+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-8514 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | 3D reconstruction of the CaMKIIa holoenzyme. | |||||||||
 Map data Map data | CaMKII holoenzyme structure resolved by negative stain EM. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Calcium/calmodulin-dependent kinase II (CaMKII) / cell signaling / calcium / calmodulin (CaM) / long-term potentiation (LTP) / long-term depression (LTD) / synaptic plasticity / cooperativity / electron microscopy (EM) / single particle reconstruction / intrinsic disorder / TRANSFERASE | |||||||||
| Function / homology | :  Function and homology information Function and homology information | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 20.0 Å | |||||||||
 Authors Authors | Myers J / Reichow SL | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2017 Journal: Nat Commun / Year: 2017Title: The CaMKII holoenzyme structure in activation-competent conformations. Authors: Janette B Myers / Vincent Zaegel / Steven J Coultrap / Adam P Miller / K Ulrich Bayer / Steve L Reichow /  Abstract: The Ca/calmodulin-dependent protein kinase II (CaMKII) assembles into large 12-meric holoenzymes, which is thought to enable regulatory processes required for synaptic plasticity underlying learning, ...The Ca/calmodulin-dependent protein kinase II (CaMKII) assembles into large 12-meric holoenzymes, which is thought to enable regulatory processes required for synaptic plasticity underlying learning, memory and cognition. Here we used single particle electron microscopy (EM) to determine a pseudoatomic model of the CaMKIIα holoenzyme in an extended and activation-competent conformation. The holoenzyme is organized by a rigid central hub complex, while positioning of the kinase domains is highly flexible, revealing dynamic holoenzymes ranging from 15-35 nm in diameter. While most kinase domains are ordered independently, ∼20% appear to form dimers and <3% are consistent with a compact conformation. An additional level of plasticity is revealed by a small fraction of bona-fide 14-mers (<4%) that may enable subunit exchange. Biochemical and cellular FRET studies confirm that the extended state of CaMKIIα resolved by EM is the predominant form of the holoenzyme, even under molecular crowding conditions. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_8514.map.gz emd_8514.map.gz | 7.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-8514-v30.xml emd-8514-v30.xml emd-8514.xml emd-8514.xml | 13.8 KB 13.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_8514.png emd_8514.png | 30.8 KB | ||
| Filedesc metadata |  emd-8514.cif.gz emd-8514.cif.gz | 6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-8514 http://ftp.pdbj.org/pub/emdb/structures/EMD-8514 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8514 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8514 | HTTPS FTP |
-Validation report
| Summary document |  emd_8514_validation.pdf.gz emd_8514_validation.pdf.gz | 370.6 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_8514_full_validation.pdf.gz emd_8514_full_validation.pdf.gz | 370.1 KB | Display | |
| Data in XML |  emd_8514_validation.xml.gz emd_8514_validation.xml.gz | 4.9 KB | Display | |
| Data in CIF |  emd_8514_validation.cif.gz emd_8514_validation.cif.gz | 5.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8514 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8514 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8514 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8514 | HTTPS FTP |
-Related structure data
| Related structure data |  5u6yMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_8514.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_8514.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | CaMKII holoenzyme structure resolved by negative stain EM. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 4.37 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
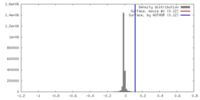
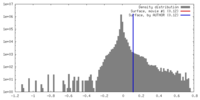
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Calcium-calmodulin dependent kinase II alpha
| Entire | Name: Calcium-calmodulin dependent kinase II alpha |
|---|---|
| Components |
|
-Supramolecule #1: Calcium-calmodulin dependent kinase II alpha
| Supramolecule | Name: Calcium-calmodulin dependent kinase II alpha / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all / Details: Full-length CaMKII alpha wild type |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 620 KDa |
-Macromolecule #1: Calcium/calmodulin-dependent protein kinase type II subunit alpha
| Macromolecule | Name: Calcium/calmodulin-dependent protein kinase type II subunit alpha type: protein_or_peptide / ID: 1 / Number of copies: 12 / Enantiomer: LEVO / EC number: Ca2+/calmodulin-dependent protein kinase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 52.281535 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MYQLFEELGK GAFSVVRRCV KVLAGQEYAA KIINTKKLSA RDHQKLEREA RICRLLKHPN IVRLHDSISE EGHHYLIFDL VTGGELFED IVAREYYSEA DASHCIQQIL EAVLHCHQMG VVHRDLKPEN LLLASKLKGA AVKLADFGLA IEVEGEQQAW F GFAGTPGY ...String: MYQLFEELGK GAFSVVRRCV KVLAGQEYAA KIINTKKLSA RDHQKLEREA RICRLLKHPN IVRLHDSISE EGHHYLIFDL VTGGELFED IVAREYYSEA DASHCIQQIL EAVLHCHQMG VVHRDLKPEN LLLASKLKGA AVKLADFGLA IEVEGEQQAW F GFAGTPGY LSPEVLRKDP YGKPVDLWAC GVILYILLVG YPPFWDEDQH RLYQQIKAGA YDFPSPEWDT VTPEAKDLIN KM LTINPSK RITAAEALKH PWISHRSTVA SCMHRQETVD CLKKFNARRK LKGAILTTML ATRNFSGGKS GGNKKSDGVK ESS ESTNTT IEDEDTKVRK QEIIKVTEQL IEAISNGDFE SYTKMCDPGM TAFEPEALGN LVEGLDFHRF YFENLWSRNS KPVH TTILN PHIHLMGDES ACIAYIRITQ YLDAGGIPRT AQSEETRVWH RRDGKWQIVH FHRSGA UniProtKB: UNIPROTKB: P11275 |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 Component:
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Staining | Type: NEGATIVE / Material: Uranyl Formate / Details: 0.75% (wt/vol) uranyl formate | ||||||||||||
| Grid | Model: Ted Pella / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 20 sec. | ||||||||||||
| Details | 100 nM complex |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI 12 |
|---|---|
| Image recording | Film or detector model: FEI EAGLE (2k x 2k) / Average exposure time: 1.0 sec. / Average electron dose: 20.0 e/Å2 |
| Electron beam | Acceleration voltage: 120 kV / Electron source: TUNGSTEN HAIRPIN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal magnification: 49000 |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Protocol: RIGID BODY FIT / Target criteria: Correlation Coefficient | ||||||
| Output model |  PDB-5u6y: |
 Movie
Movie Controller
Controller



 UCSF Chimera
UCSF Chimera

 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)