+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7pbd | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | a1b3 GABA-A receptor + GABA | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN / pLGIC GABA Neurotransmission | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationcircadian sleep/wake cycle, REM sleep / reproductive behavior / GABA receptor complex / hard palate development / cellular response to histamine / GABA receptor activation / inner ear receptor cell development / inhibitory synapse assembly / GABA-A receptor activity / GABA-gated chloride ion channel activity ...circadian sleep/wake cycle, REM sleep / reproductive behavior / GABA receptor complex / hard palate development / cellular response to histamine / GABA receptor activation / inner ear receptor cell development / inhibitory synapse assembly / GABA-A receptor activity / GABA-gated chloride ion channel activity / GABA-A receptor complex / innervation / response to anesthetic / postsynaptic specialization membrane / inhibitory postsynaptic potential / gamma-aminobutyric acid signaling pathway / synaptic transmission, GABAergic / cellular response to zinc ion / exploration behavior / motor behavior / roof of mouth development / Signaling by ERBB4 / cochlea development / social behavior / chloride channel complex / dendrite membrane / chloride transmembrane transport / cytoplasmic vesicle membrane / cerebellum development / learning / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / GABA-ergic synapse / memory / dendritic spine / postsynaptic membrane / postsynapse / response to xenobiotic stimulus / cell surface / signal transduction / identical protein binding / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.04 Å | ||||||||||||
 Authors Authors | Miller, P.S. / Kasaragod, V.B. | ||||||||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||||||||
 Citation Citation |  Journal: Nature / Year: 2022 Journal: Nature / Year: 2022Title: Mechanisms of inhibition and activation of extrasynaptic αβ GABA receptors. Authors: Vikram Babu Kasaragod / Martin Mortensen / Steven W Hardwick / Ayla A Wahid / Valentina Dorovykh / Dimitri Y Chirgadze / Trevor G Smart / Paul S Miller /  Abstract: Type A GABA (γ-aminobutyric acid) receptors represent a diverse population in the mammalian brain, forming pentamers from combinations of α-, β-, γ-, δ-, ε-, ρ-, θ- and π-subunits. αβ, ...Type A GABA (γ-aminobutyric acid) receptors represent a diverse population in the mammalian brain, forming pentamers from combinations of α-, β-, γ-, δ-, ε-, ρ-, θ- and π-subunits. αβ, α4βδ, α6βδ and α5βγ receptors favour extrasynaptic localization, and mediate an essential persistent (tonic) inhibitory conductance in many regions of the mammalian brain. Mutations of these receptors in humans are linked to epilepsy and insomnia. Altered extrasynaptic receptor function is implicated in insomnia, stroke and Angelman and Fragile X syndromes, and drugs targeting these receptors are used to treat postpartum depression. Tonic GABAergic responses are moderated to avoid excessive suppression of neuronal communication, and can exhibit high sensitivity to Zn blockade, in contrast to synapse-preferring α1βγ, α2βγ and α3βγ receptor responses. Here, to resolve these distinctive features, we determined structures of the predominantly extrasynaptic αβ GABA receptor class. An inhibited state bound by both the lethal paralysing agent α-cobratoxin and Zn was used in comparisons with GABA-Zn and GABA-bound structures. Zn nullifies the GABA response by non-competitively plugging the extracellular end of the pore to block chloride conductance. In the absence of Zn, the GABA signalling response initially follows the canonical route until it reaches the pore. In contrast to synaptic GABA receptors, expansion of the midway pore activation gate is limited and it remains closed, reflecting the intrinsic low efficacy that characterizes the extrasynaptic receptor. Overall, this study explains distinct traits adopted by αβ receptors that adapt them to a role in tonic signalling. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7pbd.cif.gz 7pbd.cif.gz | 361.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7pbd.ent.gz pdb7pbd.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  7pbd.json.gz 7pbd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pb/7pbd https://data.pdbj.org/pub/pdb/validation_reports/pb/7pbd ftp://data.pdbj.org/pub/pdb/validation_reports/pb/7pbd ftp://data.pdbj.org/pub/pdb/validation_reports/pb/7pbd | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  13290MC  7pbzC  7pc0C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Gamma-aminobutyric acid receptor subunit alpha- ... , 2 types, 2 molecules AD
| #1: Protein | Mass: 41991.133 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GABRA1 / Production host: Homo sapiens (human) / Gene: GABRA1 / Production host:  Homo sapiens (human) / References: UniProt: P14867 Homo sapiens (human) / References: UniProt: P14867 |
|---|---|
| #3: Protein | Mass: 47059.648 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GABRA1 / Production host: Homo sapiens (human) / Gene: GABRA1 / Production host:  Homo sapiens (human) / References: UniProt: P14867 Homo sapiens (human) / References: UniProt: P14867 |
-Protein / Antibody , 2 types, 4 molecules BCEF
| #2: Protein | Mass: 51615.117 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GABRB3 / Production host: Homo sapiens (human) / Gene: GABRB3 / Production host:  Homo sapiens (human) / References: UniProt: P28472 Homo sapiens (human) / References: UniProt: P28472#4: Antibody | | Mass: 56329.621 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Sugars , 3 types, 8 molecules 
| #5: Polysaccharide | Source method: isolated from a genetically manipulated source #6: Polysaccharide | #8: Sugar | |
|---|
-Non-polymers , 2 types, 3 molecules 
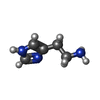

| #7: Chemical | | #9: Chemical | ChemComp-HSM / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.22 MDa / Experimental value: NO | |||||||||||||||||||||||||
| Source (natural) |
| |||||||||||||||||||||||||
| Source (recombinant) |
| |||||||||||||||||||||||||
| Buffer solution | pH: 7.4 | |||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||
| Specimen | Conc.: 0.25 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | |||||||||||||||||||||||||
| Specimen support | Grid material: GOLD / Grid type: UltrAuFoil R1.2/1.3 | |||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 95 % / Chamber temperature: 287 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: DIFFRACTION / Nominal magnification: 130000 X / Nominal defocus max: 2500 nm / Nominal defocus min: 1000 nm / C2 aperture diameter: 50 µm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 54.7 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
| 3D reconstruction | Resolution: 3.04 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 139537 / Symmetry type: POINT |
 Movie
Movie Controller
Controller















 PDBj
PDBj







