+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Straight ADP-F-actin 1 | |||||||||
 Map data Map data | Main map, straight 1 ADP-F-actin | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationStriated Muscle Contraction / skeletal muscle thin filament assembly / striated muscle thin filament / skeletal muscle fiber development / stress fiber / actin filament / Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / hydrolase activity / ATP binding Similarity search - Function | |||||||||
| Biological species |   | |||||||||
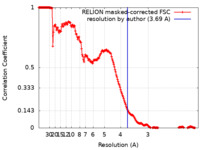
| Method | single particle reconstruction / cryo EM / Resolution: 3.69 Å | |||||||||
 Authors Authors | Reynolds MJ / Alushin GM | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2022 Journal: Nature / Year: 2022Title: Bending forces and nucleotide state jointly regulate F-actin structure. Authors: Matthew J Reynolds / Carla Hachicho / Ayala G Carl / Rui Gong / Gregory M Alushin /  Abstract: ATP-hydrolysis-coupled actin polymerization is a fundamental mechanism of cellular force generation. In turn, force and actin filament (F-actin) nucleotide state regulate actin dynamics by tuning F- ...ATP-hydrolysis-coupled actin polymerization is a fundamental mechanism of cellular force generation. In turn, force and actin filament (F-actin) nucleotide state regulate actin dynamics by tuning F-actin's engagement of actin-binding proteins through mechanisms that are unclear. Here we show that the nucleotide state of actin modulates F-actin structural transitions evoked by bending forces. Cryo-electron microscopy structures of ADP-F-actin and ADP-P-F-actin with sufficient resolution to visualize bound solvent reveal intersubunit interfaces bridged by water molecules that could mediate filament lattice flexibility. Despite extensive ordered solvent differences in the nucleotide cleft, these structures feature nearly identical lattices and essentially indistinguishable protein backbone conformations that are unlikely to be discriminable by actin-binding proteins. We next introduce a machine-learning-enabled pipeline for reconstructing bent filaments, enabling us to visualize both continuous structural variability and side-chain-level detail. Bent F-actin structures reveal rearrangements at intersubunit interfaces characterized by substantial alterations of helical twist and deformations in individual protomers, transitions that are distinct in ADP-F-actin and ADP-P-F-actin. This suggests that phosphate rigidifies actin subunits to alter the bending structural landscape of F-actin. As bending forces evoke nucleotide-state dependent conformational transitions of sufficient magnitude to be detected by actin-binding proteins, we propose that actin nucleotide state can serve as a co-regulator of F-actin mechanical regulation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27118.map.gz emd_27118.map.gz | 7.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27118-v30.xml emd-27118-v30.xml emd-27118.xml emd-27118.xml | 17.3 KB 17.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_27118_fsc.xml emd_27118_fsc.xml | 18.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_27118.png emd_27118.png | 99.4 KB | ||
| Masks |  emd_27118_msk_1.map emd_27118_msk_1.map | 512 MB |  Mask map Mask map | |
| Others |  emd_27118_half_map_1.map.gz emd_27118_half_map_1.map.gz emd_27118_half_map_2.map.gz emd_27118_half_map_2.map.gz | 411.6 MB 411.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27118 http://ftp.pdbj.org/pub/emdb/structures/EMD-27118 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27118 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27118 | HTTPS FTP |
-Validation report
| Summary document |  emd_27118_validation.pdf.gz emd_27118_validation.pdf.gz | 775 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_27118_full_validation.pdf.gz emd_27118_full_validation.pdf.gz | 774.6 KB | Display | |
| Data in XML |  emd_27118_validation.xml.gz emd_27118_validation.xml.gz | 23.3 KB | Display | |
| Data in CIF |  emd_27118_validation.cif.gz emd_27118_validation.cif.gz | 30.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27118 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27118 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27118 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27118 | HTTPS FTP |
-Related structure data
| Related structure data |  8d17MC  8d13C  8d14C  8d15C  8d16C  8d18C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
| EM raw data |  EMPIAR-11128 (Title: Cryo-EM of ADP-F-actin / Data size: 938.5 EMPIAR-11128 (Title: Cryo-EM of ADP-F-actin / Data size: 938.5 Data #1: Unaligned multi-frame micrographs of ADP-F-actin [micrographs - multiframe] Data #2: Polished single-frame particles of bent ADP-F-actin segments [picked particles - single frame - processed] Data #3: Polished single-frame particles of straight ADP-F-actin segments, Subset 1 [picked particles - single frame - processed] Data #4: Polished single-frame particles of straight ADP-F-actin segments, Subset 2 [picked particles - single frame - processed] Data #5: Polished single-frame particles of helical ADP-F-actin segments, for high-resolution [picked particles - single frame - processed]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_27118.map.gz / Format: CCP4 / Size: 8.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27118.map.gz / Format: CCP4 / Size: 8.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Main map, straight 1 ADP-F-actin | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.03 Å | ||||||||||||||||||||||||||||||||||||
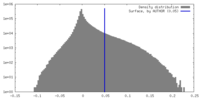
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_27118_msk_1.map emd_27118_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
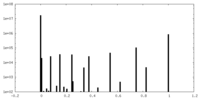
| Density Histograms |
-Half map: Half-map 1, straight 1 ADP-F-actin
| File | emd_27118_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map 1, straight 1 ADP-F-actin | ||||||||||||
| Projections & Slices |
| ||||||||||||
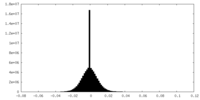
| Density Histograms |
-Half map: Half-map 2, straight 1 ADP-F-actin
| File | emd_27118_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map 2, straight 1 ADP-F-actin | ||||||||||||
| Projections & Slices |
| ||||||||||||
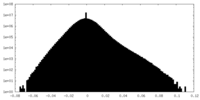
| Density Histograms |
- Sample components
Sample components
-Entire : Straight F-actin 1, ADP nucleotide state
| Entire | Name: Straight F-actin 1, ADP nucleotide state |
|---|---|
| Components |
|
-Supramolecule #1: Straight F-actin 1, ADP nucleotide state
| Supramolecule | Name: Straight F-actin 1, ADP nucleotide state / type: complex / Chimera: Yes / ID: 1 / Parent: 0 / Macromolecule list: #1 Details: Mechanically deformed filamentous actin in the aged ADP state |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 15.1 kDa/nm |
-Macromolecule #1: Actin, alpha skeletal muscle
| Macromolecule | Name: Actin, alpha skeletal muscle / type: protein_or_peptide / ID: 1 / Number of copies: 7 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 42.109973 KDa |
| Sequence | String: MCDEDETTAL VCDNGSGLVK AGFAGDDAPR AVFPSIVGRP RHQGVMVGMG QKDSYVGDEA QSKRGILTLK YPIE(HIC)G IIT NWDDMEKIWH HTFYNELRVA PEEHPTLLTE APLNPKANRE KMTQIMFETF NVPAMYVAIQ AVLSLYASGR TTGIVLD SG DGVTHNVPIY ...String: MCDEDETTAL VCDNGSGLVK AGFAGDDAPR AVFPSIVGRP RHQGVMVGMG QKDSYVGDEA QSKRGILTLK YPIE(HIC)G IIT NWDDMEKIWH HTFYNELRVA PEEHPTLLTE APLNPKANRE KMTQIMFETF NVPAMYVAIQ AVLSLYASGR TTGIVLD SG DGVTHNVPIY EGYALPHAIM RLDLAGRDLT DYLMKILTER GYSFVTTAER EIVRDIKEKL CYVALDFENE MATAASSS S LEKSYELPDG QVITIGNERF RCPETLFQPS FIGMESAGIH ETTYNSIMKC DIDIRKDLYA NNVMSGGTTM YPGIADRMQ KEITALAPST MKIKIIAPPE RKYSVWIGGS ILASLSTFQQ MWITKQEYDE AGPSIVHRKC F |
-Macromolecule #2: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 7 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Macromolecule #3: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 3 / Number of copies: 7 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-40 / Number grids imaged: 1 / Average exposure time: 10.0 sec. / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 4.0 µm / Nominal defocus min: 2.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller












 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)