[English] 日本語
 Yorodumi
Yorodumi- EMDB-8653: Cryo-EM structure of the activated Glucagon-like peptide-1 recept... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-8653 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the activated Glucagon-like peptide-1 receptor in complex with G protein | |||||||||
 Map data Map data | Structure of the activated Glucagon-like peptide-1 receptor in complex with G protein | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationglucagon receptor binding / glucagon-like peptide 1 receptor activity / glucagon receptor activity / positive regulation of blood pressure / negative regulation of execution phase of apoptosis / G-protein activation / Activation of the phototransduction cascade / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste ...glucagon receptor binding / glucagon-like peptide 1 receptor activity / glucagon receptor activity / positive regulation of blood pressure / negative regulation of execution phase of apoptosis / G-protein activation / Activation of the phototransduction cascade / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 / Ca2+ pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G alpha (z) signalling events / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / feeding behavior / Adrenaline,noradrenaline inhibits insulin secretion / ADP signalling through P2Y purinoceptor 12 / G alpha (q) signalling events / Thrombin signalling through proteinase activated receptors (PARs) / G alpha (i) signalling events / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Glucagon-type ligand receptors / G alpha (12/13) signalling events / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Adrenaline,noradrenaline inhibits insulin secretion / alkylglycerophosphoethanolamine phosphodiesterase activity / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Thrombin signalling through proteinase activated receptors (PARs) / Ca2+ pathway / Extra-nuclear estrogen signaling / G alpha (z) signalling events / G alpha (s) signalling events / G alpha (q) signalling events / photoreceptor outer segment membrane / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / spectrin binding / Vasopressin regulates renal water homeostasis via Aquaporins / positive regulation of calcium ion import / cellular response to glucagon stimulus / positive regulation of insulin secretion involved in cellular response to glucose stimulus / regulation of insulin secretion / peptide hormone binding / photoreceptor outer segment / Synthesis, secretion, and deacylation of Ghrelin / positive regulation of gluconeogenesis / cardiac muscle cell apoptotic process / protein kinase A signaling / photoreceptor inner segment / positive regulation of peptidyl-threonine phosphorylation / response to activity / gluconeogenesis / adenylate cyclase-activating G protein-coupled receptor signaling pathway / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / hormone activity / Glucagon signaling in metabolic regulation / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Glucagon-type ligand receptors / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / cellular response to catecholamine stimulus / sensory perception of taste / adenylate cyclase-activating dopamine receptor signaling pathway / cellular response to prostaglandin E stimulus / G-protein beta-subunit binding / heterotrimeric G-protein complex / signaling receptor complex adaptor activity / positive regulation of peptidyl-serine phosphorylation / GTPase binding / glucose homeostasis / retina development in camera-type eye / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / cellular response to hypoxia / cell body / G alpha (s) signalling events / G alpha (q) signalling events / secretory granule lumen / cell population proliferation / positive regulation of ERK1 and ERK2 cascade / cell surface receptor signaling pathway / G protein-coupled receptor signaling pathway / endoplasmic reticulum lumen / signaling receptor binding / GTPase activity / dendrite Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) / Homo sapiens (human) /    | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.1 Å | |||||||||
 Authors Authors | Zhang Y / Sun B / Feng D / Hu H / Chu M / Qu Q / Tarrasch JT / Li S / Kobilka TS / Kobilka BK / Skiniotis G | |||||||||
 Citation Citation |  Journal: Nature / Year: 2017 Journal: Nature / Year: 2017Title: Cryo-EM structure of the activated GLP-1 receptor in complex with a G protein. Authors: Yan Zhang / Bingfa Sun / Dan Feng / Hongli Hu / Matthew Chu / Qianhui Qu / Jeffrey T Tarrasch / Shane Li / Tong Sun Kobilka / Brian K Kobilka / Georgios Skiniotis /  Abstract: Glucagon-like peptide 1 (GLP-1) is a hormone with essential roles in regulating insulin secretion, carbohydrate metabolism and appetite. GLP-1 effects are mediated through binding to the GLP-1 ...Glucagon-like peptide 1 (GLP-1) is a hormone with essential roles in regulating insulin secretion, carbohydrate metabolism and appetite. GLP-1 effects are mediated through binding to the GLP-1 receptor (GLP-1R), a class B G-protein-coupled receptor (GPCR) that signals primarily through the stimulatory G protein G. Class B GPCRs are important therapeutic targets; however, our understanding of their mechanism of action is limited by the lack of structural information on activated and full-length receptors. Here we report the cryo-electron microscopy structure of the peptide-activated GLP-1R-G complex at near atomic resolution. The peptide is clasped between the N-terminal domain and the transmembrane core of the receptor, and further stabilized by extracellular loops. Conformational changes in the transmembrane domain result in a sharp kink in the middle of transmembrane helix 6, which pivots its intracellular half outward to accommodate the α5-helix of the Ras-like domain of G. These results provide a structural framework for understanding class B GPCR activation through hormone binding. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_8653.map.gz emd_8653.map.gz | 4.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-8653-v30.xml emd-8653-v30.xml emd-8653.xml emd-8653.xml | 17.6 KB 17.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_8653.png emd_8653.png | 36.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-8653 http://ftp.pdbj.org/pub/emdb/structures/EMD-8653 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8653 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8653 | HTTPS FTP |
-Validation report
| Summary document |  emd_8653_validation.pdf.gz emd_8653_validation.pdf.gz | 313.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_8653_full_validation.pdf.gz emd_8653_full_validation.pdf.gz | 313.4 KB | Display | |
| Data in XML |  emd_8653_validation.xml.gz emd_8653_validation.xml.gz | 5.3 KB | Display | |
| Data in CIF |  emd_8653_validation.cif.gz emd_8653_validation.cif.gz | 6.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8653 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8653 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8653 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8653 | HTTPS FTP |
-Related structure data
| Related structure data |  5vaiMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_8653.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_8653.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of the activated Glucagon-like peptide-1 receptor in complex with G protein | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
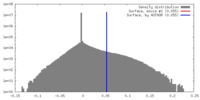
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : the activated glucagon-like peptide-1 receptor in complex with th...
| Entire | Name: the activated glucagon-like peptide-1 receptor in complex with the heterotrimeric Gs protein |
|---|---|
| Components |
|
-Supramolecule #1: the activated glucagon-like peptide-1 receptor in complex with th...
| Supramolecule | Name: the activated glucagon-like peptide-1 receptor in complex with the heterotrimeric Gs protein type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Molecular weight | Theoretical: 150 KDa |
-Macromolecule #1: Uncharacterized protein
| Macromolecule | Name: Uncharacterized protein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 52.581793 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDAAAGQPG NGSAFLLAPN RSHAPGGGGS LEVLFQGPGG GSRPQGATVS LSETVQKWRE YRRQCQHFL TEAPPLATGL FCNRTFDDYA CWPDGAPGSF VNVSCPWYLP WASNVLQGHV YRFCTAEGHW LPKDNSSLPW R DLSECEES ...String: MKTIIALSYI FCLVFADYKD DDDAAAGQPG NGSAFLLAPN RSHAPGGGGS LEVLFQGPGG GSRPQGATVS LSETVQKWRE YRRQCQHFL TEAPPLATGL FCNRTFDDYA CWPDGAPGSF VNVSCPWYLP WASNVLQGHV YRFCTAEGHW LPKDNSSLPW R DLSECEES RRGEKSSPEE RLLSLYIIYT VGYALSFSAL VIASAILLGF RHLHCTRNYI HLNLFASFIL RALSVFIKDA AL KWMYSTA AQQHQWDGLL SYQDSLGCRL VFLLMQYCVA ANYYWLLVEG AYLYTLLAFA VFSEQRIFKL YLSIGWGVPL LFV IPWGIV KYLYEDEGCW TRNSNMNYWL IIRLPILFAI GVNFLIFIRV ICIVVSKLKA NLMCKTDIKC RLAKSTLTLI PLLG THEVI FAFVMDEHAR GTLRFVKLFT ELSFTSFQGL MVAILYCFVN NEVQMEFRKS WERWRL |
-Macromolecule #2: Glucagon-like peptide 1
| Macromolecule | Name: Glucagon-like peptide 1 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 3.359699 KDa |
| Sequence | String: HAEGTFTSDV SSYLEGQAAK EFIAWLVKGR G |
-Macromolecule #3: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 44.370168 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG DSEKATKVQD IKNNLKEAI ETIVAAMSNL VPPVELANPE NQFRVDYILS VMNVPDFDFP PEFYEHAKAL WEDEGVRACY ERSNEYQLID C AQYFLDKI ...String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG DSEKATKVQD IKNNLKEAI ETIVAAMSNL VPPVELANPE NQFRVDYILS VMNVPDFDFP PEFYEHAKAL WEDEGVRACY ERSNEYQLID C AQYFLDKI DVIKQDDYVP SDQDLLRCRV LTSGIFETKF QVDKVNFHMF DVGGQRDERR KWIQCFNDVT AIIFVVASSS YN MVIREDN QTNRLQEALN LFKSIWNNRW LRTISVILFL NKQDLLAEKV LAGKSKIEDY FPEFARYTTP EDATPEPGED PRV TRAKYF IRDEFLRIST ASGDGRHYCY PHFTCAVDTE NIRRVFNDCR DIIQRMHLRQ YELL |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 38.744371 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MHHHHHHGSL LQSELDQLRQ EAEQLKNQIR DARKACADAT LSQITNNIDP VGRIQMRTRR TLRGHLAKIY AMHWGTDSRL LVSASQDGK LIIWDSYTTN KVHAIPLRSS WVMTCAYAPS GNYVACGGLD NICSIYNLKT REGNVRVSRE LAGHTGYLSC C RFLDDNQI ...String: MHHHHHHGSL LQSELDQLRQ EAEQLKNQIR DARKACADAT LSQITNNIDP VGRIQMRTRR TLRGHLAKIY AMHWGTDSRL LVSASQDGK LIIWDSYTTN KVHAIPLRSS WVMTCAYAPS GNYVACGGLD NICSIYNLKT REGNVRVSRE LAGHTGYLSC C RFLDDNQI VTSSGDTTCA LWDIETGQQT TTFTGHTGDV MSLSLAPDTR LFVSGACDAS AKLWDVREGM CRQTFTGHES DI NAICFFP NGNAFATGSD DATCRLFDLR ADQELMTYSH DNIICGITSV SFSKSGRLLL AGYDDFNCNV WDALKADRAG VLA GHDNRV SCLGVTDDGM AVATGSWDSF LKIWN |
-Macromolecule #5: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 7.56375 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFC |
-Macromolecule #6: nanobody 35
| Macromolecule | Name: nanobody 35 / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 15.140742 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: QVQLQESGGG LVQPGGSLRL SCAASGFTFS NYKMNWVRQA PGKGLEWVSD ISQSGASISY TGSVKGRFTI SRDNAKNTLY LQMNSLKPE DTAVYYCARC PAPFTRDCFD VTSTTYAYRG QGTQVTVSSH HHHHHEPEA |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 90.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 50000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal magnification: 29000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)