+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-23306 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of human Apo CNGA1 channel in K+/Ca2+ | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ALPHA-HELICAL / MEMBRANE PROTEIN / ION CHANNEL | |||||||||
| Function / homology |  Function and homology information Function and homology informationintracellular cyclic nucleotide activated cation channel complex / intracellularly cGMP-activated cation channel activity / intracellularly cAMP-activated cation channel activity / rod photoreceptor outer segment / photoreceptor outer segment membrane / sodium ion transport / sodium channel activity / monoatomic cation transmembrane transport / cGMP binding / cAMP binding ...intracellular cyclic nucleotide activated cation channel complex / intracellularly cGMP-activated cation channel activity / intracellularly cAMP-activated cation channel activity / rod photoreceptor outer segment / photoreceptor outer segment membrane / sodium ion transport / sodium channel activity / monoatomic cation transmembrane transport / cGMP binding / cAMP binding / visual perception / calcium channel activity / Activation of the phototransduction cascade / calcium ion transport / Inactivation, recovery and regulation of the phototransduction cascade / protein-containing complex binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
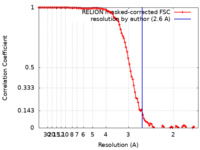
| Method | single particle reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||
 Authors Authors | Xue J / Han Y | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Neuron / Year: 2021 Journal: Neuron / Year: 2021Title: Structural mechanisms of gating and selectivity of human rod CNGA1 channel. Authors: Jing Xue / Yan Han / Weizhong Zeng / Yan Wang / Youxing Jiang /  Abstract: Mammalian cyclic nucleotide-gated (CNG) channels play an essential role in the signal transduction of the visual and olfactory sensory systems. Here we reveal the structural mechanism of ligand ...Mammalian cyclic nucleotide-gated (CNG) channels play an essential role in the signal transduction of the visual and olfactory sensory systems. Here we reveal the structural mechanism of ligand gating in human rod CNGA1 channel by determining its cryo-EM structures in both the apo closed and cGMP-bound open states. Distinct from most other members of voltage-gated tetrameric cation channels, CNGA1 forms a central channel gate in the middle of the membrane, occluding the central cavity. Structural analyses of ion binding profiles in the selectivity filters of the wild-type channel and the E365Q filter mutant allow us to unambiguously define the two Ca binding sites inside the selectivity filter, providing structural insights into Ca blockage and permeation in CNG channels. The structure of the E365Q mutant also reveals two alternative side-chain conformations at Q365, providing a plausible explanation for the voltage-dependent gating of CNG channel acquired upon E365 mutation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_23306.map.gz emd_23306.map.gz | 85.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-23306-v30.xml emd-23306-v30.xml emd-23306.xml emd-23306.xml | 10.8 KB 10.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_23306_fsc.xml emd_23306_fsc.xml | 10.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_23306.png emd_23306.png | 181.5 KB | ||
| Filedesc metadata |  emd-23306.cif.gz emd-23306.cif.gz | 5.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-23306 http://ftp.pdbj.org/pub/emdb/structures/EMD-23306 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23306 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23306 | HTTPS FTP |
-Related structure data
| Related structure data |  7lftMC  7lfwC  7lfxC  7lfyC  7lg1C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10722 (Title: Cryo-EM structure of human rod CNGA1 channel in apo-state EMPIAR-10722 (Title: Cryo-EM structure of human rod CNGA1 channel in apo-stateData size: 84.9 Data #1: Final particle stack of the CNGA1 channel in apo state [picked particles - multiframe - processed]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_23306.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_23306.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.844 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Apo CNGA1 homotetramer in K+/Ca2+
| Entire | Name: Apo CNGA1 homotetramer in K+/Ca2+ |
|---|---|
| Components |
|
-Supramolecule #1: Apo CNGA1 homotetramer in K+/Ca2+
| Supramolecule | Name: Apo CNGA1 homotetramer in K+/Ca2+ / type: cell / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: cGMP-gated cation channel alpha-1
| Macromolecule | Name: cGMP-gated cation channel alpha-1 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 64.650434 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MDYKDDDDKG GSASKDKKEE EKKEVVVIDP SGNTYYNWLF CITLPVMYNW TMVIARACFD ELQSDYLEYW LILDYVSDIV YLIDMFVRT RTGYLEQGLL VKEELKLINK YKSNLQFKLD VLSLIPTDLL YFKLGWNYPE IRLNRLLRFS RMFEFFQRTE T RTNYPNIF ...String: MDYKDDDDKG GSASKDKKEE EKKEVVVIDP SGNTYYNWLF CITLPVMYNW TMVIARACFD ELQSDYLEYW LILDYVSDIV YLIDMFVRT RTGYLEQGLL VKEELKLINK YKSNLQFKLD VLSLIPTDLL YFKLGWNYPE IRLNRLLRFS RMFEFFQRTE T RTNYPNIF RISNLVMYIV IIIHWNACVF YSISKAIGFG NDTWVYPDIN DPEFGRLARK YVYSLYWSTL TLTTIGETPP PV RDSEYVF VVVDFLIGVL IFATIVGNIG SMISNMNAAR AEFQARIDAI KQYMHFRNVS KDMEKRVIKW FDYLWTNKKT VDE KEVLKY LPDKLRAEIA INVHLDTLKK VRIFADCEAG LLVELVLKLQ PQVYSPGDYI CKKGDIGREM YIIKEGKLAV VADD GVTQF VVLSDGSYFG EISILNIKGS KAGNRRTANI KSIGYSDLFC LSKDDLMEAL TEYPDAKTML EEKGKQILMK DGLLD LNIA NAGSDPKDLE EKVTRMEGSV DLLQTRFARI LAEYESMQQK LKQRLTKVEK FLKPLIDTEF SSIEGPGAES GPIDST UniProtKB: Cyclic nucleotide-gated channel alpha-1 |
-Macromolecule #2: CHOLESTEROL
| Macromolecule | Name: CHOLESTEROL / type: ligand / ID: 2 / Number of copies: 8 / Formula: CLR |
|---|---|
| Molecular weight | Theoretical: 386.654 Da |
| Chemical component information |  ChemComp-CLR: |
-Macromolecule #3: 1-PALMITOYL-2-LINOLEOYL-SN-GLYCERO-3-PHOSPHOCHOLINE
| Macromolecule | Name: 1-PALMITOYL-2-LINOLEOYL-SN-GLYCERO-3-PHOSPHOCHOLINE / type: ligand / ID: 3 / Number of copies: 24 / Formula: CPL |
|---|---|
| Molecular weight | Theoretical: 758.06 Da |
| Chemical component information |  ChemComp-CPL: |
-Macromolecule #4: POTASSIUM ION
| Macromolecule | Name: POTASSIUM ION / type: ligand / ID: 4 / Number of copies: 3 / Formula: K |
|---|---|
| Molecular weight | Theoretical: 39.098 Da |
-Macromolecule #5: water
| Macromolecule | Name: water / type: ligand / ID: 5 / Number of copies: 4 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 1.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)