+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
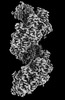
| タイトル | RAD51 filament on dsDNA bound by the BRCA2 c-terminus | |||||||||
 マップデータ マップデータ | RAD51 filament on dsDNA bound by the BRCA2 c-terminus | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | RAD51 / BRCA2 / Filament / Complex / RECOMBINATION | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報BRCA2-MAGE-D1 complex / negative regulation of mammary gland epithelial cell proliferation / presynaptic intermediate filament cytoskeleton / mitotic recombination-dependent replication fork processing / cellular response to camptothecin / chromosome organization involved in meiotic cell cycle / establishment of protein localization to telomere / telomere maintenance via telomere lengthening / positive regulation of DNA ligation / Impaired BRCA2 translocation to the nucleus ...BRCA2-MAGE-D1 complex / negative regulation of mammary gland epithelial cell proliferation / presynaptic intermediate filament cytoskeleton / mitotic recombination-dependent replication fork processing / cellular response to camptothecin / chromosome organization involved in meiotic cell cycle / establishment of protein localization to telomere / telomere maintenance via telomere lengthening / positive regulation of DNA ligation / Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / double-strand break repair involved in meiotic recombination / nuclear ubiquitin ligase complex / cellular response to hydroxyurea / replication-born double-strand break repair via sister chromatid exchange / lateral element / telomere maintenance via recombination / DNA recombinase assembly / histone H4 acetyltransferase activity / histone H3 acetyltransferase activity / regulation of DNA damage checkpoint / mitotic recombination / Impaired BRCA2 binding to PALB2 / DNA strand invasion / HDR through MMEJ (alt-NHEJ) / gamma-tubulin binding / DNA strand exchange activity / reciprocal meiotic recombination / DNA repair complex / Defective homologous recombination repair (HRR) due to BRCA1 loss of function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA1 binding function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA2/RAD51/RAD51C binding function / Homologous DNA Pairing and Strand Exchange / Resolution of D-loop Structures through Synthesis-Dependent Strand Annealing (SDSA) / response to UV-C / oocyte maturation / Resolution of D-loop Structures through Holliday Junction Intermediates / single-stranded DNA helicase activity / inner cell mass cell proliferation / ATP-dependent DNA damage sensor activity / DNA damage response, signal transduction by p53 class mediator resulting in transcription of p21 class mediator / hematopoietic stem cell proliferation / HDR through Single Strand Annealing (SSA) / Impaired BRCA2 binding to RAD51 / regulation of double-strand break repair via homologous recombination / nuclear chromosome / female gonad development / replication fork processing / male meiosis I / DNA unwinding involved in DNA replication / Transcriptional Regulation by E2F6 / Presynaptic phase of homologous DNA pairing and strand exchange / centrosome duplication / response to X-ray / intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator / ATP-dependent activity, acting on DNA / interstrand cross-link repair / DNA polymerase binding / condensed chromosome / positive regulation of mitotic cell cycle / meiotic cell cycle / regulation of cytokinesis / condensed nuclear chromosome / secretory granule / male germ cell nucleus / cellular response to ionizing radiation / nucleotide-excision repair / response to gamma radiation / regulation of protein phosphorylation / double-strand break repair via homologous recombination / HDR through Homologous Recombination (HRR) / brain development / PML body / Meiotic recombination / cellular senescence / double-strand break repair / site of double-strand break / single-stranded DNA binding / spermatogenesis / double-stranded DNA binding / DNA recombination / protease binding / chromosome, telomeric region / mitochondrial matrix / DNA repair / centrosome / DNA damage response / chromatin binding / regulation of DNA-templated transcription / chromatin / nucleolus / positive regulation of DNA-templated transcription / perinuclear region of cytoplasm / enzyme binding / ATP hydrolysis activity / protein-containing complex / mitochondrion / nucleoplasm / ATP binding / identical protein binding 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) / synthetic construct (人工物) Homo sapiens (ヒト) / synthetic construct (人工物) | |||||||||
| 手法 | らせん対称体再構成法 / クライオ電子顕微鏡法 / 解像度: 2.83 Å | |||||||||
 データ登録者 データ登録者 | Appleby R / Pellegrini L | |||||||||
| 資金援助 |  英国, 2件 英国, 2件
| |||||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2023 ジャーナル: Nat Commun / 年: 2023タイトル: Structural basis for stabilisation of the RAD51 nucleoprotein filament by BRCA2. 著者: Robert Appleby / Luay Joudeh / Katie Cobbett / Luca Pellegrini /  要旨: The BRCA2 tumour suppressor protein preserves genomic integrity via interactions with the DNA-strand exchange RAD51 protein in homology-directed repair. The RAD51-binding TR2 motif at the BRCA2 C- ...The BRCA2 tumour suppressor protein preserves genomic integrity via interactions with the DNA-strand exchange RAD51 protein in homology-directed repair. The RAD51-binding TR2 motif at the BRCA2 C-terminus is essential for protection and restart of stalled replication forks. Biochemical evidence shows that TR2 recognises filamentous RAD51, but existing models of TR2 binding to RAD51 lack a structural basis. Here we used cryo-electron microscopy and structure-guided mutagenesis to elucidate the mechanism of TR2 binding to nucleoprotein filaments of human RAD51. We find that TR2 binds across the protomer interface in the filament, acting as a brace for adjacent RAD51 molecules. TR2 targets an acidic-patch motif on human RAD51 that serves as a recruitment hub in fission yeast Rad51 for recombination mediators Rad52 and Rad55-Rad57. Our findings provide a structural rationale for RAD51 filament stabilisation by BRCA2 and reveal a common recruitment mechanism of recombination mediators to the RAD51 filament. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_17585.map.gz emd_17585.map.gz | 28.6 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-17585-v30.xml emd-17585-v30.xml emd-17585.xml emd-17585.xml | 19.6 KB 19.6 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_17585.png emd_17585.png | 76.8 KB | ||
| Filedesc metadata |  emd-17585.cif.gz emd-17585.cif.gz | 6.1 KB | ||
| その他 |  emd_17585_half_map_1.map.gz emd_17585_half_map_1.map.gz emd_17585_half_map_2.map.gz emd_17585_half_map_2.map.gz | 23.5 MB 23.5 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17585 http://ftp.pdbj.org/pub/emdb/structures/EMD-17585 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17585 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17585 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_17585_validation.pdf.gz emd_17585_validation.pdf.gz | 814.4 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_17585_full_validation.pdf.gz emd_17585_full_validation.pdf.gz | 813.9 KB | 表示 | |
| XML形式データ |  emd_17585_validation.xml.gz emd_17585_validation.xml.gz | 10.8 KB | 表示 | |
| CIF形式データ |  emd_17585_validation.cif.gz emd_17585_validation.cif.gz | 12.6 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17585 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17585 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17585 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17585 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8pbdMC  8pbcC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_17585.map.gz / 形式: CCP4 / 大きさ: 30.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_17585.map.gz / 形式: CCP4 / 大きさ: 30.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RAD51 filament on dsDNA bound by the BRCA2 c-terminus | ||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.304 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: Half-map 1
| ファイル | emd_17585_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half-map 1 | ||||||||||||
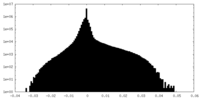
| 投影像・断面図 |
| ||||||||||||
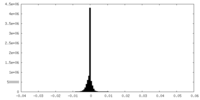
| 密度ヒストグラム |
-ハーフマップ: Half-map 2
| ファイル | emd_17585_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half-map 2 | ||||||||||||
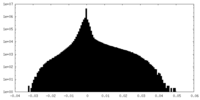
| 投影像・断面図 |
| ||||||||||||
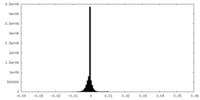
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
+全体 : RAD51 filament formed on dsDNA and bound with a peptide correspon...
+超分子 #1: RAD51 filament formed on dsDNA and bound with a peptide correspon...
+超分子 #2: DNA repair protein RAD51 homolog 1
+超分子 #3: Breast cancer type 2 susceptibility protein
+超分子 #4: DNA strand 1 and 2
+分子 #1: DNA repair protein RAD51 homolog 1
+分子 #2: Breast cancer type 2 susceptibility protein
+分子 #3: DNA strand 1
+分子 #4: DNA strand 2
+分子 #5: ADENOSINE-5'-TRIPHOSPHATE
+分子 #6: CALCIUM ION
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | らせん対称体再構成法 |
| 試料の集合状態 | filament |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| 凍結 | 凍結剤: ETHANE / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 平均電子線量: 52.6 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 0.9 µm / 倍率(公称値): 130000 |
| 試料ステージ | ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 最終 再構成 | 想定した対称性 - らせんパラメータ - Δz: 15.8 Å 想定した対称性 - らせんパラメータ - ΔΦ: 56.0 ° 想定した対称性 - らせんパラメータ - 軸対称性: C1 (非対称) 解像度のタイプ: BY AUTHOR / 解像度: 2.83 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: RELION (ver. 3.1) / 使用した粒子像数: 1 |
|---|---|
| 初期モデル | モデルのタイプ: OTHER |
| 最終 角度割当 | タイプ: NOT APPLICABLE / ソフトウェア - 名称: RELION (ver. 3.1) |
-原子モデル構築 1
| 精密化 | 空間: REAL / プロトコル: OTHER |
|---|---|
| 得られたモデル |  PDB-8pbd: |
 ムービー
ムービー コントローラー
コントローラー













 Z
Z Y
Y X
X