyde-3-phosphate dehydrogenase B, chloroplastic |
-分子 #2: Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic,Glycera...
| 分子 | 名称: Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic,Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic,Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic,Glyceraldehyde-3- ...名称: Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic,Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic,Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic,Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic タイプ: protein_or_peptide / ID: 2 / コピー数: 2 / 光学異性体: LEVO EC番号: glyceraldehyde-3-phosphate dehydrogenase (NADP+) (phosphorylating) |
|---|---|
| 由来(天然) | 生物種:  Spinacia oleracea (ホウレンソウ) / 器官: chloroplast Spinacia oleracea (ホウレンソウ) / 器官: chloroplast |
| 分子量 | 理論値: 36.256391 KDa |
| 配列 | 文字列: KLKVAINGFG RIGRNFLRCW HGRKDSPLDV VVINDTGGVK QASHLLKYDS ILGTFDADVK TAGDSAISVD GKVIKVVSDR NPVNLPWGD MGIDLVIEGT GVFVDRDGAG KHLQAGAKKV LITAPGKGDI PTYVVGVNEE GYTHADTIIS NASCTTNCLA P FVKVLDQK ...文字列: KLKVAINGFG RIGRNFLRCW HGRKDSPLDV VVINDTGGVK QASHLLKYDS ILGTFDADVK TAGDSAISVD GKVIKVVSDR NPVNLPWGD MGIDLVIEGT GVFVDRDGAG KHLQAGAKKV LITAPGKGDI PTYVVGVNEE GYTHADTIIS NASCTTNCLA P FVKVLDQK FGIIKGTMTT THSYTGDQRL LDASHRDLRR ARAACLNIVP TSTGAAKAVA LVLPNLKGKL NGIALRVPTP NV SVVDLVV QVSKKTFAEE VNAAFRESAD NELKGILSVC DEPLVSIDFR CTDVSSTIDS SLTMVMGDDM VKVIAWYDNE WGY SQRVVD LADIVANKWQ A UniProtKB: Glyceraldehyde-3-phosphate dehydrogenase A, chloroplastic |
-分子 #3: NICOTINAMIDE-ADENINE-DINUCLEOTIDE
| 分子 | 名称: NICOTINAMIDE-ADENINE-DINUCLEOTIDE / タイプ: ligand / ID: 3 / コピー数: 4 / 式: NAD |
|---|---|
| 分子量 | 理論値: 663.425 Da |
| Chemical component information |  ChemComp-NAD: |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| グリッド | モデル: C-flat-1.2/1.3 / 材質: COPPER / メッシュ: 400 |
| 凍結 | 凍結剤: ETHANE / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI POLARA 300 |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: COUNTING / 平均電子線量: 42.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 3.5 µm / 最小 デフォーカス(公称値): 1.5 µm |
| 実験機器 |  モデル: Tecnai Polara / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー