+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Hexameric human IgG3 Fc complex | ||||||||||||
 Map data Map data | Subtomogram average of IgG3-Fc hexamer formed on lipid bilayers | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | IgG3 / antibody / Fc hexamer / IMMUNE SYSTEM | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationimmunoglobulin complex / adaptive immune response / extracellular region / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
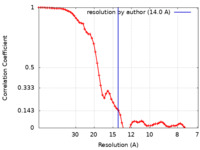
| Method | subtomogram averaging / cryo EM / Resolution: 14.0 Å | ||||||||||||
 Authors Authors | Abendstein L / Sharp TH | ||||||||||||
| Funding support | European Union,  Netherlands, 3 items Netherlands, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Complement is activated by elevated IgG3 hexameric platforms and deposits C4b onto distinct antibody domains. Authors: Leoni Abendstein / Douwe J Dijkstra / Rayman T N Tjokrodirijo / Peter A van Veelen / Leendert A Trouw / Paul J Hensbergen / Thomas H Sharp /  Abstract: IgG3 is unique among the IgG subclasses due to its extended hinge, allotypic diversity and enhanced effector functions, including highly efficient pathogen neutralisation and complement activation. ...IgG3 is unique among the IgG subclasses due to its extended hinge, allotypic diversity and enhanced effector functions, including highly efficient pathogen neutralisation and complement activation. It is also underrepresented as an immunotherapeutic candidate, partly due to a lack of structural information. Here, we use cryoEM to solve structures of antigen-bound IgG3 alone and in complex with complement components. These structures reveal a propensity for IgG3-Fab clustering, which is possible due to the IgG3-specific flexible upper hinge region and may maximise pathogen neutralisation by forming high-density antibody arrays. IgG3 forms elevated hexameric Fc platforms that extend above the protein corona to maximise binding to receptors and the complement C1 complex, which here adopts a unique protease conformation that may precede C1 activation. Mass spectrometry reveals that C1 deposits C4b directly onto specific IgG3 residues proximal to the Fab domains. Structural analysis shows this to be caused by the height of the C1-IgG3 complex. Together, these data provide structural insights into the role of the unique IgG3 extended hinge, which will aid the development and design of upcoming immunotherapeutics based on IgG3. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_16227.map.gz emd_16227.map.gz | 1.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-16227-v30.xml emd-16227-v30.xml emd-16227.xml emd-16227.xml | 16.3 KB 16.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_16227_fsc.xml emd_16227_fsc.xml | 8.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_16227.png emd_16227.png | 179.5 KB | ||
| Masks |  emd_16227_msk_1.map emd_16227_msk_1.map | 22.2 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-16227.cif.gz emd-16227.cif.gz | 5.6 KB | ||
| Others |  emd_16227_half_map_1.map.gz emd_16227_half_map_1.map.gz emd_16227_half_map_2.map.gz emd_16227_half_map_2.map.gz | 16.8 MB 17 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-16227 http://ftp.pdbj.org/pub/emdb/structures/EMD-16227 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16227 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16227 | HTTPS FTP |
-Related structure data
| Related structure data |  8btbMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_16227.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_16227.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Subtomogram average of IgG3-Fc hexamer formed on lipid bilayers | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 3.48 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_16227_msk_1.map emd_16227_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map of subtomogram average of IgG3-Fc hexamer...
| File | emd_16227_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map of subtomogram average of IgG3-Fc hexamer formed on lipid bilayers | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map of subtomogram average of IgG3-Fc hexamer...
| File | emd_16227_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map of subtomogram average of IgG3-Fc hexamer formed on lipid bilayers | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Fc domain of human IgG3
| Entire | Name: Fc domain of human IgG3 |
|---|---|
| Components |
|
-Supramolecule #1: Fc domain of human IgG3
| Supramolecule | Name: Fc domain of human IgG3 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: FLJ00385 protein (Fragment)
| Macromolecule | Name: FLJ00385 protein (Fragment) / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.768957 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: LGGPSVFLFP PKPKDTLMIS RTPEVTCVVV DVSHEDPEVQ FKWYVDGVEV HNAKTKPREE QFNSTFRVVS VLTVLHQDWL NGKEYKCKV SNKALPAPIE KTISKTKGQP REPQVYTLPP SREEMTKNQV SLTCLVKGFY PSDIAVEWES SGQPENNYNT T PPMLDSDG ...String: LGGPSVFLFP PKPKDTLMIS RTPEVTCVVV DVSHEDPEVQ FKWYVDGVEV HNAKTKPREE QFNSTFRVVS VLTVLHQDWL NGKEYKCKV SNKALPAPIE KTISKTKGQP REPQVYTLPP SREEMTKNQV SLTCLVKGFY PSDIAVEWES SGQPENNYNT T PPMLDSDG SFFLYSKLTV DKSRWQQGNI FSCSVMHEAL HNRFTQKSLS L UniProtKB: FLJ00385 protein |
-Macromolecule #2: FLJ00385 protein (Fragment)
| Macromolecule | Name: FLJ00385 protein (Fragment) / type: protein_or_peptide / ID: 2 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.782943 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: GPSVFLFPPK PKDTLMISRT PEVTCVVVDV SHEDPEVQFK WYVDGVEVHN AKTKPREEQF NSTFRVVSVL TVLHQDWLNG KEYKCKVSN KALPAPIEKT ISKTKGQPRE PQVYTLPPSR EEMTKNQVSL TCLVKGFYPS DIAVEWESSG QPENNYNTTP P MLDSDGSF ...String: GPSVFLFPPK PKDTLMISRT PEVTCVVVDV SHEDPEVQFK WYVDGVEVHN AKTKPREEQF NSTFRVVSVL TVLHQDWLNG KEYKCKVSN KALPAPIEKT ISKTKGQPRE PQVYTLPPSR EEMTKNQVSL TCLVKGFYPS DIAVEWESSG QPENNYNTTP P MLDSDGSF FLYSKLTVDK SRWQQGNIFS CSVMHEALHN RFTQKSLSLS P UniProtKB: FLJ00385 protein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | subtomogram averaging |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 / Details: PBS |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 1.5 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 6.0 µm / Nominal defocus min: 3.0 µm |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)