[English] 日本語
 Yorodumi
Yorodumi- EMDB-12142: ASCT2 in the presence of the inhibitor Lc-BPE in the outward-open... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12142 | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | ASCT2 in the presence of the inhibitor Lc-BPE in the outward-open conformation. | ||||||||||||||||||||||||
 Map data Map data | Cryo-EM map of ASCT2 in the presence of the inhibitor Lc-BPE at 3.43 A resolution in the outward-open conformation. Map was sharpened with a b factor of 189 A2 | ||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||
 Keywords Keywords | Solute carrier transporter / membrane protein / homology modeling / cancer metabolism / glutamine deprivation / cryo-EM | ||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationL-glutamine secretion / L-glutamine import across plasma membrane / L-glutamine transmembrane transporter activity / L-serine transmembrane transporter activity / L-glutamine transport / ligand-gated channel activity / L-aspartate transmembrane transporter activity / neutral amino acid transport / L-aspartate import across plasma membrane / neutral L-amino acid transmembrane transporter activity ...L-glutamine secretion / L-glutamine import across plasma membrane / L-glutamine transmembrane transporter activity / L-serine transmembrane transporter activity / L-glutamine transport / ligand-gated channel activity / L-aspartate transmembrane transporter activity / neutral amino acid transport / L-aspartate import across plasma membrane / neutral L-amino acid transmembrane transporter activity / Amino acid transport across the plasma membrane / symporter activity / amino acid transmembrane transporter activity / antiporter activity / RHOJ GTPase cycle / protein homotrimerization / RHOQ GTPase cycle / amino acid transport / RHOH GTPase cycle / RAC3 GTPase cycle / transport across blood-brain barrier / RAC1 GTPase cycle / basal plasma membrane / erythrocyte differentiation / centriolar satellite / melanosome / virus receptor activity / signaling receptor activity / ciliary basal body / extracellular exosome / membrane / metal ion binding / plasma membrane Similarity search - Function | ||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.43 Å | ||||||||||||||||||||||||
 Authors Authors | Garibsingh RA / Ndaru E | ||||||||||||||||||||||||
| Funding support |  Netherlands, Netherlands,  United States, 7 items United States, 7 items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2021 Journal: Proc Natl Acad Sci U S A / Year: 2021Title: Rational design of ASCT2 inhibitors using an integrated experimental-computational approach. Authors: Rachel-Ann A Garibsingh / Elias Ndaru / Alisa A Garaeva / Yueyue Shi / Laura Zielewicz / Paul Zakrepine / Massimiliano Bonomi / Dirk J Slotboom / Cristina Paulino / Christof Grewer / Avner Schlessinger /    Abstract: ASCT2 (SLC1A5) is a sodium-dependent neutral amino acid transporter that controls amino acid homeostasis in peripheral tissues. In cancer, ASCT2 is up-regulated where it modulates intracellular ...ASCT2 (SLC1A5) is a sodium-dependent neutral amino acid transporter that controls amino acid homeostasis in peripheral tissues. In cancer, ASCT2 is up-regulated where it modulates intracellular glutamine levels, fueling cell proliferation. Nutrient deprivation via ASCT2 inhibition provides a potential strategy for cancer therapy. Here, we rationally designed stereospecific inhibitors exploiting specific subpockets in the substrate binding site using computational modeling and cryo-electron microscopy (cryo-EM). The final structures combined with molecular dynamics simulations reveal multiple pharmacologically relevant conformations in the ASCT2 binding site as well as a previously unknown mechanism of stereospecific inhibition. Furthermore, this integrated analysis guided the design of a series of unique ASCT2 inhibitors. Our results provide a framework for future development of cancer therapeutics targeting nutrient transport via ASCT2, as well as demonstrate the utility of combining computational modeling and cryo-EM for solute carrier ligand discovery. | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12142.map.gz emd_12142.map.gz | 28.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12142-v30.xml emd-12142-v30.xml emd-12142.xml emd-12142.xml | 20.7 KB 20.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_12142.png emd_12142.png | 70.6 KB | ||
| Masks |  emd_12142_msk_1.map emd_12142_msk_1.map | 30.5 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-12142.cif.gz emd-12142.cif.gz | 6.7 KB | ||
| Others |  emd_12142_half_map_1.map.gz emd_12142_half_map_1.map.gz emd_12142_half_map_2.map.gz emd_12142_half_map_2.map.gz | 23.1 MB 23.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12142 http://ftp.pdbj.org/pub/emdb/structures/EMD-12142 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12142 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12142 | HTTPS FTP |
-Related structure data
| Related structure data |  7bcqMC  7bcsMC  7bctC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12142.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12142.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM map of ASCT2 in the presence of the inhibitor Lc-BPE at 3.43 A resolution in the outward-open conformation. Map was sharpened with a b factor of 189 A2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.012 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_12142_msk_1.map emd_12142_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half-map 1 used for post processing step and...
| File | emd_12142_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half-map 1 used for post processing step and FSC resolution calculation. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half-map 2 used for post processing step and...
| File | emd_12142_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half-map 2 used for post processing step and FSC resolution calculation. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : ASCT2 in the presence of the inhibitor Lc-BPE in the outward-open...
| Entire | Name: ASCT2 in the presence of the inhibitor Lc-BPE in the outward-open conformation. |
|---|---|
| Components |
|
-Supramolecule #1: ASCT2 in the presence of the inhibitor Lc-BPE in the outward-open...
| Supramolecule | Name: ASCT2 in the presence of the inhibitor Lc-BPE in the outward-open conformation. type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Neutral amino acid transporter B(0)
| Macromolecule | Name: Neutral amino acid transporter B(0) / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 56.638902 KDa |
| Recombinant expression | Organism:  Komagataella pastoris (fungus) Komagataella pastoris (fungus) |
| Sequence | String: MVADPPRDSK GLAAAEPTAN GGLALASIED QGAAAGGYCG SRDQVRRCLR ANLLVLLTVV AVVAGVALGL GVSGAGGALA LGPERLSAF VFPGELLLRL LRMIILPLVV CSLIGGAASL DPGALGRLGA WALLFFLVTT LLASALGVGL ALALQPGAAS A AINASVGA ...String: MVADPPRDSK GLAAAEPTAN GGLALASIED QGAAAGGYCG SRDQVRRCLR ANLLVLLTVV AVVAGVALGL GVSGAGGALA LGPERLSAF VFPGELLLRL LRMIILPLVV CSLIGGAASL DPGALGRLGA WALLFFLVTT LLASALGVGL ALALQPGAAS A AINASVGA AGSAENAPSK EVLDSFLDLA RNIFPSNLVS AAFRSYSTTY EERNITGTRV KVPVGQEVEG MNILGLVVFA IV FGVALRK LGPEGELLIR FFNSFNEATM VLVSWIMWYA PVGIMFLVAG KIVEMEDVGL LFARLGKYIL CCLLGHAIHG LLV LPLIYF LFTRKNPYRF LWGIVTPLAT AFGTSSSSAT LPLMMKCVEE NNGVAKHISR FILPIGATVN MDGAALFQCV AAVF IAQLS QQSLDFVKII TILVTATASS VGAAGIPAGG VLTLAIILEA VNLPVDHISL ILAVDWLVDR SCTVLNVEGD ALGAG LLQN YVDRTESRST EPELIQVKSE LPLDPLPVPT EEGNPLLKHY RGPAGDATVA SEKESVM UniProtKB: Neutral amino acid transporter B(0) |
-Macromolecule #2: 4-(4-phenylphenyl)carbonyloxypyrrolidine-2-carboxylic acid
| Macromolecule | Name: 4-(4-phenylphenyl)carbonyloxypyrrolidine-2-carboxylic acid type: ligand / ID: 2 / Number of copies: 3 / Formula: TG2 |
|---|---|
| Molecular weight | Theoretical: 311.332 Da |
| Chemical component information | 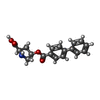 ChemComp-TG2: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Details: 20 mM Tris-HCl, pH 7.4, 200 mM NaCl |
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Details: at 5mA |
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 100 % / Chamber temperature: 288 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Temperature | Min: 90.0 K / Max: 105.0 K |
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3838 pixel / Digitization - Dimensions - Height: 3710 pixel / Digitization - Frames/image: 1-60 / Number grids imaged: 3 / Number real images: 6233 / Average exposure time: 9.0 sec. / Average electron dose: 53.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 2.0 µm / Calibrated defocus min: 0.3 µm / Calibrated magnification: 49407 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.3 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| Refinement | Space: REAL |
| Output model |  PDB-7bcq:  PDB-7bcs: |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)