+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | ubiquitinated DNA-bound FANCD2-FANCI3D | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | nucleic acid protein complex / DNA clamp / solenoid domains / Fanconi anemia / DNA repair / DNA damage / DNA interstrand crosslink / DNA BINDING PROTEIN / ubiquitination | ||||||||||||
| Biological species |   Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | ||||||||||||
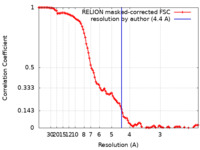
| Method | single particle reconstruction / cryo EM / Resolution: 4.4 Å | ||||||||||||
 Authors Authors | Passmore LA / Sijacki T / Alcon P | ||||||||||||
| Funding support |  United Kingdom, United Kingdom,  Germany, European Union, 3 items Germany, European Union, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2022 Journal: Nat Struct Mol Biol / Year: 2022Title: The DNA-damage kinase ATR activates the FANCD2-FANCI clamp by priming it for ubiquitination. Authors: Tamara Sijacki / Pablo Alcón / Zhuo A Chen / Stephen H McLaughlin / Shabih Shakeel / Juri Rappsilber / Lori A Passmore /    Abstract: DNA interstrand cross-links are tumor-inducing lesions that block DNA replication and transcription. When cross-links are detected at stalled replication forks, ATR kinase phosphorylates FANCI, which ...DNA interstrand cross-links are tumor-inducing lesions that block DNA replication and transcription. When cross-links are detected at stalled replication forks, ATR kinase phosphorylates FANCI, which stimulates monoubiquitination of the FANCD2-FANCI clamp by the Fanconi anemia core complex. Monoubiquitinated FANCD2-FANCI is locked onto DNA and recruits nucleases that mediate DNA repair. However, it remains unclear how phosphorylation activates this pathway. Here, we report structures of FANCD2-FANCI complexes containing phosphomimetic FANCI. We observe that, unlike wild-type FANCD2-FANCI, the phosphomimetic complex closes around DNA, independent of the Fanconi anemia core complex. The phosphomimetic mutations do not substantially alter DNA binding but instead destabilize the open state of FANCD2-FANCI and alter its conformational dynamics. Overall, our results demonstrate that phosphorylation primes the FANCD2-FANCI clamp for ubiquitination, showing how multiple posttranslational modifications are coordinated to control DNA repair. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15103.map.gz emd_15103.map.gz | 80.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15103-v30.xml emd-15103-v30.xml emd-15103.xml emd-15103.xml | 28.8 KB 28.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_15103_fsc.xml emd_15103_fsc.xml | 10.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_15103.png emd_15103.png | 45.4 KB | ||
| Filedesc metadata |  emd-15103.cif.gz emd-15103.cif.gz | 7.7 KB | ||
| Others |  emd_15103_additional_1.map.gz emd_15103_additional_1.map.gz emd_15103_additional_2.map.gz emd_15103_additional_2.map.gz emd_15103_half_map_1.map.gz emd_15103_half_map_1.map.gz emd_15103_half_map_2.map.gz emd_15103_half_map_2.map.gz | 8.8 MB 96.3 MB 80.9 MB 81.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15103 http://ftp.pdbj.org/pub/emdb/structures/EMD-15103 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15103 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15103 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_15103.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15103.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.145 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_15103_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: #2
| File | emd_15103_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_15103_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_15103_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : ubiquitinated DNA-bound FANCD2-FANCI that contains phosphomimetic...
| Entire | Name: ubiquitinated DNA-bound FANCD2-FANCI that contains phosphomimetic FANCI |
|---|---|
| Components |
|
-Supramolecule #1: ubiquitinated DNA-bound FANCD2-FANCI that contains phosphomimetic...
| Supramolecule | Name: ubiquitinated DNA-bound FANCD2-FANCI that contains phosphomimetic FANCI type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 349 KDa |
-Supramolecule #2: FANCD2
| Supramolecule | Name: FANCD2 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|
-Supramolecule #3: FANCI3D
| Supramolecule | Name: FANCI3D / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2-#4 |
|---|
-Supramolecule #4: Polyubiquitin-C
| Supramolecule | Name: Polyubiquitin-C / type: complex / ID: 4 / Parent: 1 / Macromolecule list: #5 |
|---|
-Macromolecule #1: FANCD2
| Macromolecule | Name: FANCD2 / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MVSKRKLSKI DAAEESSKTD LQSRCPETKR SRISDKRAPS QGGLENEGVF EELLRTSGII LKVGEGQNEI AVDQTAFQKK LRVALEKHP SYPGVVNEFI SGLESHIKDR SQFKNCLLPC TPARTEGSRT LVHSYCESLI KLLLGIKILQ PAVVTLLLEK I PEFFFDVV ...String: MVSKRKLSKI DAAEESSKTD LQSRCPETKR SRISDKRAPS QGGLENEGVF EELLRTSGII LKVGEGQNEI AVDQTAFQKK LRVALEKHP SYPGVVNEFI SGLESHIKDR SQFKNCLLPC TPARTEGSRT LVHSYCESLI KLLLGIKILQ PAVVTLLLEK I PEFFFDVV GTFGTNFPRL IVNQFKWLDG LLDSQDLVKK LMQMLSVSPV PIQHDIITSL PEILEDSQQN EVARELSCLL KQ GRRLTVP ILDALSRLDL DAELLAKVRQ SAMTIVPSVK LEDLPVVIKF ILHNVKAADA VEVISDLRKS LDLSSCVLPL QLL GSQRKL KSQAQASSSM SQVTTSQNCV KLLFDVIKLA VRFQKDVSEA WIKAIENSTS VSDHKVLDLI VLLLIHSTNS KNRK QTEKV LRSKIRLGCM PEQLMQNAFQ NHSMVIKDFF PSILSLAQTF LHSAHPAVVS FGSCMYKQAF AVFDSYCQQE VVCAL VTHV CSGNETELDI SLDVLTDLVI LHPSLLLRYA TFVKTILDSM QKLNPCQIRK LFYILSTLAF SQRQEGSYIQ DDMHMV IRK WLSSSVPNHK QMGIIGAVTM MGSVALKRNE ADGGLLERPE LSIECDGQLS TLLDLVGFCC EQTPEVLALY YDELANL IE KQKGNLDLQL LDKFGKSLVE DFPNDFVVDL SPTVDGSFLF PVKSLYNLDE DETQGAIAIN LLPLVSQSEP GRVADEMS N SRKRVVSPIC LSPCFRLLRL YTGEQNNGSL EEIDALLGCP LYLTDLEVEG KLDSLSKQER EFLCSLLFYA LNWFREVVN AFCQQQDAEM KGKVLTRLQN ITELQNVLGK CLAATPGYVP PPATFDSEAP EGVPSINAGG PVRKKNGKKR KSDSSKACSA ERTQADESS DGNQPDTELS ELEKSAAEKE TGNPLAQLQS YRPYFRELDL EVFSVLHCGL LTKSILDTEM HTEASEVVQL G PAELCFLL DDMCWKLEHV LTPGSTRRVP FLKERGNKDV GFSHLCQRSP KEVAVCVVKL LKPLCNHMEN MHNYFQTVIP NQ GVVDESG LNIQEYQLMS SCYHQLLLAF RLLFAWSGFS QHENSNLLRS ALQVLADRLK PGETEFLPLE ELISESFQYL LNF QASIPS FQCAFILTQV LMAISEKPMT GWKREKMASL AKQFLCQSWM KPGGDREKGS HFNSALHTLL CVYLEHTDNI LKAI EEISS VGVPELINSA KDGCSSTYPT LSRQTFPVFF RVMMAQLESS VKSIPAGKPS DSGEVQLEKL LKWNIAVRNF HILIN LVKV FDSRPVLSIC LKYGRLFVEA FLKLAMPLLD HSFKKHRDDV QSLLKTLQLS TRQLHHMCGH SKIHQDLGLT NHVPLL KKS LEQFVYRVKA MLAFNHCQEA FWVGVLKNRD LQGEEILSQA SAAPEEDSAE GSEEDTEDSA AEEPDGTDSD SGGAGRL EV LFQGPWSHPQ FEKGSAGSAA GSGAGWSHPQ FEK |
-Macromolecule #2: Fanconi anemia complementation group I
| Macromolecule | Name: Fanconi anemia complementation group I / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MAQRILQLAA EGSPERLQEA LQGLTEGELG DMVTRQALRG RETAALLKGI FKGSPCSQQS GVLRRLQVYK HCVSLVESGD LHVGKVSEI IGLLMLEARQ LPGHALAELA TLFVEVIKRG SLSNGKSLEL FSTVLTALSN SKESLAYGKG ELNGEEFKKQ L INTLCSSK ...String: MAQRILQLAA EGSPERLQEA LQGLTEGELG DMVTRQALRG RETAALLKGI FKGSPCSQQS GVLRRLQVYK HCVSLVESGD LHVGKVSEI IGLLMLEARQ LPGHALAELA TLFVEVIKRG SLSNGKSLEL FSTVLTALSN SKESLAYGKG ELNGEEFKKQ L INTLCSSK WDPQCVIHLA NMFRDIPLSG EELQFVVEKV LRMFSKLDLQ EIPPLVYQLL LLSAKGSKKT VLEGIISFFN QL DKRQKEE QRVPQSADLE VATVPLDQLR HVEGTVILHI VSAINLDQDI GEELIKHLKT EQQKDPGKAL CPFSVSLLLS TAV KHRLQE QIFDFLKTSI TRSCKDLQIL QASKFLQDLC PQQYDVTAVI LEVVKNSAFG WDHVTQGLVD LGFSLMESYE PKKS FGGKA AETNLGLSKM PAQQACKLGA SILLETFKVH EPIRSDILEQ VLNRVLTKAA SPVSHFIDLL SNIVVSAPLV LQNSS SRVT ETFDNLSFLP IDTVQGLLRA VQPLLKVSMS VRDSLILVLQ KAIFSRQLDA RKAAVAGFLL LLRNFKILGS LTSDQC DQA IGADQVQADV HACYNSAANE AFCLEILGSL RRCLSQQADV RLMLYEGFYD VLRRNSQLAS SIMETLLSQI KQYYLPQ QD LLPPLKLEGC IMAQGDQIFL QEPLAHLLCC IQHCLAWYKS TVHLCKGAED EEEEEDVGFE QNFEEMLESV TRRMIKSE L EDFELDKSAD FSPSSGVGVK NNIYAIQVMG ICEVLIEYNF KIGNFSKNKF EDVLGLFTCY NKLSEILKEK AGKNKSTLG NRIARSFLSM GFVSTLLTAL FRDNAQSHEE SLAVLRSSTE FMRYAVSVAL QKVQQLEEMG QTDGPDGQNP EKMFQNLCKI TRVLLWRYT SIPTAVEESG KKKGKSISLL CLEGLLRIFN TMQQLYAARI PQFLQALDIT DGDAEEADIN VTEKAAFQIR Q FQRSLVNQ LSSAEDDFNS KETQLLITIL STLSKLLDPG SQQFLQFLTW TVKICKENAL EDLSCCKGLL TLLFSLHVLY KS PVSLLRE LAQDIHACLG DIDQDVEIES RSHFAIVNVK TAAPTVCLLV LGQADKVLEE VDWLIKRLTI LGSDTSEDST QAS NQTQAL EKGVILQLGT LLTVFHELVQ TALPAGSCVD SLLRSLSKTY AILTSLIKHY IQACRSTSNT VPGRLEKLVK LSGS HLTPQ CYSFITYVQN IHSESLSFAE EKKKKKKEDE TAVVSTVMAK VLRDTKPIPN LIFAIEQYEK FLIHLSKKSK VNLMQ YMKL STSRDFRINA SMLDSVLQEQ NTEDAENEPD NNQSGTAEQP DENQEPQKKR RRKKHHHHHH |
-Macromolecule #5: Polyubiquitin-C
| Macromolecule | Name: Polyubiquitin-C / type: protein_or_peptide / ID: 5 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  |
| Sequence | String: MQIFVKTLTG KTITLEVEPS DTIENVKAKI QDKEGIPPDQ QRLIFAGKQL EDGRTLSDYN IQKESTLHLV LRLRGG |
-Macromolecule #3: DNA chain S
| Macromolecule | Name: DNA chain S / type: dna / ID: 3 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Sequence | String: (DA)(DT)(DC)(DG)(DA)(DT)(DG)(DT)(DC)(DT) (DC)(DT)(DA)(DG)(DA)(DC)(DA)(DG)(DC)(DT) (DG)(DC)(DT)(DC)(DA)(DG)(DG)(DA)(DT) (DT)(DG)(DA)(DT)(DC)(DT)(DG)(DT)(DA)(DA) (DT) (DG)(DG)(DC)(DC) |
-Macromolecule #4: DNA chain T
| Macromolecule | Name: DNA chain T / type: dna / ID: 4 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Sequence | String: (DG)(DG)(DC)(DC)(DA)(DT)(DT)(DA)(DC)(DA) (DG)(DA)(DT)(DC)(DA)(DA)(DT)(DC)(DC)(DT) (DG)(DA)(DG)(DC)(DA)(DG)(DC)(DT)(DG) (DT)(DC)(DT)(DA)(DG)(DA)(DG)(DA)(DC)(DA) (DT) (DC)(DG)(DA)(DT) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.5 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| |||||||||||||||
| Grid | Model: UltrAuFoil R0.6/1 / Pretreatment - Type: PLASMA CLEANING / Pretreatment - Time: 45 sec. | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 6515 / Average exposure time: 7.8 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)